Abstract
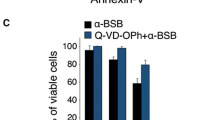
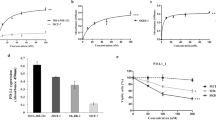
Breast cancer is the most frequently diagnosed cancer among women worldwide. Despite the initial clinical response obtained with the widely used conventional chemotherapy, an improved prognosis for breast cancer patients has been missing in the clinic because of the high toxicity to normal cells, induction of drug resistance, and the potential immunosuppressive effects of these agents. Therefore, we aimed to investigate the potential anti-carcinogenic effect of some boron derivatives (sodium pentaborate pentahydrate (SPP) and sodium perborate tetrahydrate (SPT)), which showed a promising effect on some types of cancers in the literature, on breast cancer cell lines, as well as immuno-oncological side effects on tumor-specific T cell activity. These findings suggest that both SPP and SPT suppressed proliferation and induced apoptosis in MCF7 and MDA-MB-231 cancer cell lines through downregulation of the monopolar spindle-one-binder (MOB1) protein. On the other hand, these molecules increased the expression of PD-L1 protein through their effect on the phosphorylation level of Yes-associated protein (Phospho-YAP (Ser127). In addition, they reduced the concentrations of pro-inflammatory cytokines such as IFN-γ and cytolytic effector cytokines such as sFasL, perforin, granzyme A, Granzyme B, and granulysin and increased the expression of PD-1 surface protein in activated T cells. In conclusion, SPP, SPT, and their combination could have growth inhibitory (antiproliferative) effects and could be a potential treatment for breast cancer. However, their stimulatory effects on the PD-1/PD-L1 signaling pathway and their effects on cytokines could ultimately account for the observed repression of the charging of specifically activated effector T cells against breast cancer cells.






Similar content being viewed by others
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
References
World Health Organization. (n.d.). Cancer. Retrieved March 18, 2023, from http://www.who.int/news-room/fact-sheets/detail/cancer
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65(2):87–108. https://doi.org/10.3322/caac.21262
Dizon DS, Krilov L, Cohen E et al (2016) Clinical Cancer Advances 2016: Annual Report on Progress Against Cancer From the American Society of Clinical Oncology. J Clin Oncol 34(9):987–1011. https://doi.org/10.1200/jco.2015.65.8427
Masood S (2016) Neoadjuvant chemotherapy in breast cancers. Womens Health (Lond) 12(5):480–491. https://doi.org/10.1177/1745505716677139
Uluisik I, Karakaya HC, Koc A (2018) The importance of boron in biological systems. Journal of Trace Elem Med Biol 45:156–162. https://doi.org/10.1016/j.jtemb.2017.10.008
Khaliq H, Juming Z, Ke-Mei P (2018) The physiological role of boron on health. Biological Trace Element Research 186(1):31–51. https://doi.org/10.1007/s12011-018-1284-3
Scorei RI, Popa R Jr (2010) Boron-containing compounds as preventive and chemotherapeutic agents for cancer. Anticancer Agents Med Chem 10(4):346–351. https://doi.org/10.2174/187152010791162289
Romero-Aguilar KS, Arciniega-Martínez IM, Farfán-García ED, Campos-Rodríguez R, Reséndiz-Albor AA, Soriano-Ursúa MA (2019) Effects of boron-containing compounds on immune responses: review and patenting trends. Expert Opin Ther Pat 29(5):339–351. https://doi.org/10.1080/13543776.2019.1612368
Wang Y, Zhao Y, Chen X (2008) Experimental study on the estrogen-like effect of boric acid. Biol Trace Elem Res 121(2):160–170. https://doi.org/10.1007/s12011-007-8041-3
Palumbo A, Gay F, Bringhen S et al (2008) Bortezomib, doxorubicin and dexamethasone in advanced multiple myeloma. Ann Oncol: Off J Eur Soc Med Oncol 19(6):1160–1165. https://doi.org/10.1093/annonc/mdn018
Teicher BA, Ara G, Herbst R, Palombella VJ, Adams J (1999) The proteasome inhibitor PS-341 in cancer therapy. Clin Cancer Res : an Off J Am Assoc Cancer Res 5(9):2638–2645
