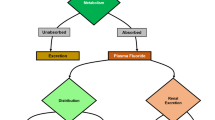
Abstract
Fluorosis is a major public health problem globally. The non-availability of specific treatment and the irreversible nature of dental and skeletal lesions poses a challenge in the management of fluorosis. Oxidative stress is known to be one of the most important mechanisms of fluoride toxicity. Fluoride promotes the accumulation of reactive oxygen species by inhibiting the activity of antioxidant enzymes, resulting in the excessive production of reactive oxygen species at the cellular level which further leads to activation of cell death processes such as apoptosis. Phytochemicals that act as antioxidants have the potential to protect cells from oxidative stress. Evidence confirms that clinical symptoms of fluorosis can be mitigated to some extent or prevented by long-term intake of antioxidants and plant products. The primary purpose of this review is to examine recent findings that focus on the amelioration of fluoride-induced oxidative stress and apoptosis by natural and synthetic phytochemicals and their molecular mechanisms of action.
Similar content being viewed by others
Data Availability
Not applicable.
References
WHO (2019) Preventing diseases through healthy environments. Inadequate or excess fluoride: A major public health concern [Internet]. [cited 2020 Sep 13]. Available from: https://apps.who.int/iris/bitstream/handle/10665/329484/WHO-CED-PHE-EPE-19.4.5-eng.pdf?ua=1
Wei W, Pang S, Sun D (2019) The pathogenesis of endemic fluorosis: research progress in the last 5 years. J Cell Mol Med. 23(4):2333–2342. https://doi.org/10.1111/jcmm.14185
WHO (2017) Guidelines for drinking-water quality, 4th edition, incorporating the 1st addendum [Internet]. WHO. World Health Organization; 2017 [cited 2020 Sep 13]. Available from: http://www.who.int/water_sanitation_health/publications/drinking-water-quality-guidelines-4-including-1st-addendum/en/
Malago J (2017) Fluoride levels in surface and groundwater in Africa: a review. Am J Water Sci Eng [Internet] 3(1):1. [cited 2020 Sep 14]. Available from: http://www.sciencepublishinggroup.com/journal/paperinfo?journalid=369&doi=10.11648/j.ajwse.20170301.11. https://doi.org/10.11648/j.ajwse.20170301.11
Khairnar MR, Dodamani AS, Jadhav HC, Naik RG, Deshmukh MA (2015) Mitigation of fluorosis — a review [Internet]. Vol. 9, Journal of Clinical and Diagnostic Research. Journal of Clinical and Diagnostic Research:ZE05–ZE09. [cited 2020 Sep 24] Available from: /pmc/articles/PMC4525626/?report=abstract. https://doi.org/10.7860/JCDR/2015/13261.6085
Shenoy PS, Sen U, Kapoor S, Ranade AV, Chowdhury CR, Bose B (2019) Sodium fluoride induced skeletal muscle changes: degradation of proteins and signaling mechanism. Environ Pollut. 244:534–548. https://doi.org/10.1016/j.envpol.2018.10.034
Asawa K, Singh A, Bhat N, Tak M, Shinde K, Jain S (2015) Association of temporomandibular joint signs & symptoms with dental fluorosis & skeletal manifestations in endemic fluoride areas of Dungarpur district, Rajasthan, India. J Clin Diagnostic Res [Internet] 9(12):ZC18–ZC21. [cited 2020 Sep 24] Available from: /pmc/articles/PMC4717726/?report=abstract. https://doi.org/10.7860/JCDR/2015/15807.6958
Leela Lyengar. 22 Technologies for fluoride removal [Internet]. [cited 2020 Nov 9]. Available from: https://www.samsamwater.com/library/TP40_22_Technologies_for_fluoride_removal.pdf
Fewtrell L, Smith S, Kay D, Bartram J (2006) An attempt to estimate the global burden of disease due to fluoride in drinking water. J Water Health. 4(4):533–542. https://doi.org/10.2166/wh.2006.045
Thole B (2013 [cited 2020 Nov 6]. Available from: 10.5772/54985) Ground water contamination with fluoride and potential fluoride removal technologies for East and Southern Africa. In: Dar IA, MA (eds) Perspectives in Water Pollution [Internet]. InTech. https://doi.org/10.5772/54985
Qin X, Wang S, Yu M, Zhang L, Li X, Zuo Z et al (2009) Child skeletal fluorosis from indoor burning of coal in Southwestern China. J Environ Public Health. https://doi.org/10.1155/2009/969764
Brindha K, Elango L Elango B and Fluoride in groundwater: causes, implications and mitigation measures. Department of Geology, Anna University, Chennai- 600025, India Appl Environ Manag. 2011;111-136
Brunt R, Vasak L, Griffioen J (2004) Fluoride in groundwater: probability of occurrence of excessive concentration on global scale. Int Groundwater Resources Accessment Centre, UNESCO [Internet] [cited 2020 Nov 6];Report nr. SP 2(April):3. Available from: http://www.wateraid.org
Kaseva ME (2006) Contribution of trona (magadi) into excessive fluorosis—a case study in Maji ya Chai ward, northern Tanzania. Sci Total Environ. 366(1):92–100. https://doi.org/10.1016/j.scitotenv.2005.08.049
WHO (2020) WHO | Water-related diseases [Internet]. [cited 2020 Nov 6]. Available from: https://www.who.int/water_sanitation_health/diseases-risks/diseases/fluorosis/en/
Everett ET (2011) Critical reviews in oral biology & medecine: fluoride’s effects on the formation of teeth and bones, and the influence of genetics [Internet]. J Dental Res International Association for Dental Research 90:552–60 [cited 2020 Nov 6]. Available from: /pmc/articles/PMC3144112/?report=abstract. https://doi.org/10.1177/0022034510384626
Kurdi MS (2016) Chronic fluorosis: The disease and its anaesthetic implications. Indian J Anaesth [Internet]. 60(3):157–62 [cited 2020 Nov 7]. Available from: /pmc/articles/PMC4800930/?report=abstract. https://doi.org/10.4103/0019-5049.177867
Li Y, Liang C, Slemenda CW, Ji R, Sun S, Cao J et al (2001) Effect of long-term exposure to fluoride in drinking water on risks of bone fractures. J Bone Miner Res. 16(5):932–939. https://doi.org/10.1359/jbmr.2001.16.5.932
Susheela AK, Toteja GS (2018) Prevention & control of fluorosis & linked disorders: —Developments in the 21 st century — reaching out to patients in the community & hospital settings for recovery [Internet]. Indian J Med Res Wolters Kluwer Medknow Publications 148:539–47. [cited 2020 Nov 7]. Available from: /pmc/articles/PMC6366265/?report=abstract. https://doi.org/10.4103/ijmr.IJMR_1775_18
Mohapatra M, Anand S, Mishra BK, Giles DE, Singh P (2009) Review of fluoride removal from drinking water. J Environ Manage. 91(1):67–77. https://doi.org/10.1016/j.jenvman.2009.08.015
WHO (2008) Guidelines for drinking-water quality third edition incorporating the first and second addenda volume 1 recommendations Geneva 2008 WHO library cataloguing-in-publication data. Guidel Drink Qual. 1.[cited 2020 Nov 9]
Viswanathan G, Jaswanth A, Gopalakrishnan S, Siva Ilango S (2009) Mapping of fluoride endemic areas and assessment of fluoride exposure. Sci Total Environ [Internet]. 407(5):1579–87 15 [cited 2020 Nov 9]. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0048969708010504. https://doi.org/10.1016/j.scitotenv.2008.10.020
Luo Q, Cui H, Deng H, Kuang P, Liu H, Lu Y, et al. (2017) Histopathological findings of renal tissue induced by oxidative stress due to different concentrations of fluoride. Oncotarget [Internet]. 8(31):50430–46. [cited 2020 Sep 24]. Available from: /pmc/articles/PMC5584147/?report=abstract. https://doi.org/10.18632/oncotarget.17365
Shanmugam T, Selvaraj M, Poomalai S (2016) Epigallocatechin gallate potentially abrogates fluoride induced lung oxidative stress, inflammation via Nrf2/Keap1 signaling pathway in rats: An in-vivo and in-silico study. Int Immunopharmacol [Internet]. 39:128–39 1 [cited 2020 Oct 18]. Available from: https://pubmed.ncbi.nlm.nih.gov/27472294/. https://doi.org/10.1016/j.intimp.2016.07.022
Lu Y, Luo Q, Cui H, Deng H, Kuang P, Liu H, et al. (2017) Sodium fluoride causes oxidative stress and apoptosis in the mouse liver. Aging (Albany NY) [Internet]. 9(6):1623–39. [cited 2020 Sep 24]. Available from: /pmc/articles/PMC5509460/?report=abstract. https://doi.org/10.18632/aging.101257
Basha PM, Madhusudhan N (2010) Pre and post natal exposure of fluoride induced oxidative macromolecular alterations in developing central nervous system of rat and amelioration by antioxidants. Neurochem Res [Internet]. 35(7):1017–28. [cited 2020 Oct 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/20336367/. https://doi.org/10.1007/s11064-010-0150-2
Sun Z, Li S, Guo Z, Li R, Wang J, Niu R, et al. (2018) Effects of fluoride on SOD and CAT in testis and epididymis of mice. Biol Trace Elem Res [Internet]. 184(1):148–53. [cited 2020 Oct 18]. Available from: https://pubmed.ncbi.nlm.nih.gov/28990137/. https://doi.org/10.1007/s12011-017-1181-1
Ozbey U, Deger Y, Yur F, Çambay Z, Ozbeye G (2017) Investigation of blood antioxidant enzyme levels and glutathione peroxidase, catalase, and superoxide dismutase gene polymorphism in sheep with fluorosis. Fluoride. 50(3):374–382
Yilmaz S, Yur F (2012) Nitric oxide oxidation products and the activities of catalase and carbonic anhydrase in sheep with fluorosis. Fluoride. 45(3):247–250
Yur F, Mert N, Dede S, Deǧer Y, Ertekin A, Mert H et al (2013) Evaluation of serum lipoprotein and tissue antioxidant levels in sheep with fluorosis. Fluoride. 46(2):90–96
Shivarajashankara YM, Shivashankara AR, Hanumanth Rao S, Gopalakrishna BP (2001) Oxidative stress in children with endemic skeletal fluorosis. Fluoride. 34(2):103–107
Ailani V, Gupta RC, Gupta SK, Gupta K (2009) Oxidative stress in cases of chronic fluoride intoxication. Indian J Clin Biochem. 24(4):426–429. https://doi.org/10.1007/s12291-009-0076-0
