Abstract
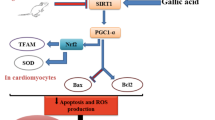
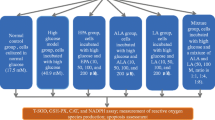
Epidemiological studies have shown that high glucose levels and oxidative stress cause elevation of advanced glycation end products (AGEs) that are known to contribute to diabetic complications. Thus, agents that hamper reactive oxygen species (ROS) load can be used as a potential drug against AGEs-mediated complications. Hence, the present study investigated the protective role of gallic acid (GA) against the effects of AGEs in cardiac H9C2(2-1) cells. Exposure of cells to AGEs resulted in release of ROS (P < 0.05) with significant (P < 0.05) decline in antioxidant enzyme levels and increase in collagen (P < 0.01) content. In addition, the altered mitochondrial membrane potential (mmp) (P < 0.01) was also observed in cells exposed to AGEs, whereas AGEs-exposed cells pretreated with GA prevented the release of ROS, and there were no significant changes in the antioxidant status, collagen content and mmp. Thus, the results of the present study provide evidence that GA exhibits protective role against AGEs-induced cardiovascular complications probably through its free radical scavenging activity.






Similar content being viewed by others
References
Ahmed, N. (2005). Advanced glycation end products role in pathology of diabetic complications. Diabetes Research and Clinical Practice, 67, 3–21.
Forbes, J. M., Coughlan, M. T., & Cooper, M. E. (2008). Oxidative stress as a major culprit in kidney disease in diabetes. Diabetes, 57, 1446–1454.
Schleicher, E., & Friess, U. (2007). Oxidative stress, AGE, and atherosclerosis. Kidney International, Supplement 106, S17–S26.
Bierhaus, A., Humpert, P. M., Morcos, M., Wendt, T., Chavakis, T., Arnold, B., et al. (2005). Understanding RAGE, the receptor for advanced glycation end products. Journal of molecular medicine (Berlin, Germany), 83, 876–886.
Basta, G., Schmidt, A. M., & De Caterina, R. (2004). Advanced glycation end products and vascular inflammation: Implications for accelerated atherosclerosis in diabetes. Cardiovascular Research, 63, 582–592.
Farrar, J. L., Hartle, D. K., Hargrove, J. L., & Greenspan, P. (2007). Inhibition of protein glycation by skins and seeds of the muscadine grape. BioFactors, 30, 193–200.
Su-Chen, H., Szu-Pei, W., Shyh-Mirn, L., & Ya-Li, T. (2010). Comparison of anti-glycation capacities of several herbal infusions with that of green tea. Food Chemistry, 122, 768–774.
Priscilla, D. H., & Prince, P. S. (2009). Cardioprotective effect of gallic acid on cardiac troponin-T, cardiac marker enzymes, lipid peroxidation products and antioxidants in experimentally induced myocardial infarction in Wistar rats. Chemico-Biological Interactions, 179, 118–124.
Shahrzad, S., Aoyagi, K., Winter, A., Koyama, A., & Bitsch, I. (2001). Pharmacokinetics of gallic acid and its relative bioavailability from tea in healthy humans. The Journal of Nutrition, 131, 1207–1210.
Andreea, S. I., Marieta, C., & Anca, D. (2008). AGEs and glucose levels modulate type I and III procollagen mRNA synthesis in dermal fibroblasts cells culture. Experimental Diabetes Research, 2008, 473603.
Ewing, J. F., & Janero, D. R. (1995). Microplate superoxide dismutase assay employing a nonenzymatic superoxide generator. Analytical Biochemistry, 232, 243–248.
Aebi, H., Suter, H., & Feinstein, R. N. (1968). Activity and stability of catalase in blood and tissues of normal and acatalasemic mice. Biochemical Genetics, 2, 245–251.
Goldin, A., Beckman, J. A., Schmidt, A. M., & Creager, M. A. (2006). Advanced glycation end products: Sparking the development of diabetic vascular injury. Circulation, 8, 597–605.
Wheeler, M. T., & McNally, E. M. (2005). The interaction of coronary tone and cardiac fibrosis. Current Atherosclerosis Reports, 7, 219–226.
Lee, H. B., Yu, M. R., Yang, Y., Jiang, Z., & Ha, H. (2003). Reactive oxygen species-regulated signaling pathways in diabetic nephropathy. Journal of the American Society of Nephrology, 14, S241–S245.