Gatto S, Scappini B, Pham L et al (2003) The proteasome inhibitor PS-341 inhibits growth and induces apoptosis in Bcr/Abl-positive cell lines sensitive and resistant to imatinib mesylate. Haematologica 88(8):853–863
Frankel A, Man S, Elliott P, Adams J, Kerbel RS (2000) Lack of multicellular drug resistance observed in human ovarian and prostate carcinoma treated with the proteasome inhibitor PS-341. Clin Cancer Res : Off J Am Assoc Cancer Res 6(9):3719–3728
Nawrocki ST, Carew JS, Pino MS et al (2006) Aggresome disruption: a novel strategy to enhance bortezomib-induced apoptosis in pancreatic cancer cells. Can Res 66(7):3773–3781. https://doi.org/10.1158/0008-5472.can-05-2961
Sunwoo JB, Chen Z, Dong G et al (2001) Novel proteasome inhibitor PS-341 inhibits activation of nuclear factor-kappa B, cell survival, tumor growth, and angiogenesis in squamous cell carcinoma. Clin Cancer Res : Off J Am Assoc Cancer Res 7(5):1419–1428
Barth RF, Coderre JA, Vicente MGaH, Blue TE (2005) Boron neutron capture therapy of cancer: current status and future prospects. Clin Cancer Res 11(11):3987–4002. https://doi.org/10.1158/1078-0432.ccr-05-0035
Gadan MA, González SJ, Batalla M, Olivera MS, Policastro L, Sztejnberg ML (2015) Reprint of Application of BNCT to the treatment of HER2+ breast cancer recurrences: research and developments in Argentina. Appl Radiat Isotopes 106:260–264. https://doi.org/10.1016/j.apradiso.2015.10.009
Cebeci E, Yüksel B, Şahin F (2022) Anti-cancer effect of boron derivatives on small-cell lung cancer. J Trace Elem Med Biol 70:126923. https://doi.org/10.1016/j.jtemb.2022.126923
Gündüz MK, Bolat M, Kaymak G, Berikten D, Köse DA (2022) Therapeutic effects of newly synthesized boron compounds (BGM and BGD) on hepatocellular carcinoma. Biol Trace Elem Res 200(1):134–146. https://doi.org/10.1007/s12011-021-02647-9
Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD (2002) Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol 3(11):991–998. https://doi.org/10.1038/ni1102-991
Standish LJ, Sweet ES, Novack J et al (2008) Breast cancer and the immune system. J Soc Integr Oncol 6(4):158–68
Dunn GP, Old LJ, Schreiber RD (2004) The three Es of cancer immunoediting. Annu Rev Immunol 22:329–360. https://doi.org/10.1146/annurev.immunol.22.012703.104803
Manjili MH, Payne KK (2012) Cancer immunotherapy: re-programming cells of the innate and adaptive immune systems. Oncoimmunology 1(2):201–204. https://doi.org/10.4161/onci.1.2.18113
Latchman Y, Wood CR, Chernova T et al (2001) PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat Immunol 2(3):261–268. https://doi.org/10.1038/85330
Prasad DV, Richards S, Mai XM, Dong C (2003) B7S1, a novel B7 family member that negatively regulates T cell activation. Immunity 18(6):863–873. https://doi.org/10.1016/s1074-7613(03)00147-x
Greenwald RJ, Freeman GJ, Sharpe AH (2005) The B7 family revisited. Annu Rev Immunol 23:515–548. https://doi.org/10.1146/annurev.immunol.23.021704.115611
Dai S, Jia R, Zhang X, Fang Q, Huang L (2014) The PD-1/PD-Ls pathway and autoimmune diseases. Cell Immunol 290(1):72–79. https://doi.org/10.1016/j.cellimm.2014.05.006
Ghebeh H, Tulbah A, Mohammed S et al (2007) Expression of B7–H1 in breast cancer patients is strongly associated with high proliferative Ki-67-expressing tumor cells. Int J Cancer J Int du Cancer 121:751–8. https://doi.org/10.1002/ijc.22703
Zheng Y, Fang YC, Li J (2019) PD-L1 expression levels on tumor cells affect their immunosuppressive activity. Oncol Lett 18(5):5399–5407. https://doi.org/10.3892/ol.2019.10903
Mandai M (2016) PD-1/PD-L1 blockage in cancer treatment-from basic research to clinical application. Int J Clin Oncol 21(3):447. https://doi.org/10.1007/s10147-016-0969-x