Rao SM, Sherlin HJ, Anuja N, Pratibha R, Priya P, Chandrasekar T (2011) Morphometry of buccal mucosal cells in fluorosis — a new paradigm. Hum Exp Toxicol [Internet]. 30(11):1761–8. [cited 2020 Sep 24]. Available from: http://journals.sagepub.com/doi/10.1177/0960327111400109. https://doi.org/10.1177/0960327111400109
Ameeramja J, Panneerselvam L, Govindarajan V, Jeyachandran S, Baskaralingam V, Perumal E (2016) Tamarind seed coat ameliorates fluoride induced cytotoxicity, oxidative stress, mitochondrial dysfunction and apoptosis in A549 cells. J Hazard Mater [Internet]. 301:554–65. [cited 2020 Sep 26]. Available from: https://pubmed.ncbi.nlm.nih.gov/26439939/. https://doi.org/10.1016/j.jhazmat.2015.09.037
Li W, Jiang B, Cao X, Xie Y, Huang T (2017) Protective effect of lycopene on fluoride-induced ameloblasts apoptosis and dental fluorosis through oxidative stress-mediated Caspase pathways. Chem Biol Interact [Internet]. 261:27–34. [cited 2020 Sep 23]. Available from: https://pubmed.ncbi.nlm.nih.gov/27871895/. https://doi.org/10.1016/j.cbi.2016.11.021
Shuhua X, Ziyou L, Ling Y, Fei W, Sun G (2012) A role of fluoride on free radical generation and oxidative stress in Bv-2 microglia cells. Mediators Inflamm. 2012. https://doi.org/10.1155/2012/102954
Birben E, Sahiner UM, Sackesen C, Erzurum S, Kalayci O (2012) Oxidative stress and antioxidant defense. World Allergy Organization Journal. https://doi.org/10.1097/WOX.0b013e3182439613
Barbier O, Arreola-Mendoza L, Del Razo LM (2010) Molecular mechanisms of fluoride toxicity. Chem Biol Interact. 188(2):319–333. https://doi.org/10.1016/j.cbi.2010.07.011
Miranda GHN, Gomes BAQ, Bittencourt LO, Aragão WAB, Nogueira LS, Dionizio AS et al (2018) Chronic exposure to sodium fluoride triggers oxidative biochemistry misbalance in mice: Effects on peripheral blood circulation. Oxid Med Cell Longev. https://doi.org/10.1155/2018/8379123
Chlubek D (2003) Fluoride and oxidative stress [Internet]. Vol. 36, Fluoride. International Society for Fluoride Research, Dunedin, pp 217–228 Available from: https://www.fluorideresearch.org/364/files/FJ2003_v36_n4_p217-228.pdf
Mahaboob Basha P, Madhusudhan N (2011) Effect of maternal exposure of fluoride on oxidative stress markers and amelioration by selected antioxidants in developing central nervous system of rats. Biologia (Bratisl) [Internet]. 66(1):187–193. Available from: https://www.degruyter.com/view/journals/biolog/66/1/article-p187.xml. https://doi.org/10.2478/s11756-010-0136-1
Connett M (2012) Fluoride & Oxidative Stress [Internet]. Fluoride Action Network. [cited 2020 Sep 25]. Available from: https://fluoridealert.org/studies/oxidative-stress/
Zhang YJ, Gan RY, Li S, Zhou Y, Li AN, Xu DP, et al. (2015) Antioxidant phytochemicals for the prevention and treatment of chronic diseases [Internet]. Molecules MDPI AG 20:21138–56. [cited 2020 Sep 25]. Available from: https://pubmed.ncbi.nlm.nih.gov/26633317/. https://doi.org/10.3390/molecules201219753
Liu RH (2013) Health-promoting components of fruits and vegetables in the diet. Adv Nutr [Internet]. 4(3):384S. [cited 2020 Sep 25]. Available from: /pmc/articles/PMC3650511/?report=abstract. https://doi.org/10.3945/an.112.003517
Webb D (2013) Phytochemicals’ role in good health. Today’s Dietitian [Internet]. 70 [cited 2020 Sep 25]. Available from: https://www.todaysdietitian.com/newarchives/090313p70.shtml
De Felice SL (1989) The NutraCeutical Revolution: fueling a powerful, new international market. Harvard Univ Adv Manag Progr Biomed Res Dev Como, Italy [Internet]. [cited 2020 Sep 25]; Available from: https://fimdefelice.org/library/the-nutraceutical-revolution-fueling-a-powerful-new-international-market/
Calvani M, Pasha A, Favre C (2020) Nutraceutical boom in cancer: inside the labyrinth of reactive oxygen species [Internet]. Int J Mol Sci MDPI AG 21: 1936. [cited 2020 Sep 25]. Available from: www.mdpi.com/journal/ijms. https://doi.org/10.3390/ijms21061936
Ashwlayan VD, Nimesh S (2018) Nutraceuticals in the management of diabetes mellitus. Pharm Pharmacol Int J [Internet]. 6(2):114–20. [cited 2020 Sep 25]. Available from: http://medcraveonline.com. https://doi.org/10.15406/ppij.2018.06.00166
Sosnowska B, Penson P, Banach M (2017) The role of nutraceuticals in the prevention of cardiovascular disease [Internet]. Cardiovasc Diagn Ther AME Publishing Company 7:S21–31. [cited 2020 Sep 25]. Available from: /pmc/articles/PMC5418215/?report=abstract. https://doi.org/10.21037/cdt.2017.03.20
Dadhania PV, Trivedi P, Vikram A, Nand Tripathi D (2016) Nutraceuticals against neurodegeneration: a mechanistic insight. Curr Neuropharmacol [Internet]. 14(6):627–40 [cited 2020 Sep 25]. Available from: /pmc/articles/PMC4981739/?report=abstract. https://doi.org/10.2174/1570159x14666160104142223
Chang CL, Lin CS, Lai GH (2012) Phytochemical characteristics, free radical scavenging activities, and neuroprotection of five medicinal plant extracts. Evidence-based Complement Altern Med. 2012. https://doi.org/10.1155/2012/984295
Salar RK, Seasotiya L (2011) Free radical scavenging activity, phenolic contents and phytochemical evaluation of different extracts of stem bark of Butea monosperma (Lam.) Kuntze. Front Life Sci [Internet]. 5(3–4):107–16. [cited 2020 Nov 7]. Available from: http://www.tandfonline.com/doi/abs/10.1080/21553769.2011.635813. https://doi.org/10.1080/21553769.2011.635813
Benmehdi H, Behilil A, Memmou F, Amrouche A (2017) Free radical scavenging activity, kinetic behaviour and phytochemical constituents of Aristolochia clematitis L. roots. Arab J Chem. 10:S1402–S1408. https://doi.org/10.1016/j.arabjc.2013.04.015
Thatoi HN, Patra JK, Das SK (2014) Free radical scavenging and antioxidant potential of mangrove plants: a review. Acta Physiol Plant 36:561–579. https://doi.org/10.1007/s11738-013-1438-z
Gad FAM, Farouk SM, Emam MA (2020) Antiapoptotic and antioxidant capacity of phytochemicals from Roselle (Hibiscus sabdariffa) and their potential effects on monosodium glutamate-induced testicular damage in rat. Environ Sci Pollut Res [Internet] 1–12. [cited 2020 Nov 7]. Available from: https://link.springer.com/article/10.1007/s11356-020-10674-7. https://doi.org/10.1007/s11356-020-10674-7
Naoi M, Shamoto-Nagai M, Maruyama W (2019) Neuroprotection of multifunctional phytochemicals as novel therapeutic strategy for neurodegenerative disorders: antiapoptotic and antiamyloidogenic activities by modulation of cellular signal pathways. Future Neurol [Internet] 14(1):FNL9. [cited 2020 Nov 7]. Available from: https://www.futuremedicine.com/doi/10.2217/fnl-2018-0028. https://doi.org/10.2217/fnl-2018-0028
Dutta K, Ghosh D, Basu A (2009) Curcumin protects neuronal cells from japanese encephalitis virus-mediated cell death and also inhibits infective viral particle formation by dysregulation of ubiquitin-proteasome system. J Neuroimmune Pharmacol [Internet]. 4(3):328–37. [cited 2020 Nov 7]. Available from: https://pubmed.ncbi.nlm.nih.gov/19434500/. https://doi.org/10.1007/s11481-009-9158-2
Sobeh M, Mahmoud MF, Hasan RA, Cheng H, El-Shazly AM, Wink M (2017) Senna singueana: Antioxidant, hepatoprotective, antiapoptotic properties and phytochemical profiling of a methanol bark extract. Molecules [Internet]. 22(9):1502. [cited 2020 Nov 7]. Available from: https://pubmed.ncbi.nlm.nih.gov/28885586/. https://doi.org/10.3390/molecules22091502
Choi YJ, Kang JS, Park JHY, Lee YJ, Choi JS, Kang YH (2003) Polyphenolic flavonoids differ in their antiapoptotic efficacy in hydrogen peroxide-treated human vascular endothelial cells. J Nutr [Internet]. 133(4):985–91. [cited 2020 Nov 7]. Available from: https://academic.oup.com/jn/article/133/4/985/4688298. https://doi.org/10.1093/jn/133.4.985
Inkielewicz-Stepniak I, Radomski MW, Wozniak M (2012) Fisetin prevents fluoride- and dexamethasone-induced oxidative damage in osteoblast and hippocampal cells. Food Chem Toxicol [Internet]. 50(3–4):583–9 [cited 2020 Oct 18]. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0278691511006818. https://doi.org/10.1016/j.fct.2011.12.015
K S (2016) Hematobiochemical and antioxidant evaluation of aloe vera whole leaf extract on fluoride induced toxicity in Wistar Albino Rats. SOJ Vet Sci 2(1):1–5. https://doi.org/10.15226/2381-2907/2/1/00115
Madhusudhan N, Mahaboob Basha P, Rai P, Ahmed F, Ravi Prasad G (2010) Effect of maternal fluoride exposure on developing CNS of rats: protective role of Aloe vera, Curcuma longa and Ocimum sanctum. Indian J Exp Biol [Internet]. 48(8):830–6. [cited 2020 Oct 18]. Available from: https://pubmed.ncbi.nlm.nih.gov/21341542/.
Bouasla A, Bouasla I, Boumendjel A, Abdennour C, El Feki A, Messarah M. (2016) Prophylactic effects of pomegranate (Punica granatum) juice on sodium fluoride induced oxidative damage in liver and erythrocytes of rats. Can J Physiol Pharmacol [Internet]. 94(7):709–18. [cited 2020 Oct 18]. Available from: https://cdnsciencepub.com/doi/abs/10.1139/cjpp-2015-0226. https://doi.org/10.1139/cjpp-2015-0226
Pritam Sadhukhan S (2016). Anti-oxidative effect of genistein and mangiferin on sodium fluoride induced oxidative insult of renal cells: a comparative study. Biomarkers J [Internet]. 2(1):1 [cited 2020 Oct 18]. Available from: https://biomarkers.imedpub.com/antioxidative-effect-of-genistein-and-mangiferin-on-sodium-fluoride-induced-oxidative-insult-of-renal-cells-a-comparative-study.php?aid=8207.