Ha, H., & Lee, H. B. (2001). Oxidative stress in diabetic nephropathy: Basic and clinical information. Current Diabetes Reports, 1, 282–287.
Franke, S., Sommer, M., Rüster, C., Bondeva, T., Marticke, J., Hofmann, G., et al. (2009). Advanced glycation end products induce cell cycle arrest and proinflammatory changes in osteoarthritic fibroblast-like synovial cells. Arthritis Research & Therapy, 11, R136.
Han, H., Wang, H., Long, H., Nattel, S., & Wang, Z. (2001). Oxidative preconditioning and apoptosis in L-cells. Roles of protein kinase B and mitogen-activated protein kinases. The Journal of biological chemistry, 276, 26357–26364.
Kulisz, A., Chen, N., Chandel, N. S., Shao, Z., & Schumacker, P. T. (2002). Mitochondrial ROS initiate phosphorylation of p38 MAP kinase during hypoxia in cardiomyocytes. American journal of physiology. Lung cellular and molecular physiology, 282, L1324–L1329.
Han, H., Long, H., Wang, H., Wang, J., Zhang, Y., & Wang, Z. (2004). Progressive apoptotic cell death triggered by transient oxidative insult in H9c2 rat ventricular cells: A novel pattern of apoptosis and the mechanisms. American Journal of Physiology. Heart and Circulatory Physiology, 286, H2169–H2182.
Bashan, N., Kovsan, J., Kachko, I., Ovadia, H., & Rudich, A. (2009). Positive and negative regulation of insulin signaling by reactive oxygen and nitrogen species. Physiological Reviews, 89, 27–71.
Tsutsui, H., Kinugawa, S., & Matsushima, S. (2009). Mitochondrial oxidative stress and dysfunction in myocardial remodelling. Cardiovascular Research, 81, 449–456.
Johansen, J. S., Harris, A. K., Rychly, D. J., & Ergul, A. (2005). Oxidative stress and the use of antioxidants in diabetes: Linking basic science to clinical practice. Cardiovascular Diabetology, 29, 4–5.
Niedowicz, D. M., & Daleke, D. L. (2005). The role of oxidative stress in diabetic complications. Cell Biochemistry and Biophysics, 43, 289–330.
Kuppan, G., Balasubramanyam, J., Monickaraj, F., Srinivasan, G., Mohan, V., & Balasubramanyam, M. (2010). Transcriptional regulation of cytokines and oxidative stress by gallic acid in human THP-1 monocytes. Cytokine, 49, 229–234.
Camelliti, P., Borg, T. K., & Kohl, P. (2005). Structural and functional characterization of cardiac fibroblasts. Cardiovascular Research, 65, 40–51.
Chen, K., Chen, J., Li, D., Zhang, X., & Mehta, J. L. (2004). Angiotensin II regulation of collagen type I expression in cardiac fibroblasts: Modulation by PPAR-gamma ligand pioglitazone. Hypertension, 44, 655–661.
Thallas-Bonke, V., Thorpe, S. R., Coughlan, M. T., Fukami, K., Yap, F. Y., Sourris, K. C., et al. (2008). Inhibition of NADPH oxidase prevents advanced glycation end product-mediated damage in diabetic nephropathy through a protein kinase C-alpha-dependent pathway. Diabetes, 57, 460–469.
Li, Y. Y., Feng, Y., McTiernan, C. F., Pei, W., Moravec, C. S., Wang, P., et al. (2001). Downregulation of matrix metalloproteinases and reduction in collagen damage in the failing human heart after support with left ventricular assist devices. Circulation, 104, 1147–1152.
Lijnen, P., Papparella, I., Petrov, V., Semplicini, A., & Fagard, R. (2006). Angiotensin II-stimulated collagen production in cardiac fibroblasts is mediated by reactive oxygen species. Journal of Hypertension, 24, 757–766.
Acknowledgments
This work was supported by UGC, JNMF and CSIR, New Delhi, India.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Umadevi, S., Gopi, V., Simna, S.P. et al. Studies on the Cardio Protective Role of Gallic Acid Against AGE-Induced Cell Proliferation and Oxidative Stress in H9C2 (2-1) Cells. Cardiovasc Toxicol 12, 304–311 (2012). https://doi.org/10.1007/s12012-012-9170-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-012-9170-2