Saleh R, Toor SM, Khalaf S, Elkord E (2019) Breast Cancer Cells and PD-1/PD-L1 blockade upregulate the expression of PD-1, CTLA-4, TIM-3 and LAG-3 immune checkpoints in CD4+ T cells. Vaccines 7(4):149. https://doi.org/10.3390/vaccines7040149
Grubczak K, Kretowska-Grunwald A, Groth D et al (2021) Differential response of MDA-MB-231 and MCF-7 breast cancer cells to in vitro inhibition with CTLA-4 and PD-1 through cancer-immune cells modified interactions. Cells 10(8):2044
Ghebeh H, Lehe C, Barhoush E et al (2010) Doxorubicin downregulates cell surface B7–H1 expression and upregulates its nuclear expression in breast cancer cells: role of B7–H1 as an anti-apoptotic molecule. Breast Cancer Res : BCR 12(4):R48. https://doi.org/10.1186/bcr2605
Luo M, Fu L (2016) The effect of chemotherapy on programmed cell death 1/programmed cell death 1 ligand axis: some chemotherapeutical drugs may finally work through immune response. Oncotarget 7(20):29794–803. https://doi.org/10.18632/oncotarget.7631
Planes-Laine G, Rochigneux P, Bertucci F et al (2019) PD-1/PD-L1 targeting in breast cancer: the first clinical evidences are emerging. A Lit Rev Cancers 11(7):1033. https://doi.org/10.3390/cancers11071033
Adams S, Schmid P, Rugo HS et al (2019) Pembrolizumab monotherapy for previously treated metastatic triple-negative breast cancer: cohort A of the phase II KEYNOTE-086 study. Annals Oncol : Off J Eur Soc Med Oncol 30(3):397–404. https://doi.org/10.1093/annonc/mdy517
Mao X, Li P, Wang Y et al (2017) CRB3 regulates contact inhibition by activating the Hippo pathway in mammary epithelial cells. Cell Death Dis 8:e2546. https://doi.org/10.1038/cddis.2016.478
Pan D (2010) The hippo signaling pathway in development and cancer. Dev Cell 19(4):491–505. https://doi.org/10.1016/j.devcel.2010.09.011
Plouffe SW, Hong AW, Guan KL (2015) Disease implications of the Hippo/YAP pathway. Trends Mol Med 21(4):212–222. https://doi.org/10.1016/j.molmed.2015.01.003
Wu S, Liu Y, Zheng Y, Dong J, Pan D (2008) The TEAD/TEF family protein Scalloped mediates transcriptional output of the Hippo growth-regulatory pathway. Dev Cell 14(3):388–398. https://doi.org/10.1016/j.devcel.2008.01.007
Kim MH, Kim CG, Kim S-K et al (2018) YAP-induced PD-L1 expression drives immune evasion in BRAFi-resistant melanoma. Cancer Immunol Res 6(3):255–266. https://doi.org/10.1158/2326-6066.Cir-17-0320
Miao J, Hsu PC, Yang YL et al (2017) YAP regulates PD-L1 expression in human NSCLC cells. Oncotarget 8(70):114576–114587. https://doi.org/10.18632/oncotarget.23051
Janse van Rensburg HJ, Azad T, Ling M et al (2018) The Hippo pathway component TAZ promotes immune evasion in human cancer through PD-L1. Cancer Res 78(6):1457–1470. https://doi.org/10.1158/0008-5472.can-17-3139
Tait JF, Gibson D, Fujikawa K (1989) Phospholipid binding properties of human placental anticoagulant protein-I, a member of the lipocortin family. Research Support, U S Gov’t P H S. J Biol Chem 264(14):7944–9
Rosenblatt J, Wu Z, Vasir B et al (2010) Generation of tumor-specific T lymphocytes using dendritic cell/tumor fusions and anti-CD3/CD28. J Immunother (Hagerstown, Md : 1997) 33(2):155–66. https://doi.org/10.1097/CJI.0b013e3181bed253
Grubczak K, Kretowska-Grunwald A, Groth D et al (2021) Differential response of MDA-MB-231 and MCF-7 breast cancer cells to in vitro inhibition with CTLA-4 and PD-1 through cancer-immune cells modified interactions. Cells 10(8):2044. https://doi.org/10.3390/cells10082044
Hong W, Guan K-L (2012) The YAP and TAZ transcription co-activators: key downstream effectors of the mammalian Hippo pathway. Semin Cell Dev Biol 23(7):785–793. https://doi.org/10.1016/j.semcdb.2012.05.004
Barranco WT, Eckhert CD (2004) Boric acid inhibits human prostate cancer cell proliferation. Cancer Lett 216(1):21–29. https://doi.org/10.1016/j.canlet.2004.06.001
Barranco WT, Eckhert CD (2004) Boric acid inhibits human prostate cancer cell proliferation. Cancer Lett 216(1):21–29. https://doi.org/10.1016/j.canlet.2004.06.001