Ameeramja J, Perumal E (2018) Possible Modulatory effect of tamarind seed coat extract on fluoride-induced pulmonary inflammation and fibrosis in rats. Inflammation [Internet]. 41(3):886–95 [cited 2020 Sep 26]. Available from: http://link.springer.com/10.1007/s10753-018-0743-5. https://doi.org/10.1007/s10753-018-0743-5
Vasant RA, Narasimhacharya AVRL (2012) Ameliorative effect of tamarind leaf on fluoride-induced metabolic alterations. Environ Health Prev Med [Internet]. 17(6):484–93. [cited 2020 Sep 13]. Available from: /pmc/articles/PMC3493631/?report=abstract. https://doi.org/10.1007/s12199-012-0277-7
Hassan HA, Abdel-Aziz AF (2010) Evaluation of free radical-scavenging and anti-oxidant properties of black berry against fluoride toxicity in rats. Food Chem Toxicol [Internet]. 48(8–9):1999–2004. Available from: https://www.researchgate.net/publication/44601241_Evaluation_of_free_radical-scavenging_and_anti-oxidant_properties_of_black_berry_against_fluoride_toxicity_in_rats. https://doi.org/10.1016/j.fct.2010.05.018
Hamza RZ, El-Shenawy NS, Ismail HAA (2015) Protective effects of blackberry and quercetin on sodium fluoride-induced oxidative stress and histological changes in the hepatic, renal, testis and brain tissue of male rat. J Basic Clin Physiol Pharmacol. 26(3):237–251. https://doi.org/10.1515/jbcpp-2014-0065
Hassan HA, Yousef MI (2009) Mitigating effects of antioxidant properties of black berry juice on sodium fluoride induced hepatotoxicity and oxidative stress in rats. Food Chem Toxicol. 47(9):2332–2337. https://doi.org/10.1016/j.fct.2009.06.023
Karadeniz A, Şimşek N, Koç F, Alp H (2009) The investigation of protective effects of the panax ginseng on oxidative damage induced by chronic fluoride toxicity in mice testis tissue. Kafkas Univ Vet Fak Derg. 15(1):1–8. https://doi.org/10.9775/kvfd.2008.23-a
Mahaboob Basha P, Saumya SM (2013) Suppression of mitochondrial oxidative phosphorylation and TCA enzymes in discrete brain regions of mice exposed to high fluoride: Amelioration by Panax ginseng (Ginseng) and Lagerstroemia speciosa (Banaba) extracts. Cell Mol Neurobiol [Internet]. 33(3):453–64. [cited 2020 Sep 17]. Available from: https://link.springer.com/article/10.1007/s10571-013-9912-0. https://doi.org/10.1007/s10571-013-9912-0
Mahaboob Basha P, Saumya SM (2013) Influence of fluoride on streptozotocin induced diabetic nephrotoxicity in mice: protective role of Asian ginseng (Panax ginseng) & banaba (Lagerstroemia speciosa) on mitochondrial oxidative stress. Indian J Med Res [Internet]. 137(2):370–9. [cited 2020 Sep 17]. Available from: /pmc/articles/PMC3657862/?report=abstract
Nabavi SF, Habtemariam S, Jafari M, Sureda A, Nabavi SM (2012) Protective role of gallic acid on sodium fluoride induced oxidative stress in rat brain. Bull Environ Contam Toxicol [Internet]. 89(1):73–7. [cited 2020 Sep 18]. Available from: https://pubmed.ncbi.nlm.nih.gov/22531840/. https://doi.org/10.1007/s00128-012-0645-4
Nabavi SM, Habtemariam S, Nabavi SF, Sureda A, Daglia M, Moghaddam AH et al (2013) Protective effect of gallic acid isolated from Peltiphyllum peltatum against sodium fluoride-induced oxidative stress in rat’s kidney. Mol Cell Biochem. 372(1–2):233–239. https://doi.org/10.1007/s11010-012-1464-y
Nabavi SF, Habtemariam S, Sureda A, Hajizadeh Moghaddam A, Daglia M, Nabavi SM (2013) In vivo protective effects of gallic acid isolated from peltiphyllum peltatum against sodium fluoride-induced oxidative stress in rat erythrocytes. Arh Hig Rada Toksikol. 64(4):553–559. https://doi.org/10.2478/10004-1254-64-2013-2353
Sinha M, Manna P, Sil PC (2008) Terminalia arjuna protects mouse hearts against sodium fluoride-induced oxidative stress. J Med Food [Internet]. 11(4):733–40. [cited 2020 Sep 18]. Available from: https://pubmed.ncbi.nlm.nih.gov/19053867/. https://doi.org/10.1089/jmf.2007.0130
Sinha M, Manna P, Sil PC (2007) Aqueous extract of the bark of Terminalia arjuna plays a protective role against sodium-fluoride-induced hepatic and renal oxidative stress. J Nat Med [Internet]. 61(3):251–60.’; [cited 2020 Sep 18]. Available from: https://link.springer.com/article/10.1007/s11418-007-0133-z. https://doi.org/10.1007/s11418-007-0133-z
Ghosh J, Das J, Manna P, Sil PC (2008) Cytoprotective effect of arjunolic acid in response to sodium fluoride mediated oxidative stress and cell death via necrotic pathway. Toxicol Vitr. 22(8):1918–1926. https://doi.org/10.1016/j.tiv.2008.09.010
Nabavi SF, Nabavi SM, Latifi AM, Mirzaei M, Habtemariam S, Moghaddam AH (2012) Mitigating role of quercetin against sodium fluoride-induced oxidative stress in the rat brain. Pharm Biol [Internet]. 50(11):1380–3. [cited 2020 Sep 22]. Available from: https://pubmed.ncbi.nlm.nih.gov/22870923/. https://doi.org/10.3109/13880209.2012.675341
Nabavi SF, Eslami S, Moghaddam AH, Jafari N, Nabavi SM, Ebrahimzadeh MA (2011) Protective effects of quercetin against sodium fluoride-induced oxidative stress in rat erythrocytes. Toxicol Environ Chem [Internet]. 93(8):1666–75. [cited 2020 Sep 22]. Available from: https://www.tandfonline.com/doi/abs/10.1080/02772248.2011.600314. https://doi.org/10.1080/02772248.2011.600314
Nabavi SM, Nabavi SF, Habtemariam S, Moghaddam AH, Latifi AM (2012) Ameliorative effects of quercetin on sodium fluoride-induced oxidative stress in rat’s kidney. Ren Fail. 34(7):901–906. https://doi.org/10.3109/0886022X.2012.687347
Nabavi SMF, Nabavi SMF, Mirzaei M, Moghaddam AH (2012) Protective effect of quercetin against sodium fluoride induced oxidative stress in rat’s heart. Food Funct [Internet]. 3(4):437–41. [cited 2020 Sep 23]. Available from: https://pubs.rsc.org/en/content/articlehtml/2012/fo/c2fo10264a. https://doi.org/10.1039/c2fo10264a
Chouhan S, Yadav A, Kushwah P, Kaul RK, Flora SJS (2011). Silymarin and quercetin abrogates fluoride induced oxidative stress and toxic effects in rats. Mol Cell Toxicol [Internet]. 7(1):25–32. [cited 2020 Sep 26]. Available from: https://link.springer.com/article/10.1007/s13273-011-0004-2. https://doi.org/10.1007/s13273-011-0004-2
Nabavi SM, Nabavi SF, Eslami S, Moghaddam AH (2012) In vivo protective effects of quercetin against sodium fluoride-induced oxidative stress in the hepatic tissue. Food Chem. 132(2):931–935. https://doi.org/10.1016/j.foodchem.2011.11.070
Mesram N, Nagapuri K, Banala RR, Nalagoni CR, Karnati PR (2017) Quercetin treatment against NaF induced oxidative stress related neuronal and learning changes in developing rats. J King Saud Univ - Sci. 29(2):221–229. https://doi.org/10.1016/j.jksus.2016.04.002
Tian Y, Xiao Y, Wang B, Sun C, Tang K, Sun F (2018) Vitamin E and lycopene reduce coal burning fluorosis-induced spermatogenic cell apoptosis via oxidative stress-mediated JNK and ERK signaling pathways. Biosci Rep [Internet]. 38(4):20171003. [cited 2020 Sep 23]. Available from: /pmc/articles/PMC6066653/?report=abstract. https://doi.org/10.1042/BSR20171003
Mansour HH, Tawfik SS (2012) Efficacy of lycopene against fluoride toxicity in rats. Pharm Biol [Internet]. 50(6):707–11. [cited 2020 Sep 24]. Available from: http://www.tandfonline.com/doi/full/10.3109/13880209.2011.618994. https://doi.org/10.3109/13880209.2011.618994
Nabavi SF, Nabavi SM, Ebrahimzadeh MA, Eslami S, Jafari N, Hajizadeh MA (2011) The protective effect of curcumin against sodium fluoride-induced oxidative stress in rat heart. Arch Biol Sci. 63(3):563–569. https://doi.org/10.2298/ABS1103563N
Nabavi SF, Eslami S, Moghaddam AH, Nabavi SM (2011) Protective effects of curcumin against fluoride-induced oxidative stress in the rat brain. Neurophysiology. 43(4):287–291. https://doi.org/10.1007/s11062-011-9228-y
Moghaddam AH, Nabavi SF, Nabavi SM, Loizzo MR, Roohbakhsh A, Setzer WN (2015) Ameliorative effects of curcumin against sodium fluoride-induced hepatotoxicity. Prog Nutr. 17(4):324–330
Sharma C, Suhalka P, Sukhwal P, Jaiswal N, Bhatnagar M (2014) Curcumin attenuates neurotoxicity induced by fluoride: an in vivo evidence. Pharmacogn Mag [Internet]. 10(37):61–5. [cited 2020 Sep 26]. Available from: /pmc/articles/PMC3969660/?report=abstract. https://doi.org/10.4103/0973-1296.126663
Nabavi SM, Nabavi SF, Moghaddam AH, Setzer WN, Mirzaei M (2012) Effect of silymarin on sodium fluoride-induced toxicity and oxidative stress in rat cardiac tissues. An Acad Bras Cienc [Internet]. 84(4):1121–6. [cited 2020 Sep 26]. Available from: https://pubmed.ncbi.nlm.nih.gov/22964841/ .https://doi.org/10.1590/S0001-37652012005000056
Nabavi SM, Sureda A, Nabavi SF, Latifi AM, Moghaddam AH, Hellio C (2012) Neuroprotective effects of silymarin on sodium fluoride-induced oxidative stress. J Fluor Chem. 142:79–82. https://doi.org/10.1016/j.jfluchem.2012.06.029
Nabavi SM, Nabavi SF, Loizzo MR, Sureda A, Amani MA, Moghaddam AH (2012) Cytoprotective effect of Silymarin against sodium fluoride-induced oxidative stress in rat erythrocytes. Fluoride. 45(1):27–34
Miltonprabu S, Thangapandiyan S (2015) Epigallocatechin gallate potentially attenuates fluoride induced oxidative stress mediated cardiotoxicity and dyslipidemia in rats. J Trace Elem Med Biol [Internet]. 29:321–35. [cited 2020 Sep 26]. Available from: https://pubmed.ncbi.nlm.nih.gov/25282272/. https://doi.org/10.1016/j.jtemb.2014.08.015
Thangapandiyan S, Miltonprabu S (2015) Epigallocatechin gallate exacerbates fluoride-induced oxidative stress mediated testicular toxicity in rats through the activation of Nrf2 signaling pathway. Asian Pacific J Reprod. 4(4):272–287. https://doi.org/10.1016/j.apjr.2015.07.005
Abdel-Wahab WM (2013) Protective effect of thymoquinone on sodium fluoride-induced hepatotoxicity and oxidative stress in rats. J Basic Appl Zool. 66(5):263–270. https://doi.org/10.1016/j.jobaz.2013.04.002
Alhusaini AM, Faddah LM, El Orabi NF, Hasan IH (2018) Role of some natural antioxidants in the modulation of some proteins expressions against sodium fluoride-induced renal injury. Biomed Res Int. 2018. https://doi.org/10.1155/2018/5614803
Abou Anza R, Salah EH (2015) Mitigation of fluoride toxicity by the use of thymoquinone in adult male Albino Rat. Ain Shams J Forensic Med Clin Toxicol 24(1):1–10. https://doi.org/10.21608/ajfm.2015.18652
Alhusaini A, Faddaa L, Ali HM, Hassan I, El Orabi NF, Bassiouni Y (2018) Amelioration of the protein expression of Cox2, NF κ B, and STAT-3 by some antioxidants in the liver of sodium fluoride–intoxicated rats. Dose-Response [Internet]. 16(3):155932581880015. [cited 2020 Oct 18]. Available from: http://journals.sagepub.com/doi/10.1177/1559325818800153. https://doi.org/10.1177/1559325818800153
Wang EH, Yu ZL, Ping GF, Zhai DS (2020) Grape seed procyanidin extract attenuate sodium fluoride-induced oxidative damage and apoptosis in rat kidneys [Internet]. Biomed Environ Sci Elsevier Ltd 33:454–7. [cited 2020 Oct 18]. p. . Available from: http://www.besjournal.com/en/article/doi/10.3967/bes2020.061. https://doi.org/10.3967/bes2020.061
