Abstract
Background
Aluminum and indium are widely used in industrial manufacturing, in pharmaceutical products, in medical treatments, and in food packaging, so they could reach organisms by different way. In order to clarify whether these elements are dangerous, we already demonstrated the ultrastructural modifications observed in the testicles, the epididymides, and the seminal vesicles of rat. Their pro-oxidative effect was also confirmed concomitantly to a decrease in anti-oxidant defenses in the blood, the testicles, and the liver. Thus, it seemed very logic to evaluate damages in the reproductive organs, especially on the exocrine and endocrine functions of the testicles.
Methods
Aluminum and indium were intraperitoneally administered to male Wistar rats. Sperm solution was obtained from cauda epididymides. Motility, viability, density, and malformation of spermatozoa solution were assessed. Serum total unconjugated testosterone concentrations were measured using RIA technique.
Results
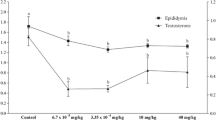
Our results showed a decrease in weight of the testicles, epididymides, and seminal vesicles of indium-treated rats and an increase in the weight of their kidneys. A decrease in motility, viability, and density of epididymides stored sperm as well as generation of many spermatozoa malformations was also observed especially in indium-treated rats. Testosterone levels were increased in indium but were enhanced in aluminum group. This confirmed our previous studies showing that aluminum and indium are toxic for the testicular tissues. This could be explained by the generation of reactive oxygen species (ROS) affecting strongly the exocrine and the endocrine functions of the testicles.
Conclusion
Aluminum and indium are disturbing elements for the exocrine and endocrine functions of rat testicles.




Similar content being viewed by others
Data Availability
All data related to the subject of this study is reported and archived in the Laboratory of Physiology in the Faculty of Medicine of Tunis (Tunisia).
References
Pilette J, (2004)Aluminium et vaccins. 60 p.
Van Hulle M, (2003-2004) Fractionation of indium and speciation of arsenic in body fluids and tissues after exposure. 230 p.
Vallot T, Vatier J, Carella G, Pospai D (1995) Médicaments utilisés dans les affections gastro-duodénales. Editions techniques. Encycl. Med. Chir (Elsevier, Paris), Thérapeutique, 25-510-A10. 15 p.
Wills MR, Savory J (1985) Water Content of Aluminum, Dialysis Dementia, and Osteomalacia. Environ Health Perspect 63:141–147
Gourrier-Frery C, Frery N (2004) Aluminium. Editions techniques Encycl Med Chir (Elsevier, Paris). Toxicologie-Pathologie 1:79–95
Sombra LL, Luconi MO, Fernandez LP, Olsina RA, Silva MF, Martinez LD (2003) Assessment of trace aluminium content in parenteral solutions by combined cloud point preconcentration - flow injection inductively coupled plasma optical emission spectrometry. J Pharm Biomed Anal 30:1451–1458
Darbre PD (2005) Aluminium, antiperspirants and breast cancer. J Inorg Biochem 99:1912–1919
Fitzgerald PG, Topp TJ, Walton JM, Jackson JR, Gillis DA (1992) The use of indium 111 leukocyte scans in children with inflammatory bowel disease. J Pediatr Surg 27(10):1298–1300
Signore A, Beales P, Sensi M, Zuccarini O, Pozzilli P (1983) Labelling of lymphocytes with indium 111 oxine: effect on cell surface phenotype and antibody-dependent cellular cytotoxicity. Immunol Lett 6(3):151–154
Hunter RE, Doherty P, Griffin TW, Gionet M, Hnatowich DJ, Bianco JA, Dillon MB (1987) Use of indium-111-labeled OC-125 monoclonal antibody in the detection of ovarian cancer. Gynecol Oncol 27(3):325–337
Durmus M, Ahsen V (2010) Water-soluble cationic gallium (III) and indium (III) phthalocyanines for photodynamic therapy. J Inorg Biochem 104(3):297–309