Yang N, Sheridan AM (2014) Cell cycle. In: Wexler P (ed) Encyclopedia of toxicology (Third Edition). Academic Press, pp 753–758
Poon RYC (2002) Cell cycle control. In: Bertino JR (ed) Encyclopedia of cancer (Second Edition). Academic Press, pp 393–403
Liu Q, Cao Y, Zhou P et al (2018) Panduratin A inhibits cell proliferation by inducing G0/G1 phase cell cycle arrest and induces apoptosis in breast cancer cells. Biomol Ther (Seoul) 26(3):328–334. https://doi.org/10.4062/biomolther.2017.042
Gu JJ, Kaufman GP, Mavis C, Czuczman MS, Hernandez-Ilizaliturri FJ (2017) Mitotic catastrophe and cell cycle arrest are alternative cell death pathways executed by bortezomib in rituximab resistant B-cell lymphoma cells. Oncotarget 8(8):12741–12753. https://doi.org/10.18632/oncotarget.14405
Shen L, Au WY, Wong KY et al (2008) Cell death by bortezomib-induced mitotic catastrophe in natural killer lymphoma cells. Mol Cancer Ther 7(12):3807–3815. https://doi.org/10.1158/1535-7163.mct-08-0641
Lignitto L, Arcella A, Sepe M et al (2013) Proteolysis of MOB1 by the ubiquitin ligase praja2 attenuates Hippo signalling and supports glioblastoma growth. Nat Commun 4:1822. https://doi.org/10.1038/ncomms2791
Kosaka Y, Mimori K, Tanaka F, Inoue H, Watanabe M, Mori M (2007) Clinical significance of the loss of MATS1 mRNA expression in colorectal cancer. Int J Oncol 31(2):333–338
Zhou D, Conrad C, Xia F et al (2009) Mst1 and Mst2 maintain hepatocyte quiescence and suppress hepatocellular carcinoma development through inactivation of the Yap1 oncogene. Cancer Cell 16(5):425–438. https://doi.org/10.1016/j.ccr.2009.09.026
Wu XS, Wang F, Li HF et al (2017) LncRNA-PAGBC acts as a microRNA sponge and promotes gallbladder tumorigenesis. EMBO Rep 18(10):1837–1853. https://doi.org/10.15252/embr.201744147
Yang B, Li Y, Zhang R et al (2020) MOB1A regulates glucose deprivation-induced autophagy via IL6-STAT3 pathway in gallbladder carcinoma. Am J Cancer Res 10(11):3896–3910
Sampath D, Winneker RC, Zhang Z (2001) Cyr61, a member of the CCN family, is required for MCF-7 cell proliferation: regulation by 17β-estradiol and overexpression in human breast cancer. Endocrinology 142(6):2540–2548. https://doi.org/10.1210/endo.142.6.8186
Tsai M-S, Hornby AE, Lakins J, Lupu R (2000) Expression and function of CYR61, an angiogenic factor, in breast cancer cell lines and tumor biopsies1. Can Res 60(20):5603–5607
Vellon L, Menendez JA, Liu H, Lupu R (2007) Up-regulation of αVβ3 integrin expression is a novel molecular response to chemotherapy-induced cell damage in a heregulin-dependent manner. Differentiation 75(9):819–830. https://doi.org/10.1111/j.1432-0436.2007.00241.x
Xie D, Miller CW, O’Kelly J et al (2001) Breast cancer Cyr61 is overexpressed, estrogen-inducible, and associated with more advanced disease. J Biol Chem 276(17):14187–94. https://doi.org/10.1074/jbc.M009755200
Yamamoto H, Ngan CY, Monden M (2008) Cancer cells survive with survivin. Cancer Sci 99(9):1709–1714. https://doi.org/10.1111/j.1349-7006.2008.00870.x
Wang C, Zheng X, Shen C, Shi Y (2012) MicroRNA-203 suppresses cell proliferation and migration by targeting BIRC5 and LASP1 in human triple-negative breast cancer cells. J Exp Clin Cancer Res 31(1):58. https://doi.org/10.1186/1756-9966-31-58
Wang H, Ye Y-F (2015) Effect of survivin siRNA on biological behaviour of breast cancer MCF7 cells. Asian Pac J Trop Med 8(3):225–228. https://doi.org/10.1016/S1995-7645(14)60320-5
Gupta M, Gupta SK, Hoffman B, Liebermann DA (2006) Gadd45a and Gadd45b protect hematopoietic cells from UV-induced apoptosis via distinct signaling pathways, including p38 activation and JNK inhibition. J Biol Chem 281(26):17552–17558. https://doi.org/10.1074/jbc.M600950200
Li D, Dai C, Zhou Y et al (2016) Effect of GADD45a on olaquindox-induced apoptosis in human hepatoma G2 cells: involvement of mitochondrial dysfunction. Environ Toxicol Pharmacol 46:140–146. https://doi.org/10.1016/j.etap.2016.07.012