Niu Q, Mu L, Li S, Xu S, Ma R, Guo S (2016) Proanthocyanidin protects human embryo hepatocytes from fluoride-induced oxidative stress by regulating iron metabolism. Biol Trace Elem Res [Internet]. 169(2):174–9. [cited 2020 Oct 18]. Available from: https://pubmed.ncbi.nlm.nih.gov/26105545/. https://doi.org/10.1007/s12011-015-0409-1
Niu Q, He P, Xu S, Ma R, Ding Y, Mu L, et al. (2018) Fluoride-induced iron overload contributes to hepatic oxidative damage in mouse and the protective role of Grape seed proanthocyanidin extract. J Toxicol Sci [Internet]. 43(5):311–9. [cited 2020 Oct 18]. Available from: https://pubmed.ncbi.nlm.nih.gov/29743442/. https://doi.org/10.2131/jts.43.311
Eşsiz D, Eraslan G, Altintaş L (2008) Antioxidant and therapeutic efficacy of proanthocyanidin in sodium fluoride-intoxicated mice. Fluoride 41:308–313
Khudiar KK (2015) Effect of grape seed oil on hepatic function in adult male rabbits treated with sodium fluoride (Part-II). Adv Anim Vet Sci 3(10):550–558. https://doi.org/10.14737/journal.aavs/2015/3.10.550.558
Giri A, Bharti VK, Angmo K, Kalia S, Kumar B (2016) Fluoride induced oxidative stress, immune system and apoptosis in animals: a review. Int J Bioassays 5(12):5174. https://doi.org/10.21746/ijbio.2016.12.0011
Chattopadhyay A, Podder S, Agarwal S, Bhattacharya S (2011) Fluoride-induced histopathology and synthesis of stress protein in liver and kidney of mice. Arch Toxicol [Internet]. 85(4):327–35. [cited 2020 Oct 16];. Available from: https://pubmed.ncbi.nlm.nih.gov/20859737/. https://doi.org/10.1007/s00204-010-0588-7
Wang HW, Zhao WP, Liu J, Tan PP, Zhang C, Zhou BH (2017) Fluoride-induced oxidative stress and apoptosis are involved in the reducing of oocytes development potential in mice. Chemosphere [Internet]. 186:911–8. [cited 2020 Oct 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/28826138/. https://doi.org/10.1016/j.chemosphere.2017.08.068
Chen T, Cui H, Cui Y, Bai C, Gong T (2011) Decreased antioxidase activities and oxidative stress in the spleen of chickens fed on high-fluorine diets. Hum Exp Toxicol [Internet]. 30(9):1282–6. [cited 2020 Oct 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/21071555/. https://doi.org/10.1177/0960327110388538
Wang W, Ji Y, Yang W, Zhang C, Angwa L, Jin B et al (2020) Inhibitors of apoptosis proteins (IAPs) are associated with T-2 toxin-induced decreased collagen II in mouse chondrocytes in vitro. Toxicon. 176:34–43. https://doi.org/10.1016/j.toxicon.2020.01.002
Nguyen Ngoc TD, Son YO, Lim SS, Shi X, Kim JG, Heo JS, et al. (2012) Sodium fluoride induces apoptosis in mouse embryonic stem cells through ROS-dependent and caspase- and JNK-mediated pathways. Toxicol Appl Pharmacol [Internet]. 259(3):329–37. [cited 2020 Oct 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/22285274/. https://doi.org/10.1016/j.taap.2012.01.010
Varol E, Icli A, Aksoy F, Bas HA, Sutcu R, Ersoy IH, et al. (2013) Evaluation of total oxidative status and total antioxidant capacity in patients with endemic fluorosis. Toxicol Ind Health [Internet]. 29(2):175–80. [cited 2020 Oct 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/22155887/. https://doi.org/10.1177/0748233711428641
Manjunath A, Dayanand C, Muninarayana C, Vegi P (2014) Trends on oxidative stress and antioxidant status in fluoride affected areas in Kolar district, India. Int J Adv Med. 1(2):1. https://doi.org/10.5455/2349-3933.ijam20140805
Ravula S, Harinarayan C V., Prasad U V., Ramalakshmi T, Rupungudi A, Madrol V (2012) Effect of fluoride on reactive oxygen species and bone metabolism in postmenopausal women. Fluoride [Internet]. 45(2):108–15. [cited 2020 Oct 16]. Available from: www.ijmse.com
Redza-Dutordoir M, Averill-Bates DA (2016) Activation of apoptosis signalling pathways by reactive oxygen species. Biochimica et Biophysica Acta - Molecular Cell Research. Elsevier B.V. 1863:2977–2992. https://doi.org/10.1016/j.bbamcr.2016.09.012
Ghatage D, Gosavi S, Hazarey V, Ganvir S (2012) Apoptosis: molecular mechanism. J Orofac Sci [Internet] 4(2):103. [cited 2020 Oct 16]. Available from: http://www.jofs.in/text.asp?2012/4/2/103/106199. https://doi.org/10.4103/0975-8844.106199
Song C, Fu B, Zhang J, Zhao J, Yuan M, Peng W, et al. (2017) Sodium fluoride induces nephrotoxicity via oxidative stress-regulated mitochondrial SIRT3 signaling pathway. Sci Rep [Internet]. 7(1):1–15. [cited 2020 Oct 16]. Available from: www.nature.com/scientificreports/. https://doi.org/10.1038/s41598-017-00796-3
Geng Y, Qiu Y, Liu X, Chen X, Ding Y, Liu S et al (2014) Sodium fluoride activates ERK and JNK via induction of oxidative stress to promote apoptosis and impairs ovarian function in rats. J Hazard Mater. 272:75–82. https://doi.org/10.1016/j.jhazmat.2014.03.011
Karube H, Nishitai G, Inageda K, Kurosu H, Matsuoka M (2009) NaF activates MAPKs and induces apoptosis in odontoblast-like cells. J Dent Res [Internet]. 88(5):461–5. [cited 2020 Oct 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/19493891/. https://doi.org/10.1177/0022034509334771
Suzuki M, Bandoski C, Bartlett JD (2015) Fluoride induces oxidative damage and SIRT1/autophagy through ROS-mediated JNK signaling. Free Radic Biol Med [Internet]. 89:369–78. [cited 2020 Oct 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/26431905/. https://doi.org/10.1016/j.freeradbiomed.2015.08.015
Zhao L, Li J, Su J, Snead ML, Ruan J (2016) LS8 cell apoptosis induced by NaF through p-ERK and p-JNK — a mechanism study of dental fluorosis. Acta Odontol Scand [Internet]. 74(7):539–49. [cited 2020 Oct 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/27624793/. https://doi.org/10.1080/00016357.2016.1214980
Refsnes M, Schwarze PE, Holme JA, Låg M (2003) Fluoride-induced apoptosis in human epithelial lung cells (A549 cells): role of different G protein-linked signal systems. Hum Exp Toxicol [Internet]. 22(3):111–23. [cited 2020 Oct 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/12723891/. https://doi.org/10.1191/0960327103ht322oa
Agalakova NI, Gusev GP (2013) Transient activation of protein kinase C contributes to fluoride-induced apoptosis of rat erythrocytes. Toxicol Vitr. 27(8):2335–2341. https://doi.org/10.1016/j.tiv.2013.10.010
Li Y, Decker S, Yuan ZA, DenBesten PK, Aragon MA, Jordan-Sciutto K et al (2005) Effects of sodium fluoride on the actin cytoskeleton of murine ameloblasts. Arch Oral Biol. 50(8):681–688. https://doi.org/10.1016/j.archoralbio.2004.11.021
Deng H, Kuang P, Cui H, Luo Q, Liu H, Lu Y et al (2017) Sodium fluoride induces apoptosis in mouse splenocytes by activating ROS-dependent NF-κB signaling. Oncotarget 8:114428–114441. https://doi.org/10.18632/oncotarget.22826
Wang J, Xu H, Cheng X, Yang J, Yan Z, Ma H, et al. (2020) Calcium relieves fluoride-induced bone damage through the PI3K/AKT pathway. In: Food and Function [Internet]. Royal Society of Chemistry. [cited 2020 Nov 8]. 11(1):1155–1164. Available from: https://doi.org/10.1039/c9fo02491c
Wu J, Cheng M, Liu Q, Yang J, Wu S, Lu X, et al. (2015) Protective role of tert-butylhydroquinone against sodium fluoride-induced oxidative stress and apoptosis in PC12 cells. Cell Mol Neurobiol [Internet]. 35(7):1017–25. [cited 2020 Nov 8]. Available from: https://pubmed.ncbi.nlm.nih.gov/25911493/. https://doi.org/10.1007/s10571-015-0196-4
Tu W, Zhang Q, Liu Y, Han L, Wang Q, Chen P, et al. (2018) Fluoride induces apoptosis via inhibiting SIRT1 activity to activate mitochondrial p53 pathway in human neuroblastoma SH-SY5Y cells. Toxicol Appl Pharmacol [Internet]. 347:60–9. [cited 2020 Nov 8]. Available from: https://pubmed.ncbi.nlm.nih.gov/29609003/. https://doi.org/10.1016/j.taap.2018.03.030
Anuradha CD, Kanno S, Hirano S (2001) Oxidative damage to mitochondria is a preliminary step to caspase-3 activation in fluoride-induced apoptosis in HL-60 cells. Free Radic Biol Med. 31(3):367–373. https://doi.org/10.1016/S0891-5849(01)00591-3
Song JS, Lee HY, Lee E, Hwang HJ, Kim JH (2002) Cytotoxicity and apoptosis induction of sodium fluoride in human promyelocytic leukemia (HL-60) cells. Environ Toxicol Pharmacol. 11(2):85–91. https://doi.org/10.1016/S1382-6689(01)00108-9
Lee JH, Jung JY, Jeong YJ, Park JH, Yang KH, Choi NK et al (2008) Involvement of both mitochondrial- and death receptor-dependent apoptotic pathways regulated by Bcl-2 family in sodium fluoride-induced apoptosis of the human gingival fibroblasts. Toxicology. 243(3):340–347. https://doi.org/10.1016/j.tox.2007.10.026
Otsuki S, Morshed SRM, Chowdhury SA, Takayama F, Satoh T, Hashimoto K et al (2005) Possible link between glycolysis and apoptosis induced by sodium fluoride. J Dent Res. 84(10):919–923. https://doi.org/10.1177/154405910508401009
Chou R-H, Hsieh S-C, Yu Y-L, Huang M-H, Huang Y-C, Hsieh Y-H (2013) Fisetin inhibits migration and invasion of human cervical cancer cells by down-regulating urokinase plasminogen activator expression through suppressing the p38 MAPK-dependent NF-κB signaling pathway. Ahmad A, editor. PLoS One [Internet]. 8(8):e71983. [cited 2020 Nov 8]. Available from: https://dx.plos.org/10.1371/journal.pone.0071983. https://doi.org/10.1371/journal.pone.0071983
Tesoriere L, Attanzio A, Allegra M, Gentile C, Livrea MA (2013) Phytochemical indicaxanthin suppresses 7-ketocholesterol-induced THP-1 cell apoptosis by preventing cytosolic Ca2+ increase and oxidative stress. Br J Nutr. 110:230–240. https://doi.org/10.1017/S000711451200493X
Vauzour D, Vafeiadou K, Rice-Evans C, Williams RJ, Spencer JPE (2007) Activation of pro-survival Akt and ERK1/2 signalling pathways underlie the anti-apoptotic effects of flavanones in cortical neurons. J Neurochem [Internet]. 103(4):1355–67. [cited 2020 Nov 8]. Available from: https://pubmed.ncbi.nlm.nih.gov/17961201/. https://doi.org/10.1111/j.1471-4159.2007.04841.x
Yan W-J, Liu R-B, Wang L-K, Ma Y-B, Ding S-L, Deng F, et al. (2018) Sirt3-Mediated autophagy contributes to resveratrol-induced protection against ER stress in HT22 cells. Front Neurosci [Internet]. 12:116. [cited 2020 Nov 8]. Available from: http://journal.frontiersin.org/article/10.3389/fnins.2018.00116/full. https://doi.org/10.3389/fnins.2018.00116
Hannan MA, Dash R, Sohag AAM, Haque MN, Moon IS (2020) Neuroprotection against oxidative stress: phytochemicals targeting TrkB signaling and the Nrf2-ARE antioxidant system [Internet]. Front Mol Neurosci. Frontiers Media S.A. 13:116. [cited 2020 Nov 8]. Available from: www.frontiersin.org. https://doi.org/10.3389/fnmol.2020.00116
Shi N, Chen F, Zhang X, Clinton SK, Tang X, Sun Z, et al. (2017) Suppression of oxidative stress and NFκB/MAPK signaling by lyophilized black raspberries for esophageal cancer prevention in rats. Nutrients [Internet]. 9(4):413. [cited 2020 Nov 8]. Available from: /pmc/articles/PMC5409752/?report=abstract 10.3390/nu9040413