Polmear IJ (1993) The elements. In: Downs AJ (ed) Chemistry of alumnium, gallium, indium and thallium. Chapman and Hall, London, pp 81–110
Zurita JL, Jos A, del Peso A, Salguero M, Cameán AM, López-Artíguez M, Repetto G (2007) Toxicological assessment of indium nitrate on aquatic organisms and investigation of the effects on the PLHC-1 fish cell line. Sci Total Environ 387(1-3):155–165
Brindaban CR, Sampak S (2002) Use of indium hydride (cl2inh) for chemoselective reduction of the carbon-carbon double bond in conjugated alkenes. Tetrahedron Lett 43(41):740–7407
Kamboj VP, Kar A (1964) Antitesticular effect of metallic and rare earth salts. J Reprod Fertil 7:21–28
Yousef MI, El-Morsy AMA, Hassan MS (2005) Aluminium induced deterioration in reproductive performance and seminal plasma biochemistry of male rabbits: protective role of ascorbic acid. Toxicology 215:97–107
Yousef MI, Kamel KI, El-Guendi MI, El-Demerdash FM (2007) An in vitro study on reproductive toxicity of aluminium chloride on rabbit sperm: The protective role of some antioxidants. Toxicology 239:213–223
Omura M, Tanaka A, Zhao M, Hirata M, Makita Y, Inoue N, Gotoh K (1995) Toxic effects of gallium arsenide on sperm in rats by repeated intratracheal instillations. J Occup Health 37:165–166
Omura M, Hirata M, Tanaka A, Zhao M, Makita Y, Inoue N, Gotoh K, Ishinishi N (1996) Testicular toxicity evaluation of arsenic-containing binary compounds semiconductors, gallium arsenide and indium arsenide, in hamsters. Toxicol Lett 89:123–129
Omura M, Yamazaki K, Tanaka A, Hirata M, Makita Y, Inoue N (2000) Changes in the testicular damage caused by indium arsenide and indium phosphide in hamsters during two years after intratracheal instillations. J Occup Health 42:196–204
Omura M, Tanaka A, Hirata M, Inoue N, Ueno T, Homma T, Sekizawa K (2002) Testicular toxicity evaluation if indium-Tin oxide. J Occup Health 44:105–107
Maghraoui S, Ahlem A, Aouatef BA, Habib JM, Leila T (2011a) Histological and ultrastructural study of the intracellular behavior of indium in the testicular tissues. Microsc Res Tech 74(6):546–550
Maghraoui S, Clichici S, Ayadi A, Login C, Moldovan R, Daicoviciu D, Decea N, Mureşan A, Tekaya L (2014) Oxidative stress in the blood and testicles of rat following intraperitoneal administration of aluminum and indium. Acta Physiol Hung 101(1):47–58
Hazout A, Menezo Y, Madelenat P, Yazbeck C, Selva J, Cohen-Bacrie P (2008) Causes et implications cliniques des alte´rations de l’ADN des spermatozoides. Gynécol Obs Fertil 36:1109–1117
El-Demerdash FM, Yousef MI, Kedwany FS, Baghdadi HH (2004) Cadmium-induced changes in lipid peroxidation, blood hematology, biochemical parameters and semen quality of male rats: protective role of vitamin E and β-carotene. Food Chem Toxicol 42:1563–1571
Nakajima M, Kobayashi Y, Usami M, Ohno Y (1998) Teratogenic effects of indium by oral or intravenous administration in rats. Teratology 57(3):19A
Nakajima M, Sasaki M, Kobayashi Y, Awano T, Irie D, Usami M, Ohno Y (1999) Studies on developmental toxicity of indium by rat embryo culture and toxicokinetics. Teratology 59(5):25A
Nakajima M, Takahashi H, Sasaki M, Kobayashi Y, Usami M, Ohno Y (2000) Species differences in the developmental toxicity of indium between rats and mice. Teratology. 62(3):41A
Nakajima M, Sasaki M, Kobayashi Y, Usami M, Ohno Y (2001) Studies on the pathogenesis of indium-induced tail anomalies in rat fetuses. Teratology 63(4):24A
Perreault SD. (1997) The mature spermatozoa as a target for reproductive toxicants. In: Boekelheide K, Chapin RE, Hoyer PB, Harris C, editors. Comprehensive toxicology, vol. 10. Reproductive and endocrine toxicology. New York: Pergamon Press. p. 165–79.