Russo T, Piccolo V, Ferrara G et al (2017) Dermoscopy pathology correlation in melanoma. J Dermatol 44(5):507–514. https://doi.org/10.1111/1346-8138.13629
Yin F, Bruemmer D, Blaschke F, Hsueh WA, Law RE, Herle AJ (2004) Signaling pathways involved in induction of GADD45 gene expression and apoptosis by troglitazone in human MCF-7 breast carcinoma cells. Oncogene 23(26):4614–4623. https://doi.org/10.1038/sj.onc.1207598
Sheikh MS, Hollander MC, Fornace AJ (2000) Role of Gadd45 in apoptosis. Biochem Pharmacol 59(1):43–45. https://doi.org/10.1016/S0006-2952(99)00291-9
Gilad Y, Eliaz Y, Yu Y, Han SJ, O’Malley BW, Lonard DM (2019) Drug-induced PD-L1 expression and cell stress response in breast cancer cells can be balanced by drug combination. Sci Reports 9(1):15099. https://doi.org/10.1038/s41598-019-51537-7
Chieochansin T, Thepmalee C, Grainok J, Junking M, Yenchitsomanus PT (2019) Cytolytic activity of effector T-lymphocytes against hepatocellular carcinoma is improved by dendritic cells pulsed with pooled tumor antigens. Sci Rep 9(1):17668. https://doi.org/10.1038/s41598-019-54087-0
Wu Y-g, Wu G-z, Wang L, Zhang Y-Y, Li Z, Li D-C (2010) Tumor cell lysate-pulsed dendritic cells induce a T cell response against colon cancer in vitro and in vivo. Med Oncol 27(3):736–742
Xie BH, Yang JY, Li HP et al (2014) Dendritic cells transfected with hepatocellular carcinoma (HCC) total RNA induce specific immune responses against HCC in vitro and in vivo. Clin Transl Oncol 16(8):753–760. https://doi.org/10.1007/s12094-013-1145-7
Tada F, Abe M, Hirooka M et al (2012) Phase I/II study of immunotherapy using tumor antigen-pulsed dendritic cells in patients with hepatocellular carcinoma. Int J Oncol 41(5):1601–1609. https://doi.org/10.3892/ijo.2012.1626
Caballero-Baños M, Benitez-Ribas D, Tabera J et al (2016) Phase II randomised trial of autologous tumour lysate dendritic cell plus best supportive care compared with best supportive care in pre-treated advanced colorectal cancer patients. Eur J Cancer 64:167–174. https://doi.org/10.1016/j.ejca.2016.06.008
Zimmermann K, Kuehle J, Dragon AC et al (2020) Design and characterization of an “all-in-one” lentiviral vector system combining constitutive anti-GD2 CAR expression and inducible cytokines. Cancers 12(2):375. https://doi.org/10.3390/cancers12020375
Acknowledgements
We acknowledge and appreciate Dr. A. Asli Hizli Deniz for her valuable efforts in improving the use of English in the manuscript. We also would like to express our very great appreciation to Dr. Ahmed Eltokhi, University of Washington, for his valuable and constructive suggestions during the preparation and revision of the manuscript. Their willingness to give their time so generously has been greatly appreciated.
Funding
This work was generously supported by Yeditepe University.
Author information
Authors and Affiliations
Contributions
Eslam Essam Mohammed, Nezaket Türkel, Altay Burak Dalan, and Fikrettin Sahin conceived the idea of the manuscript. Study design, material preparation, experiments, and data collection and analysis were performed by Eslam Essam Mohammed and Ummuhan Miray Yiğit. Eslam Essam Mohammed made the first draft of the manuscript and Nezaket Türkel and Fikrettin Sahin edited and revised the manuscript based on the concept.
Corresponding author
Ethics declarations
Institutional Review Board Statement
Not applicable.
Informed Consent
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.








Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohammed, E.E., Türkel, N., Yigit, U.M. et al. Boron Derivatives Inhibit the Proliferation of Breast Cancer Cells and Affect Tumor-Specific T Cell Activity In Vitro by Distinct Mechanisms. Biol Trace Elem Res 201, 5692–5707 (2023). https://doi.org/10.1007/s12011-023-03632-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03632-0