Zhai X, Lin M, Zhang F, Hu Y, Xu X, Li Y, et al. (2013) Dietary flavonoid genistein induces Nrf2 and phase II detoxification gene expression via ERKs and PKC pathways and protects against oxidative stress in Caco-2 cells. Mol Nutr Food Res [Internet]. 57(2):249–59. [cited 2020 Nov 8]. Available from: http://doi.wiley.com/10.1002/mnfr.201200536. https://doi.org/10.1002/mnfr.201200536
Meng D, Zhang P, Li S, Ho CT, Zhao H (2017) Antioxidant activity evaluation of dietary phytochemicals using Saccharomyces cerevisiae as a model. J Funct Foods. 38:36–44. https://doi.org/10.1016/j.jff.2017.08.041
Kuru P (2014) Tamarindus indica and its health related effects [Internet]. Asian Pac J Trop Biomed. Asian Pacific Tropical Biomedicine Press 4:676–81 [cited 2020 Sep 13]. Available from: https://www.sciencedirect.com/science/article/pii/S2221169115300885. https://doi.org/10.12980/APJTB.4.2014APJTB-2014-0173
Havinga RM, Hartl A, Putscher J, Prehsler S, Buchmann C, Vogl CR (2010) Tamarindus indica L. (Fabaceae): patterns of use in traditional African medicine. Journal of Ethnopharmacology. Elsevier 127:573–588. https://doi.org/10.1016/j.jep.2009.11.028
Murugan M, Subramanian E (2006) Studies on defluoridation of water by Tamarind seed, an unconventional biosorbent. J Water Health [Internet]. 4(4):453–61. [cited 2020 Sep 14]. Available from: http://iwaponline.com/jwh/article-pdf/4/4/453/396531/453.pdf. https://doi.org/10.2166/wh.2006.0029
Kumar NP, Kumar NS, Krishnaiah A (2012) Defluoridation of water using Tamarind (tamarindus indica) fruit Cover: kinetics and equilibrium studies. J Chil Chem Soc [Internet]. 57(3):1224–31. [cited 2020 Sep 14]. Available from: https://scielo.conicyt.cl/scielo.php?script=sci_arttext&pid=S0717-97072012000300006&lng=es&nrm=iso&tlng=en. https://doi.org/10.4067/S0717-97072012000300006
Ranjan R, Swarup D, Patra RC, Chandra V (2009) Tamarindus indica L. and moringa oleifera m. extract administration ameliorates fluoride toxicity in rabbits. Indian J Exp Biol [Internet]. 47(11):900–905 Available from: https://www.researchgate.net/publication/41137780_Tamarindus_indica_L_and_Moringa_oleifera_M_extract_administration_ameliorates_fluoride_toxicity_in_rabbits
Dey S, Swarup D, Saxena A, Dan A (2011) In vivo efficacy of tamarind (Tamarindus indica) fruit extract on experimental fluoride exposure in rats. Res Vet Sci [Internet]. 91(3):422–5 [cited 2020 Sep 14]. Available from: https://pubmed.ncbi.nlm.nih.gov/20980037/. https://doi.org/10.1016/j.rvsc.2010.09.013
Sudjaroen Y, Haubner R, Würtele G, Hull WE, Erben G, Spiegelhalder B et al (2005) Isolation and structure elucidation of phenolic antioxidants from Tamarind (Tamarindus indica L.) seeds and pericarp. Food Chem Toxicol. 43(11):1673–1682. https://doi.org/10.1016/j.fct.2005.05.013
Khandare AL, Rao GS, Lakshmaiah N (2002) Effect of tamarind ingestion on fluoride excretion in humans. Eur J Clin Nutr [Internet]. 56(1):82–5. [cited 2020 Sep 14]. Available from: https://www.nature.com/articles/1601287. https://doi.org/10.1038/sj.ejcn.1601287
Bowen-Forbes CS, Zhang Y, Nair MG (2010) Anthocyanin content, antioxidant, anti-inflammatory and anticancer properties of blackberry and raspberry fruits. J Food Compos Anal. 23:554–560. https://doi.org/10.1016/j.jfca.2009.08.012
U.S. Department of Agriculture (2019) Agricultural Research Service. FoodData Central [Internet]. FoodData Central. Blackberries, raw. [cited 2020 Sep 14]. p. fdc.nal.usda.gov. Available from: https://fdc.nal.usda.gov/fdc-app.html#/food-details/786757/nutrients
Khoo HE, Azlan A, Tang ST, Lim SM (2017) Anthocyanidins and anthocyanins: colored pigments as food, pharmaceutical ingredients, and the potential health benefits [Internet]. Food Nutr Res Swedish Nutrition Foundation 61:1361779. [cited 2020 Sep 15]. Available from: /pmc/articles/PMC5613902/?report=abstract. https://doi.org/10.1080/16546628.2017.1361779
Cervoni B (2020) Blackberry Nutrition Facts and Health Benefits [Internet]. [cited 2020 Sep 14]. Available from: https://www.verywellfit.com/blackberry-nutrition-facts-calories-and-health-benefits-4109221
Kähkönen MP, Hopia AI, Heinonen M (2001) Berry phenolics and their antioxidant activity. J Agric Food Chem [Internet] 49(8):4076–4082. Available from:. https://doi.org/10.1021/jf010152t10.1021/jf010152t
Heinonen M (2007) Antioxidant activity and antimicrobial effect of berry phenolics — a Finnish perspective. Mol Nutr Food Res. 51(6):684–691. https://doi.org/10.1002/mnfr.200700006
Paredes-López O, Cervantes-Ceja ML, Vigna-Pérez M, Hernández-Pérez T (2010) Berries: Improving human health and healthy aging, and promoting quality life—a review. Plant Foods Hum Nutr [Internet]. 65(3):299–308. Available from: https://www.researchgate.net/publication/45276258_Berries_Improving_Human_Health_and_Healthy_Aging_and_Promoting_Quality_Life-A_Review. https://doi.org/10.1007/s11130-010-0177-1
Zhang H, Zhang H, Abid S, Ahn JC, Mathiyalagan R, Kim YJ, et al. (2020) Characteristics of Panax ginseng cultivars in Korea and China [Internet]. Molecules. MDPI AG 25:25 [cited 2020 Sep 15]x. Available from: /pmc/articles/PMC7321059/?report=abstract. https://doi.org/10.3390/molecules25112635
Kim YJ, Zhang D, Yang DC (2015) Biosynthesis and biotechnological production of ginsenosides [Internet]. Biotechnol Adv Elsevier Inc. 33:717–35 [cited 2020 Sep 15]. Available from: https://pubmed.ncbi.nlm.nih.gov/25747290/. https://doi.org/10.1016/j.biotechadv.2015.03.001
Fernández-Moriano C, González-Burgos E, Iglesias I, Lozano R, Gómez-Serranillos MP (2017) Evaluation of the adaptogenic potential exerted by ginsenosides Rb1 and Rg1 against oxidative stress-mediated neurotoxicity in an in vitro neuronal model. PLoS One [Internet]. 12(8):e0182933 [cited 2020 Sep 16]. Available from: /pmc/articles/PMC5558939/?report=abstract. https://doi.org/10.1371/journal.pone.0182933
Yao F, Xue Q, Li K, Cao X, Sun L, Liu Y (2019) Phenolic compounds and ginsenosides in ginseng shoots and their antioxidant and anti-inflammatory capacities in lps-induced raw264.7 mouse macrophages. Int J Mol Sci [Internet]. 20(12):2951. [cited 2020 Sep 16]. Available from: /pmc/articles/PMC6627944/?report=abstract. https://doi.org/10.3390/ijms20122951
Shen CY, Jiang JG, Yang L, Wang DW, Zhu W (2017) Anti-ageing active ingredients from herbs and nutraceuticals used in traditional Chinese medicine: pharmacological mechanisms and implications for drug discovery. Br J Pharmacol. 174(11):1395–1425. https://doi.org/10.1111/bph.13631
Choi KS, Song H, Kim EH, Choi JH, Hong H, Han YM et al (2012) Inhibition of hydrogen sulfide-induced angiogenesis and inflammation in vascular endothelial cells: Potential mechanisms of gastric cancer prevention by Korean red ginseng. J Ginseng Res. 36(2):135–145. https://doi.org/10.5142/jgr.2012.36.2.135
Jin TY, Rong PQ, Liang HY, Zhang PP, Zheng GQ, Lin Y (2020) Clinical and preclinical systematic review of Panax ginseng C. A. mey and its compounds for fatigue. Front Pharmacol [Internet]. 11:1031. [cited 2020 Sep 16]. Available from: /pmc/articles/PMC7379339/?report=abstract. https://doi.org/10.3389/fphar.2020.01031
Mancuso C, Santangelo R (2017) Panax ginseng and Panax quinquefolius: from pharmacology to toxicology. Food Chem Toxicol [Internet]. 107:362. [cited 2020 Sep 16]. Available from: /pmc/articles/PMC7116968/?report=abstract. https://doi.org/10.1016/J.FCT.2017.07.019
Lee YM, Yoon H, Park HM, Song BC, Yeum KJ (2017) Implications of red Panax ginseng in oxidative stress associated chronic diseases [Internet]. J Ginseng Res Elsevier B.V. 41:113–9. [cited 2020 Sep 17]. Available from: /pmc/articles/PMC5386131/?report=abstract. https://doi.org/10.1016/j.jgr.2016.03.003
Bak MJ, Jun M, Jeong WS (2012) Antioxidant and hepatoprotective effects of the red ginseng essential oil in H 2O 2-treated HepG2 cells and CCL 4-treated mice. Int J Mol Sci [Internet]. 13(2):2314–30. [cited 2020 Sep 17]. Available from: /pmc/articles/PMC3292025/?report=abstract. https://doi.org/10.3390/ijms13022314
Park HM, Kim SJ, Mun AR, Go HK, Kim GB, Kim SZ, et al. 2012 Korean red ginseng and its primary ginsenosides inhibit ethanol-induced oxidative injury by suppression of the MAPK pathway in TIB-73 cells. J Ethnopharmacol [Internet]. 141(3):1071–6. [cited 2020 Sep 17]. Available from: https://pubmed.ncbi.nlm.nih.gov/22472111/. https://doi.org/10.1016/j.jep.2012.03.038
Park SH, Jang JH, Chen CY, Na HK, Surh YJ (2010) A formulated red ginseng extract rescues PC12 cells from PCB-induced oxidative cell death through Nrf2-mediated upregulation of heme oxygenase-1 and glutamate cysteine ligase. Toxicology [Internet]. 278(1):131–9. [cited 2020 Sep 17]. Available from: https://pubmed.ncbi.nlm.nih.gov/20399244/. https://doi.org/10.1016/j.tox.2010.04.003
Choubey S, Varughese LRache, Kumar V, Beniwal V (2015) Medicinal importance of gallic acid and its ester derivatives: a patent review [Internet]. Pharm Patent Anal Pharm Pat Anal 4:305–15. [cited 2020 Sep 18]. Available from: https://pubmed.ncbi.nlm.nih.gov/26174568/. https://doi.org/10.4155/ppa.15.14
Kahkeshani N, Farzaei F, Fotouhi M, Alavi SS, Bahramsoltani R, Naseri R, et al. (2019) Pharmacological effects of gallic acid in health and disease: a mechanistic review. Iran J Basic Med Sci [Internet]. 22(3):225–37. [cited 2020 Sep 18]. Available from: /pmc/articles/PMC6528712/?report=abstract. https://doi.org/10.22038/ijbms.2019.32806.7897
Badhani B, Sharma N, Kakkar R (2015) Gallic acid: A versatile antioxidant with promising therapeutic and industrial applications. RSC Adv [Internet]. 5(35):27540–57. [cited 2020 Sep 18]. Available from: https://pubs.rsc.org/en/content/articlehtml/2015/ra/c5ra01911g. 10.1039/c5ra01911g
Gao J, Hu J, Hu D, Yang X (2019) A role of gallic acid in oxidative damage diseases: a comprehensive review. Nat Prod Commun [Internet]. 14(8):1934578X1987417. [cited 2020 Sep 18]. Available from: http://journals.sagepub.com/doi/10.1177/1934578X19874174. https://doi.org/10.1177/1934578X19874174
Kongpichitchoke T, Chiu MT, Huang TC, Hsu JL (2016) Gallic acid content in taiwanese teas at different degrees of fermentation and its antioxidant activity by inhibiting pkcδ activation: In vitro and in silico studies. Molecules [Internet]. 21(10):1346. [cited 2020 Sep 18]. Available from: /pmc/articles/PMC6273586/?report=abstract. https://doi.org/10.3390/molecules21101346
Scassellati-Sforzolini G, Villarini M, Moretti M, Marcarelli M, Pasquini R, Fatigoni C et al (1999) Antigenotoxic properties of Terminalia arjuna bark extracts. J Environ Pathol Toxicol Oncol. 18(2):119–125