Exley C (2004) The pro-oxidant activity of aluminum. Free Radic Biol Med 36:380–387
Yousef MI (2004) Aluminium-induced changes in hematobiochemical parameters, lipid peroxidation and enzyme activities of male rabbits: protective role of ascorbic acid. Toxicology 199:47–57
Saberwal GS, Gill‐Sharma MK, Balasinor N, Choudhary J, Padwal V, Juneja HS (2003) Tamoxifen, protein kinase C and rat sperm mitochondria. Cell Biol Int 27:761–768
Klinfelter G (2002) Actions of toxicants on the structure and function of the epididymis. In: Robaire B, Hinton BT (eds) the Epididymis: from Molecule to Medical Practice. Kluwer Academic/Olenum, New York, pp 353–369
Dacheux J, Castella S, Gatti J, Dacheux F (2005) Epididymal cell secretory activities and the role of proteins in boar sperm maturation. Theriogenology 63:319–341
Klinefelter GR, Suarez JD (1997) Toxicant-induced acceleration of epididymal sperm transit: Androgen-dependent proteins may be involved repdoctive toxicology. Reprod Toxicol 11(4):511–519
Maghraoui S et al (2011b) Behavior of indium study in intestinal mucosa, liver, kidney and testicle after intraperitoneal and intragastric administration to rat. J Electron Microsc 60(2):183–190
Ecroyd H, Jones RC, Aitken RJ (2003) Tyrosine phosphorylationof HSP-90 during mammalian sperm capacitation. Biol Reprod 69:1801–1807
El-Taieb MAA, Herwig R, Nada EA, Greilberger J, Marberger M (2009) Oxidative stress and epididymal sperm transport, motility and morphological defects. Eur J Obstet Gynecol Reprod Biol 144S(2009):S199–S203
Kumar V, Bal A, Gill KD (2009) Susceptibility of mitochondrial superoxide dismutase to aluminium induced oxidative damage. Toxicology 255:117–123
Gatti J, Castella S, Dacheux F, Ecroyd H, Métayer S, Thimon V, Dacheux JL (2004) Posttesticular sperm environment and fertility. Anim Reprod Sci 2004(82-83):321–339
Tanaka A (2004) Toxicity of indium arsenide, gallium arsenide, and aluminium gallium arsenide. Toxicol Appl Pharmacol 198:405–411
Hermo L, Pelletier RM, Cyr DG, Smith CE (2010) Surfing the wave, cycle, life history, and genes/proteins expressed by testicular germ cells. Part 2: Changes in spermatid organelles associated with development of spermatozoa. Microsc Res Tech 73:279–319
Koksal IT, Usta M, Orhan I, Abbasoglu S, Kadioglu A (2003) Potential role of reactive oxygen species on testicular pathology associated with infertility. Asian J Androl 5:95–99
Aitken RJ, Clarkson JS, Fishel S (1989) Generation of reactive oxygen species, lipid peroxidation, and human sperm function. Biol Reprod 41:183–197
Plante M, de Lamirande E, Gagnon C (1994) Reactive oxygen species released by activated neutrophils, but not by deficient spermatozoa, are sufficient to affect normal sperm motility. Fertil Steril 62:387–393
Sharma RK, Agarwal A (1996) Role of reactive oxygen species in male infertility. Urology 48:835–850
De Lamirande E, Jiang H, Zini A, Kodama H, Gagnon C (1997) Reactive oxygen species and sperm physiology. Rev Reprod 2:48–54
Taylor CT (2001) Antioxidants and reactive oxygen species in human fertility. Environ Toxicol Pharmacol 10:189–198
Aitken RJ, Harkiss D, Buckingham D (1993) Relationship between iron-catalyzed lipid peroxidation and human sperm function. J Reprod Fertil 98:257–265
Sanocka D, Kurpisz M (2004) Reactive oxygen species and sperm cells. Reprod Biol Endocrinol 2:1–7
Acknowledgements
* Special thanks to Madam Boussaidi Samiha, technique agent of the Laboratory of Medical Bacteriology of the Pasteur Institute of Tunis (Tunisia) for her precious help in the learning of the techniques of determination of fertility parameters: motility, viability assessment, and density sperm.
* Big thanks are addressed to Professor Ben Rayana Mohamed Chiheb, chief of the Medical Biology Laboratory of the Nutrition Institute of Tunis (Tunisia), for allowing us using the gamma counter for total unconjugated testosterone level measurements.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and experimentation were performed by Dr Maghraoui Samira and Dr Ayadi Ahlem. The SEM imaging and figure interpretation were performed by Dr Florea Adrian. The first draft of the manuscript was written by Dr Maghraoui Samira. Dr Florea Adrian and Pr Matei Horea commented on previous versions of the manuscript. All authors read and approved the final manuscript. Pr Tekaya Leila supervised the different steps of the work and corrected the manuscript in the different phases of redaction.
Corresponding author
Ethics declarations
Ethics Approval
The ethic committee approved the study twice. The first time it was approved by the ethic committee of the Pasteur Institute of Tunis (Tunisia). The second time it was approved by the ethic committee of Iuliu Haţieganu University of Medicine and Pharmacy (Cluj-Napoca; Romania).
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maghraoui, S., Florea, A., Ayadi, A. et al. Changes in Organ Weight, Sperm Quality and Testosterone Levels After Aluminum (Al) and Indium (In) Administration to Wistar Rats. Biol Trace Elem Res 201, 766–775 (2023). https://doi.org/10.1007/s12011-022-03180-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-022-03180-z