Amalraj A, Gopi S (2017) Medicinal properties of Terminalia arjuna (Roxb.) Wight & Arn.: A review [Internet]. J Tradit Complement Med National Taiwan University 7:65–78. [cited 2020 Sep 18]. Available from: /pmc/articles/PMC5198828/?report=abstract. https://doi.org/10.1016/j.jtcme.2016.02.003
Ramya EM, Kumar GP, Anand T, Anilakumar KR (2017) Modulatory effects of Terminalia arjuna against domoic acid induced toxicity in Caco-2 cell line. Cytotechnology [Internet]. 69(4):725–39. [cited 2020 Sep 19]. Available from: /pmc/articles/PMC5507850/?report=abstract. https://doi.org/10.1007/s10616-017-0080-9
Gupta R, Singhal S, Goyle A, Sharma VN (2001) Antioxidant and hypocholesterolaemic effects of Terminalia arjuna tree-bark powder: a randomised placebo-controlled trial. J Assoc Physicians India. 49:231–235
Subramaniam S, Ramachandran S, Uthrapathi S, Gnamanickam VR, Dubey GP (2011) Anti-hyperlipidemic and antioxidant potential of different fractions of Terminalia arjuna Roxb. bark against PX-407 induced hyperlipidemia. Indian J Exp Biol. 49(4):282–288
Kumar S, Enjamoori R, Jaiswal A, Ray R, Seth S, Maulik SK (2009) Catecholamine-induced myocardial fibrosis and oxidative stress is attenuated by <I>Terminalia arjuna</I> (Roxb.). J Pharm Pharmacol [Internet] 61(11):1529–36. [cited 2020 Sep 18]. Available from: https://pubmed.ncbi.nlm.nih.gov/19903379/. https://doi.org/10.1211/jpp/61.11.0013
Mandal S, Patra A, Samanta A, Roy S, Mandal A, Mahapatra T Das, et al. (2013) Analysis of phytochemical profile of Terminalia arjuna bark extract with antioxidative and antimicrobial properties. Asian Pac J Trop Biomed [Internet]. 3(12):960–6. [cited 2020 Sep 18]. Available from: /pmc/articles/PMC3805097/?report=abstract. https://doi.org/10.1016/S2221-1691(13)60186-0
Manu TM, Anand T, Pandareesh MD, Kumar PB, Khanum F (2019) Terminalia arjuna extract and arjunic acid mitigate cobalt chloride–induced hypoxia stress–mediated apoptosis in H9c2 cells. Naunyn Schmiedebergs Arch Pharmacol. 392(9):1107–1119. https://doi.org/10.1007/s00210-019-01654-x
Ghosh J, Das J, Manna P, Sil PC (2010) Protective effect of the fruits of Terminalia arjuna against cadmium-induced oxidant stress and hepatic cell injury via MAPK activation and mitochondria dependent pathway. Food Chem. 123(4):1062–1075. https://doi.org/10.1016/j.foodchem.2010.05.062
Thangaraju MM, Tamatam A, Bhat P V., Deshetty UM, Babusha ST, Khanum F (2020) Terminalia arjuna extract attenuates isoproterenol-induced cardiac stress in Wistar rats via an anti-apoptotic pathway. Proc Natl Acad Sci India Sect B - Biol Sci [Internet]. 1–12. [cited 2020 Sep 19]. Available from: https://link.springer.com/article/10.1007/s40011-020-01180-4. https://doi.org/10.1007/s40011-020-01180-4
Lakhanpal P, Rai DK (2007) Quercetin: a versatile flavonoid. Internet J Med Updat - EJOURNAL [Internet]. 2(2). [cited 2020 Sep 22]. Available from: https://www.akspublication.com/paper05_jul-dec2007.htm. https://doi.org/10.4314/ijmu.v2i2.39851
Costa LG, Garrick JM, Roquè PJ, Pellacani C (2016) Mechanisms of neuroprotection by quercetin: counteracting oxidative stress and more, vol 2016. Oxidative Medicine and Cellular Longevity. Hindawi Limited. https://doi.org/10.1155/2016/2986796
El-Saber Batiha G, Beshbishy AM, Ikram M, Mulla ZS, Abd El-Hack ME, Taha AE, et al. (2020) The pharmacological activity, biochemical properties, and pharmacokinetics of the major natural polyphenolic flavonoid: Quercetin [Internet]. Foods. MDPI Multidisciplinary Digital Publishing Institute 9:374 [cited 2020 Sep 23]. Available from: /pmc/articles/PMC7143931/?report=abstract. https://doi.org/10.3390/foods9030374
Anand David AV, Arulmoli R, Parasuraman S (2016) Overviews of biological importance of quercetin: a bioactive flavonoid [Internet]. Pharmacognosy Reviews. Medknow Publications 10:84–9 [cited 2020 Sep 22]. Available from: /pmc/articles/PMC5214562/?report=abstract. https://doi.org/10.4103/0973-7847.194044
Denny Joseph KM, Muralidhara (2013) Enhanced neuroprotective effect of fish oil in combination with quercetin against 3-nitropropionic acid induced oxidative stress in rat brain. Prog Neuro-Psychopharmacology Biol Psychiatry [Internet] 40(1):83–92. [cited 2020 Sep 22]. Available from: https://pubmed.ncbi.nlm.nih.gov/22960609/. https://doi.org/10.1016/j.pnpbp.2012.08.018
Bao D, Wang J, Pang X, Liu H (2017) Protective effect of quercetin against oxidative stress-induced cytotoxicity in rat pheochromocytoma (PC-12) cells. Molecules [Internet] 22(7):1122. [cited 2020 Sep 22]. Available from: https://pubmed.ncbi.nlm.nih.gov/28684704/. https://doi.org/10.3390/molecules22071122
Boots AW, Drent M, de Boer VCJ, Bast A, Haenen GRMM (2011) Quercetin reduces markers of oxidative stress and inflammation in sarcoidosis. Clin Nutr. 30(4):506–512. https://doi.org/10.1016/j.clnu.2011.01.010
Kook D, Wolf AH, Yu AL, Neubauer AS, Priglinger SG, Kampik A et al (2008) The protective effect of quercetin against oxidative stress in the human RPE in vitro. Investig Ophthalmol Vis Sci. 49(4):1712–1720. https://doi.org/10.1167/iovs.07-0477
Afanas’ev IB, Dcrozhko AI, Brodskii A V., Kostyuk VA, Potapovitch AI (1989) Chelating and free radical scavenging mechanisms of inhibitory action of rutin and quercetin in lipid peroxidation. Biochem Pharmacol [Internet]. 38(11):1763–9. [cited 2020 Sep 23]. Available from: https://pubmed.ncbi.nlm.nih.gov/2735934/. https://doi.org/10.1016/0006-2952(89)90410-3
Ferrali M, Signorini C, Caciotti B, Sugherini L, Ciccoli L, Giachetti D, et al. (1997) Protection against oxidative damage of erythrocyte membrane by the flavonoid quercetin and its relation to iron chelating activity. FEBS Lett [Internet]. 416(2):123–9. [cited 2020 Sep 23]. Available from: http://doi.wiley.com/10.1016/S0014-5793%2897%2901182-4. https://doi.org/10.1016/S0014-5793(97)01182-4
Sorata Y, Takahama U, Kimura M (1984) Protective effect of quercetin and rutin on photosensitized lysis of human erythrocytes in the presence of hematoporphyrin. BBA - Gen Subj [Internet]. 799(3):313–7. [cited 2020 Sep 23]. Available from: https://pubmed.ncbi.nlm.nih.gov/6733152/. https://doi.org/10.1016/0304-4165(84)90276-9
Shoskes D, Lapierre C, Cruz-Corerra M, Muruve N, Rosario R, Fromkin B et al (2005) Beneficial effects of the bioflavonoids curcumin and quercetin on early function in cadaveric renal transplantation: a randomized placebo controlled trial. Transplantation. 80(11):1556–1559. https://doi.org/10.1097/01.tp.0000183290.64309.21
Nagao A, Seki M, Kobayashi H (1999) Inhibition of xanthine oxidase by flavonoids. Biosci Biotechnol Biochem [Internet]. 63(10):1787–90. [cited 2020 Sep 23]. Available from: https://www.tandfonline.com/action/journalInformation?journalCode=tbbb20. https://doi.org/10.1271/bbb.63.1787
Iio M, Ono Y, Kai S, Fukumoto M (1986) Effects of Flavonoids on xanthine oxidation as well as on cytochrome c reduction by milk xanthine oxidase. J Nutr Sci Vitaminol (Tokyo) [Internet]. 32(6):635–42. [cited 2020 Sep 23]. Available from: https://pubmed.ncbi.nlm.nih.gov/3035152/. https://doi.org/10.3177/jnsv.32.635
Karabulut E, Otlu O, Pakdemirli A, Yarım M, Salt A, Cenesiz S. The effects of quercetin on the fluorosis toxicity in kidney of mice. 2019.
Colunga Biancatelli RML, Berrill M, Catravas JD, Marik PE (2020) Quercetin and vitamin C: an experimental, synergistic therapy for the prevention and treatment of SARS-CoV-2 Related Disease (COVID-19) [Internet]. Front Immunol Frontiers Media S.A. 11:1451. [cited 2020 Sep 23]. Available from: www.frontiersin.org. https://doi.org/10.3389/fimmu.2020.01451
Dias AS, Porawski M, Alonso M, Marroni N, Collado PS, González-Gallego J (2005) Quercetin decreases oxidative stress, NF-κB activation, and iNOS overexpression in liver of streptozotocin-induced diabetic rats. J Nutr. 135(10):2299–2304. https://doi.org/10.1093/jn/135.10.2299
Sánchez-González P, López-Hernández FJ, Prierto M, Vicente-Vicente L, López-Novoa JM, Morales AI (2010) Renoprotective effects of quercetin on cisplatin-induced nephrotoxicity. Role of TNF-alpha, NF-kappaB and iNOS. Toxicol Lett [Internet]. 196(196):S242. [cited 2020 Sep 23]. Available from: https://www.infona.pl//resource/bwmeta1.element.elsevier-3a243135-2be6-3e14-9e30-e4114bd312e6. https://doi.org/10.1016/j.toxlet.2010.03.809
National Center for Biotechnology Information (n.d.). PubChem Compound Summary for CID 446925, Lycopene. [Internet]. [cited 2020 Sep 23]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Lycopene
Juturu V (2013) Polyphenols and cardiometabolic syndrome. In: Polyphenols in Human Health and Disease. Elsevier Inc., pp 1067–1076. https://doi.org/10.1016/B978-0-12-398456-2.00082-7
Hedayati N, Naeini MB, Nezami A, Hosseinzadeh H, Wallace Hayes A, Hosseini S, et al. (2019) Protective effect of lycopene against chemical and natural toxins: a review [Internet]. BioFactors. Blackwell Publishing Inc. 45:5–23. [cited 2020 Sep 23]. Available from: https://pubmed.ncbi.nlm.nih.gov/30339717/. https://doi.org/10.1002/biof.1458
Przybylska S (2020) Lycopene – a bioactive carotenoid offering multiple health benefits: a review. Int J Food Sci Technol [Internet]. 55(1):11–32. [cited 2020 Sep 23]. Available from: https://onlinelibrary.wiley.com/doi/abs/10.1111/ijfs.14260. https://doi.org/10.1111/ijfs.14260
Mozos I, Stoian D, Caraba A, Malainer C, Horbanczuk JO, Atanasov AG (2018) Lycopene and vascular health [Internet]. Front Pharmacol Frontiers Media S.A. 9:521. [cited 2020 Sep 23]. Available from: /pmc/articles/PMC5974099/?report=abstract. https://doi.org/10.3389/fphar.2018.00521
Zhu R, Chen B, Bai Y, Miao T, Rui L, Zhang H, et al (2020) Lycopene in protection against obesity and diabetes: a mechanistic review. Pharmacol Res 159:104966. https://doi.org/10.1016/j.phrs.2020.104966
Story EN, Kopec RE, Schwartz SJ, Keith Harris G (2010) An update on the health effects of tomato lycopene. Annu Rev Food Sci Technol [Internet]. 1(1):189–210. [cited 2020 Sep 23]. Available from: /pmc/articles/PMC3850026/?report=abstract. https://doi.org/10.1146/annurev.food.102308.124120
Yue R, Hu H, Yiu KH, Luo T, Zhou Z, Xu L, et al. 2012 Lycopene protects against hypoxia/reoxygenation-induced apoptosis by preventing mitochondrial dysfunction in primary neonatal mouse cardiomyocytes. Gallyas F, editor. PLoS One [Internet]. 7(11):e50778. [cited 2020 Sep 23];. Available from: https://dx.plos.org/10.1371/journal.pone.0050778. https://doi.org/10.1371/journal.pone.0050778
Goncu T, Oğuz E, Sezen H, Koçarslan S, Oğuz H, Akal A, et al. (2016) Anti-inflammatory effect of lycopene on endotoxin-induced uveitis in rats. Arq Bras Oftalmol [Internet]. 79(6):357–62. [cited 2020 Sep 23]. Available from: https://pubmed.ncbi.nlm.nih.gov/28076559/. https://doi.org/10.5935/0004-2749.20160102
Chernyshova MP, Pristenskiy DV, Lozbiakova MV, Chalyk NE, Bandaletova TY, Petyaev IM (2019) Systemic and skin-targeting beneficial effects of lycopene-enriched ice cream: A pilot study. J Dairy Sci [Internet] 102(1):14–25. [cited 2020 Sep 23]. Available from. https://doi.org/10.3168/jds.2018-15282
Grether-Beck S, Marini A, Jaenicke T, Stahl W, Krutmann J (2017) Molecular evidence that oral supplementation with lycopene or lutein protects human skin against ultraviolet radiation: results from a double-blinded, placebo-controlled, crossover study. Br J Dermatol [Internet]. 176(5):1231–40. [cited 2020 Sep 23]. Available from: http://doi.wiley.com/10.1111/bjd.15080. https://doi.org/10.1111/bjd.15080
Russo C, Ferro Y, Maurotti S, Salvati MA, Mazza E, Pujia R, et al. (2020) Lycopene and bone: an in vitro investigation and a pilot prospective clinical study. J Transl Med [Internet]. 18(1):43. [cited 2020 Sep 23]. Available from: https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-020-02238-7. https://doi.org/10.1186/s12967-020-02238-7
Jiang W, Guo MH, Hai X (2016) Hepatoprotective and antioxidant effects of lycopene on non-alcoholic fatty liver disease in rat. World J Gastroenterol [Internet]. 22(46):10180–8. [cited 2020 Sep 23]. Available from: /pmc/articles/PMC5155177/?report=abstract. https://doi.org/10.3748/wjg.v22.i46.10180
Chen D, Huang C, Chen Z (2019) A review for the pharmacological effect of lycopene in central nervous system disorders. Biomedicine and Pharmacotherapy. Elsevier Masson SAS 111:791–801. https://doi.org/10.1016/j.biopha.2018.12.151
Durairajanayagam D, Agarwal A, Ong C, Prashast P (2014) Lycopene and male infertility [Internet]. Asian J Androl Medknow Publications. 16:420–5 [cited 2020 Sep 23]. Available from: /pmc/articles/PMC4023371/?report=abstract. https://doi.org/10.4103/1008-682X.126384
Zhao B, Ren B, Guo R, Zhang W, Ma S, Yao Y, et al. (2017) Supplementation of lycopene attenuates oxidative stress induced neuroinflammation and cognitive impairment via Nrf2/NF-κB transcriptional pathway. Food Chem Toxicol [Internet]. 109(Pt 1):505–16. [cited 2020 Sep 23]. Available from: https://pubmed.ncbi.nlm.nih.gov/28974442/. https://doi.org/10.1016/j.fct.2017.09.050
Bandeira ACB, da Silva TP, de Araujo GR, Araujo CM, da Silva RC, Lima WG, et al. (2017) Lycopene inhibits reactive oxygen species production in SK-Hep-1 cells and attenuates acetaminophen-induced liver injury in C57BL/6 mice. Chem Biol Interact [Internet]. 263:7–17. [cited 2020 Sep 23]. Available from: https://pubmed.ncbi.nlm.nih.gov/27989599/. https://doi.org/10.1016/j.cbi.2016.12.011
Xu XY, Meng X, Li S, Gan RY, Li Y, Li H Bin (2018) Bioactivity, health benefits, and related molecular mechanisms of curcumin: current progress, challenges, and perspectives [Internet]. Nutrients. MDPI AG. 10:1553. [cited 2020 Oct 13]. Available from: /pmc/articles/PMC6213156/?report=abstract. https://doi.org/10.3390/nu10101553
Pulido-Moran M, Moreno-Fernandez J, Ramirez-Tortosa C, Ramirez-Tortosa MC (2016) Curcumin and health [Internet]. Molecules. MDPI AG. 21:264. [cited 2020 Oct 13]. Available from: /pmc/articles/PMC6273481/?report=abstract. https://doi.org/10.3390/molecules21030264
Tomeh MA, Hadianamrei R, Zhao X (2019) A review of curcumin and its derivatives as anticancer agents [Internet]. Int J Mol Sci MDPI AG. 20:1033. [cited 2020 Oct 13]. Available from: /pmc/articles/PMC6429287/?report=abstract. https://doi.org/10.3390/ijms20051033
Grama CN, Suryanarayana P, Patil MA, Raghu G, Balakrishna N, Ravi Kumar MNV, et al. (2013) Efficacy of biodegradable curcumin nanoparticles in delaying cataract in diabetic rat model. PLoS One [Internet]. 8(10):e78217. [cited 2020 Oct 13]. Available from: https://pubmed.ncbi.nlm.nih.gov/24155984/. https://doi.org/10.1371/journal.pone.0078217
Wu J, Li Q, Wang X, Yu S, Li L, Wu X, et al. (2013) Neuroprotection by Curcumin in ischemic brain injury involves the Akt/Nrf2 pathway. PLoS One [Internet]. 8(3):e59843. [cited 2020 Oct 13]. Available from: https://pubmed.ncbi.nlm.nih.gov/23555802/. https://doi.org/10.1371/journal.pone.0059843
Suryanarayana P, Satyanarayana A, Balakrishna N, Kumar PU, Bhanuprakash RG (2007) Effect of turmeric and curcumin on oxidative stress and antioxidant enzymes in streptozotocin-induced diabetic rat. Med Sci Monit. 13(12):BR286–BR292
Burge K, Gunasekaran A, Eckert J, Chaaban H (2019) Curcumin and intestinal inflammatory diseases: molecular mechanisms of protection. Int J Mol Sci [Internet]. 20(8):1912. [cited 2020 Oct 13]. Available from: /pmc/articles/PMC6514688/?report=abstract. https://doi.org/10.3390/ijms20081912
Jagetia GC, Aggarwal BB (2007) “Spicing up” of the immune system by curcumin [Internet]. Journal of Clinical Immunology. Springer. 27:19–35. [cited 2020 Oct 13]. Available from: https://link.springer.com/article/10.1007/s10875-006-9066-7. https://doi.org/10.1007/s10875-006-9066-7
Dai C, Tang S, Li D, Zhao K, Xiao X (2015) Curcumin attenuates quinocetone-induced oxidative stress and genotoxicity in human hepatocyte L02 cells. Toxicol Mech Methods [Internet]. 25(4):340–6. [cited 2020 Oct 13]. Available from: https://www.tandfonline.com/doi/abs/10.3109/15376516.2015.1045659. https://doi.org/10.3109/15376516.2015.1045659
Khurana S, Venkataraman K, Hollingsworth A, Piche M, Tai TC (2013) Polyphenols: benefits to the cardiovascular system in health and in aging [Internet]. Nutrients. MDPI AG. 5:3779–827. [cited 2020 Oct 13]. Available from: /pmc/articles/PMC3820045/?report=abstract. https://doi.org/10.3390/nu5103779
Li Y, Li J, Li S, Li Y, Wang X, Liu B, et al. (2015) Curcumin attenuates glutamate neurotoxicity in the hippocampus by suppression of ER stress-associated TXNIP/NLRP3 inflammasome activation in a manner dependent on AMPK. Toxicol Appl Pharmacol [Internet]. 286(1):53–63. [cited 2020 Oct 13]. Available from: https://pubmed.ncbi.nlm.nih.gov/25791922/. https://doi.org/10.1016/j.taap.2015.03.010
Fu X Yan, Yang M Feng, Cao M Zhi, Li D Wei, Yang X Yi, Sun J Yi, et al. (2016) Strategy to suppress oxidative damage-induced neurotoxicity in PC12 cells by curcumin: the role of ROS-mediated DNA damage and the MAPK and AKT pathways. Mol Neurobiol [Internet]. 53(1):369–78. [cited 2020 Oct 13]. Available from: https://link.springer.com/article/10.1007/s12035-014-9021-1. https://doi.org/10.1007/s12035-014-9021-1
Pulla Reddy AC, Lokesh BR (1994) Effect of dietary turmeric (curcuma longa) on iron-induced lipid peroxidation in the rat liver. Food Chem Toxicol [Internet]. 32(3):279–83. [cited 2020 Oct 13]. Available from: https://pubmed.ncbi.nlm.nih.gov/8157223/. https://doi.org/10.1016/0278-6915(94)90201-1
Sökmen M, Akram Khan M (2016) The antioxidant activity of some curcuminoids and chalcones. Inflammopharmacology [Internet]. 24(2–3):81–6. [cited 2020 Oct 13];. Available from: /pmc/articles/PMC4883448/?report=abstract. https://doi.org/10.1007/s10787-016-0264-5
Priyadarsini KI, Maity DK, Naik GH, Kumar MS, Unnikrishnan MK, Satav JG et al (2003) Role of phenolic O-H and methylene hydrogen on the free radical reactions and antioxidant activity of curcumin. Free Radic Biol Med. 35(5):475–484. https://doi.org/10.1016/S0891-5849(03)00325-3
Hackett ES, Twedt DC, Gustafson DL (2013) Milk thistle and its derivative compounds: a review of opportunities for treatment of liver disease [Internet]. J Vet Intern Med. 27:10–6. [cited 2020 Oct 14]. Available from: http://doi.wiley.com/10.1111/jvim.12002. https://doi.org/10.1111/jvim.12002
Bijak M (2017) Silybin, a major bioactive component of milk thistle (Silybum marianum L. Gaernt.)—chemistry, bioavailability, and metabolism [Internet]. Molecules. MDPI AG. 22:1942. [cited 2020 Oct 15]. Available from: /pmc/articles/PMC6150307/?report=abstract. https://doi.org/10.3390/molecules22111942
Anthony K, Subramanya G, Uprichard S, Hammouda F, Saleh M (2013) Antioxidant and anti-hepatitis C viral activities of commercial milk thistle food supplements. Antioxidants. 2(1):23–36. https://doi.org/10.3390/antiox2010023
Surai PF (2015) Silymarin as a natural antioxidant: an overview of the current evidence and perspectives. Antioxidants [Internet]. 4(1):204–47. [cited 2020 Oct 15]. Available from: /pmc/articles/PMC4665566/?report=abstract. https://doi.org/10.3390/antiox4010204
Chtourou Y, Fetoui H, Sefi M, Trabelsi K, Barkallah M, Boudawara T, et al. (2010) Silymarin, a natural antioxidant, protects cerebral cortex against manganese-induced neurotoxicity in adult rats. BioMetals [Internet]. 23(6):985–96. [cited 2020 Oct 15]. Available from: https://link.springer.com/article/10.1007/s10534-010-9345-x. https://doi.org/10.1007/s10534-010-9345-x
Jia R, Cao L, Du J, Xu P, Jeney G, Yin G (2013) The protective effect of silymarin on the carbon tetrachloride (CCl 4)-induced liver injury in common carp (Cyprinus carpio). Vitr Cell Dev Biol - Anim [Internet]. 49(3):155–61. [cited 2020 Oct 15]. Available from: https://pubmed.ncbi.nlm.nih.gov/23435858/. https://doi.org/10.1007/s11626-013-9587-3
Ghosh S, Sarkar A, Bhattacharyya S, Sil PC (2016) Silymarin protects mouse liver and kidney from thioacetamide induced toxicity by scavenging reactive oxygen species and activating PI3K-Akt pathway. Front Pharmacol [Internet]. 7(DEC):481. [cited 2020 Oct 15]. Available from: /pmc/articles/PMC5156955/?report=abstract. https://doi.org/10.3389/fphar.2016.00481
Chtourou Y, Garoui EM, Boudawara T, Zeghal N (2013) Therapeutic efficacy of silymarin from milk thistle in reducing manganese-induced hepatic damage and apoptosis in rats. Hum Exp Toxicol [Internet]. 32(1):70–81. [cited 2020 Oct 15]. Available from: https://pubmed.ncbi.nlm.nih.gov/22899727/. https://doi.org/10.1177/0960327112455674
Patel N, Joseph C, Corcoran GB, Ray SD (2010) Silymarin modulates doxorubicin-induced oxidative stress, Bcl-xL and p53 expression while preventing apoptotic and necrotic cell death in the liver. Toxicol Appl Pharmacol. 245(2):143–152. https://doi.org/10.1016/j.taap.2010.02.002
Singh BN, Shankar S, Srivastava RK (2011) Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications [Internet]. Biochem Pharmacol. Elsevier Inc. 82:1807–21. [cited 2020 Oct 14]. Available from: /pmc/articles/PMC4082721/?report=abstract. https://doi.org/10.1016/j.bcp.2011.07.093
He J, Xu L, Yang L, Wang X (2018) Epigallocatechin gallate is the most effective catechin against antioxidant stress via hydrogen peroxide and radical scavenging activity. Med Sci Monit [Internet]. 24:8198–206. [cited 2020 Oct 14]. Available from: /pmc/articles/PMC6247744/?report=abstract. https://doi.org/10.12659/MSM.911175
Murakami C, Hirakawa Y, Inui H, Nakano Y, Yoshida H (2002) Effect of tea catechins on cellular lipid peroxidation and cytotoxicity in HepG2 cells. Biosci Biotechnol Biochem [Internet]. 66(7):1559–62. [cited 2020 Oct 14]. Available from: https://www.tandfonline.com/doi/abs/10.1271/bbb.66.1559. https://doi.org/10.1271/bbb.66.1559
Srividhya R, Jyothilakshmi V, Arulmathi K, Senthilkumaran V, Kalaiselvi P (2008) Attenuation of senescence-induced oxidative exacerbations in aged rat brain by (-)-epigallocatechin-3-gallate. Int J Dev Neurosci. 26(2):217–223. https://doi.org/10.1016/j.ijdevneu.2007.12.003
Han J, Wang M, Jing X, Shi H, Ren M, Lou H (2014) (-)-Epigallocatechin gallate protects against cerebral ischemia-induced oxidative stress via Nrf2/ARE signaling. Neurochem Res [Internet]. 39(7):1292–9. [cited 2020 Oct 14]. Available from: https://pubmed.ncbi.nlm.nih.gov/24792731/. https://doi.org/10.1007/s11064-014-1311-5
Han XD, Zhang YY, Wang KL, Huang YP, Yang ZB, Liu Z (2017) The involvement of Nrf2 in the protective effects of (-)-Epigallocatechin-3-gallate (EGCG) on NaAsO2-induced hepatotoxicity. Oncotarget [Internet]. 8(39):65302–12. [cited 2020 Oct 14]. Available from: /pmc/articles/PMC5630332/?report=abstract. https://doi.org/10.18632/oncotarget.18582
Wang Y, Liu N, Bian X, Sun G, Du F, Wang B, et al. (2015) Epigallocatechin-3-gallate reduces tubular cell apoptosis in mice with ureteral obstruction. J Surg Res [Internet]. 197(1):145–54. [cited 2020 Oct 14]. Available from: https://pubmed.ncbi.nlm.nih.gov/25913488/. https://doi.org/10.1016/j.jss.2015.03.034
Granja A, Frias I, Neves AR, Pinheiro M, Reis S (2017) Therapeutic potential of epigallocatechin gallate nanodelivery systems, vol 2017. BioMed Research International, Hindawi Limited. https://doi.org/10.1155/2017/5813793
Yimer EM, Tuem KB, Karim A, Ur-Rehman N, Anwar F (2019) Nigella sativa L. (Black Cumin): a promising natural remedy for wide range of illnesses [Internet]. Evid. Based Complement. Alternat. Med.. Hindawi Limited. 2019. [cited 2020 Oct 18]. Available from: /pmc/articles/PMC6535880/?report=abstract. https://doi.org/10.1155/2019/1528635
Ismail N, Ismail M, Azmi NH, Abu Bakar MF, Basri H, Abdullah MA (2016) Modulation of hydrogen peroxide-induced oxidative stress in human neuronal cells by thymoquinone-rich fraction and thymoquinone via transcriptomic regulation of antioxidant and apoptotic signaling genes. Oxid Med Cell Longev [Internet]. 2016. [cited 2020 Oct 18]. Available from: /pmc/articles/PMC4707358/?report=abstract. https://doi.org/10.1155/2016/2528935
Atta MS, Almadaly EA, El-Far AH, Saleh RM, Assar DH, Al Jaouni SK, et al. (2017) Thymoquinone defeats diabetes-induced testicular damage in rats targeting antioxidant, inflammatory and aromatase expression. Int J Mol Sci [Internet]. 18(5):919. [cited 2020 Oct 18]. Available from: /pmc/articles/PMC5454832/?report=abstract. https://doi.org/10.3390/ijms18050919
Mabrouk A, Cheikh H Ben (2016) Thymoquinone ameliorates lead-induced suppression of the antioxidant system in rat kidneys. Libyan J Med [Internet]. 11(1):31018. [cited 2020 Oct 18]. Available from: /pmc/articles/PMC4823626/?report=abstract. https://doi.org/10.3402/ljm.v11.31018
Ali BH, Al Za’abi M, Shalaby A, Manoj P, Waly MI, Yasin J, et al. (2015) The effect of thymoquinone treatment on the combined renal and pulmonary toxicity of cisplatin and diesel exhaust particles. Exp Biol Med [Internet]. 240(12):1698–707. [cited 2020 Oct 18]. Available from: /pmc/articles/PMC4935344/?report=abstract. https://doi.org/10.1177/1535370215579013
Tas U, Ayan M, Sogut E, Kuloglu T, Uysal M, Tanriverdi H, et al. (2015) Protective effects of thymoquinone and melatonin on intestinal ischemia-reperfusion injury. Saudi J Gastroenterol [Internet]. 21(5):284–9. [cited 2020 Oct 18]. Available from: /pmc/articles/PMC4632252/?report=abstract. https://doi.org/10.4103/1319-3767.166203
de la Iglesia R, Milagro FI, Campión J, Boqué N, Martínez JA 2010 Healthy properties of proanthocyanidins. BioFactors [Internet]. 36(3):159–68. [cited 2020 Oct 21]. Available from: http://doi.wiley.com/10.1002/biof.79. https://doi.org/10.1002/biof.79
Manach C, Williamson G, Morand C, Scalbert A, Rémésy C (2005) Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. The American journal of clinical nutrition 81:230S–242S. https://doi.org/10.1093/ajcn/81.1.230s
Rodríguez-Pérez C, García-Villanova B, Guerra-Hernández E, Verardo V (2019) Grape seeds proanthocyanidins: an overview of in vivo bioactivity in animal models [Internet]. Nutrients. MDPI AG. 11:2435. [cited 2020 Oct 21]. Available from: /pmc/articles/PMC6835351/?report=abstract, https://doi.org/10.3390/nu11102435
Neilson AP, O’Keefe SF, Bolling BW (2016) High-molecular-weight proanthocyanidins in foods: overcoming analytical challenges in pursuit of novel dietary bioactive components. Annual Review of Food Science and Technology. Annual Reviews Inc. 7:43–64. https://doi.org/10.1146/annurev-food-022814-015604
Shi J, Yu J, Pohorly JE, Kakuda Y (2003) Polyphenolics in grape seeds — biochemistry and functionality. Journal of Medicinal Food. Mary Ann Liebert Inc. 6:291–299. https://doi.org/10.1089/109662003772519831
Bagchi D, Bagchi M, Stohs SJ, Das DK, Ray SD, Kuszynski CA et al (2000) Free radicals and grape seed proanthocyanidin extract: importance in human health and disease prevention. In: Toxicology, pp 187–197. https://doi.org/10.1016/S0300-483X(00)00210-9
Bian JT, Bhargava HN (1998) Protective effects of grape seed proanthocyanidins and selected antioxidants against TPA-induced hepatic and brain lipid peroxidation and DNA fragmentation, and peritoneal macrophage activation in mice. Gen Pharmacol. 30(5):771–776. https://doi.org/10.1016/S0306-3623(97)00332-7
Jia Z, Song Z, Zhao Y, Wang X, Liu P (2011) Grape seed proanthocyanidin extract protects human lens epithelial cells from oxidative stress via reducing NF-κB and MAPK protein expression. Mol Vis [Internet]. 17:210–7. [cited 2020 Oct 21]. Available from: http://www.molvis.org/molvis/v17/a25
Xia EQ, Deng GF, Guo YJ, Li H Bin 2010. Biological activities of polyphenols from grapes [Internet]. Int J Mol Sci. Multidisciplinary Digital Publishing Institute (MDPI). 11:622–46. [cited 2020 Oct 21]. Available from: /pmc/articles/PMC2852857/?report=abstract. https://doi.org/10.3390/ijms11020622
Mahady GB, Gyllenhaal C, Fong HHS, Farnsworth NR (2000) Ginsengs: a review of safety and efficacy. Nutr Clin Care [Internet]. 3(2):90–101. [cited 2021 Mar 29]. Available from: https://onlinelibrary.wiley.com/doi/abs/10.1046/j.1523-5408.2000.00020.x. https://doi.org/10.1046/j.1523-5408.2000.00020.x
Vuksan V, Sung MK, Sievenpiper JL, Stavro PM, Jenkins AL, Di Buono M et al (2008) Korean red ginseng (Panax ginseng) improves glucose and insulin regulation in well-controlled, type 2 diabetes: results of a randomized, double-blind, placebo-controlled study of efficacy and safety. Nutr Metab Cardiovasc Dis. 18(1):46–56. https://doi.org/10.1016/j.numecd.2006.04.003
Yennurajalingam S, Tannir NM, Williams JL, Lu Z, Hess KR, Frisbee-Hume S et al (2017) A double-blind, randomized, placebo-controlled trial of panax ginseng for cancer-related fatigue in patients with advanced cancer. JNCCN J Natl Compr Cancer Netw. 15(9):1111–1120. https://doi.org/10.6004/jnccn.2017.0149
Egert S, Bosy-Westphal A, Seiberl J, Kürbitz C, Settler U, Plachta-Danielzik S, et al. (2009) Quercetin reduces systolic blood pressure and plasma oxidised low-density lipoprotein concentrations in overweight subjects with a high-cardiovascular disease risk phenotype: a double-blinded, placebo-controlled cross-over study. Br J Nutr [Internet]. 102(7):1065–74. [cited 2021 Apr 1]. Available from: https://doi.org/10.1017/S0007114509359127
Lu NT, Crespi CM, Liu NM, Vu JQ, Ahmadieh Y, Wu S et al (2016) A phase i dose escalation study demonstrates quercetin safety and explores potential for bioflavonoid antivirals in patients with chronic hepatitis C. Phyther Res. 30(1):160–168. https://doi.org/10.1002/ptr.5518
Pakfetrat M, Basiri F, Malekmakan L, Roozbeh J (2014) Effects of turmeric on uremic pruritus in end stage renal disease patients: a double-blind randomized clinical trial. J Nephrol. 27(2):203–207. https://doi.org/10.1007/s40620-014-0039-2
Epelbaum R, Schaffer M, Vizel B, Badmaev V, Bar-Sela G (2010) Curcumin and gemcitabine in patients with advanced pancreatic cancer. Nutr Cancer [Internet]. 62(8):1137–41. [cited 2021 Mar 30]. Available from: https://pubmed.ncbi.nlm.nih.gov/21058202/. https://doi.org/10.1080/01635581.2010.513802
Vázquez Cisneros LC, López-Uriarte P, López-Espinoza A, Navarro Meza M, Espinoza-Gallardo AC, Guzmán Aburto MB (2017) Effects of green tea and its epigallocatechin (EGCG) content on body weight and fat mass in humans: a systematic review. Nutr Hosp [Internet]. 34(3):731–7. [cited 2021 Mar 30]. Available from: https://pubmed.ncbi.nlm.nih.gov/28627214/. https://doi.org/10.20960/nh.753
Hu J, Webster D, Cao J, Shao A (2018) The safety of green tea and green tea extract consumption in adults – results of a systematic review. Regulatory Toxicology and Pharmacology. Academic Press Inc. 95:412–433. https://doi.org/10.1016/j.yrtph.2018.03.019
Koshak A, Wei L, Koshak E, Wali S, Alamoudi O, Demerdash A et al (2017) Nigella sativa supplementation improves asthma control and biomarkers: a randomized, double-blind, placebo-controlled trial. Phyther Res. 31(3):403–409. https://doi.org/10.1002/ptr.5761
Moeen-ud-din H, Murad S, Fatima A (2014) Placebo controlled study on comparison of effects of Nigella sativa and nicotinic acid along with low fat diet and physical exercise on LDL-cholesterol and HDL-cholesterol. Pakistan J Med Heal Sci. 8(2):306–309
Handog EB, Galang DAVF, De Leon-Godinez MA, Chan GP (2009) A randomized, double-blind, placebo-controlled trial of oral procyanidin with vitamins A, C, E for melasma among Filipino women. Int J Dermatol [Internet]. 48(8):896–901. [cited 2021 Mar 31]. Available from: https://pubmed.ncbi.nlm.nih.gov/19659873/. https://doi.org/10.1111/j.1365-4632.2009.04130.x
Sano A (2017) Safety assessment of 4-week oral intake of proanthocyanidin-rich grape seed extract in healthy subjects. Food Chem Toxicol [Internet]. 108(Pt B):519–23. [cited 2021 Mar 31]. Available from: https://pubmed.ncbi.nlm.nih.gov/27889390/. https://doi.org/10.1016/j.fct.2016.11.021
Sonam KS, Guleria S (2017) Synergistic antioxidant activity of natural products. Ann Pharmacol Pharm [Internet]. 2(16):1–6. [cited 2020 Sep 25]. Available from: http://www.remedypublications.com/pharmacology-and-pharmaceutics/articles/pdfs_folder/app-v2-id1086.pdf
Code Availability
Not applicable.
Author information
Authors and Affiliations
Contributions
Conceptualization, L.M.A, D.S; writing—original draft preparation, L.M.A; writing—review and editing, L.M.A, Y.J, J.P, and D.S. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Angwa, L.M., Jiang, Y., Pei, J. et al. Antioxidant Phytochemicals for the Prevention of Fluoride-Induced Oxidative Stress and Apoptosis: a Review. Biol Trace Elem Res 200, 1418–1441 (2022). https://doi.org/10.1007/s12011-021-02729-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02729-8