Abstract
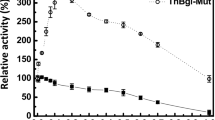
Coptotermes formosanus is a well-known wood-feeding termite that can degrade lignocellulose polysaccharides efficiently with its unique multi-enzyme catalysis system. β-glucosidase (BG) is one of the important cellulases in its enzyme system. However, there may present multiple endogenous BGs in termite digestive system for various properties and functions. This study aims to characterize two BG homologs and reveal their potential coordinative effect. In this study, two endogenous BG homologs (CfGlu1B and CfGlu1C) from C. formosanus showed different substrate specificity. CfGlu1B favors cellobiose while CfGlu1C favors sucrose. Besides, CfGlu1C exhibited higher alkali resistance than CfGlu1B. Kinetic analysis revealed that CfGlu1B enzyme’s activity toward p-NP-β-D-glucopyranoside (p-NPG) was higher than that of CfGlu1C, and the difference mainly attributes to the turnover number (K cat). In addition, the activity assay showed significant synergistic action of CfGlu1B and CfGlu1C in degrading D-lactosum. For effect of metals, Cu2+ inhibited both enzymes and Ca2+ increased the activity of CfGlu1C but not CfGlu1B. Site-directed mutagenesis analysis indicated that both enzymes lost activities when residues E190 of CfGlu1B and E168 of CfGlu1C were mutated to alanine, respectively, which were essential active centers of the GHF1 enzymes. Moreover, mutation H252N increased the activity of enzyme CfGlu1C by 2.1-fold. This study implies interesting possibilities for better practical biotechnological use in green energy production.





Similar content being viewed by others
References
Watanabe, H., & Tokuda, G. (2010). Cellulolytic systems in insects. Annual Review of Entomology, 55, 609–632.
Ohkuma, M. (2003). Termite symbiotic systems: efficient bio-recycling of lignocellulose. Applied Microbiology and Biotechnology, 61(1), 1–9.
Zhang, D., Lax, A. R., Henrissat, B., Coutinho, P., Katiya, N., Nierman, W. C., & Fedorova, N. (2012). Carbohydrate-active enzymes revealed in Coptotermes formosanus (Isoptera: Rhinotermitidae) transcriptome. Insect Molecular Biology, 21(2), 235–245.
Breznak, J. A., & Brune, A. (1994). Role of microorganisms in the digestion of lignocellulose by termites. Annual Review of Entomology, 39(1), 453–487.
Scharf, M. E., Kovaleva, E. S., Jadhao, S., Campbell, J. H., Buchman, G. W., & Boucias, D. G. (2010). Functional and translational analyses of a beta-glucosidase gene (glycosyl hydrolase family 1) isolated from the gut of the lower termite Reticulitermes flavipes. Insect Biochemistry and Molecular Biology, 40(8), 611–620.
Elshafei, A. M., Hassan, M. M., Haroun, B. M., Abdel Fatah, O. M., Atta, H. M., & Othman, A. M. (2009). Purification and properties of an endoglucanase of Aspergillus terreus DSM 826. Journal of Basic Microbiology, 49(5), 426–432.
Ketudat Cairns, J. R., & Esen, A. (2010). β-Glucosidases. Cellular and Molecular Life Sciences, 67(20), 3389–3405.
Inoue, T., Murashima, K., Azuma, J. I., Sugimoto, A., & Slaytor, M. (1997). Cellulose and xylan utilisation in the lower termite Reticulitermes speratus. Journal of Insect Physiology, 43(3), 235–242.
Henrissat, B., & Bairoch, A. (1993). New families in the classification of glycosyl hydrolases based on amino acid sequence similarities. Biochemical Journal, 293, 781–788.
Henrissat, B., Callebaut, I., Fabrega, S., Lehn, P., Mornon, J.-P., & Davies, G. (1995). Conserved catalytic machinery and the prediction of a common fold for several families of glycosyl hydrolases. Proceedings of the National Academy of Sciences, 92(15), 7090–7094.
Lo, N., Tokuda, G., & Watanabe, H. (2011). Biology of termites: a modern synthesis. In D. E. Bignell, Y. Roisin, & N. Lo (Eds.), Evolution and function of endogenous termite cellulases (pp. 51–67). Netherlands: Springer.
Shimada, K., & Maekawa, K. (2014). Gene expression and molecular phylogenetic analyses of beta-glucosidase in the termite Reticulitermes speratus (Isoptera: Rhinotermitidae). Journal of Insect Physiology, 65, 63–69.
Zhang, D., Lax, A. R., Bland, J. M., Yu, J., Fedorova, N., & Nierman, W. C. (2010). Hydrolysis of filter-paper cellulose to glucose by two recombinant endogenous glycosyl hydrolases of Coptotermes formosanus. Insect Science, 17(3), 245–252.
Watanabe, H., Noda, H., Tokuda, G., & Lo, N. (1998). A cellulase gene of termite origin. Nature, 394(6691), 330–331.
Zhang, D., Allen, A. B., & Lax, A. R. (2012). Functional analyses of the digestive β-glucosidase of Formosan subterranean termites (Coptotermes formosanus). Journal of Insect Physiology, 58(1), 205–210.
Davies, G., & Henrissat, B. (1995). Structures and mechanisms of glycosyl hydrolases. Structure, 3(9), 853–859.
Sun, J. Z., Ding, S. H., & Doran-Peterson, J. (2014). Biological conversion of biomass for fuels and chemicals: exploration from natural utilization systems. In J. Z. Sun & J. X. G. Zhou (Eds.), Lignocellulolytic systems of insects and their potential for viable biofuels (pp. 195–217). Cambridge: RSC Publishing.
Franco Cairo, J. P., Oliveira, L. C., Uchima, C. A., Alvarez, T. M., Citadini, A. P., Cota, J., Leonardo, F. C., Costa-Leonardo, A. M., Carazzolle, M. F., Costa, F. F., Pereira, G. A., & Squina, F. M. (2013). Deciphering the synergism of endogenous glycoside hydrolase families 1 and 9 from Coptotermes gestroi. Insect Biochemistry and Molecular Biology, 43(10), 970–981.
Uchima, C. A., Tokuda, G., Watanabe, H., Kitamoto, K., & Arioka, M. (2012). Heterologous expression in Pichia pastoris and characterization of an endogenous thermostable and high-glucose-tolerant-glucosidase from the termite Nasutitermes takasagoensis. Applied and Environmental Microbiology, 78(12), 4288–4293.
Liu, W., Hong, J., Bevan, D. R., & Zhang, Y. H. P. (2009). Fast identification of thermostable beta-glucosidase mutants on cellobiose by a novel combinatorial selection/screening approach. Biotechnology and Bioengineering, 103(6), 1087–1094.
Jeng, W. Y., Wang, N. C., Lin, M. H., Lin, C. T., Liaw, Y. C., Chang, W. J., Liu, C. I., Liang, P. H., & Wang, A. H. (2011). Structural and functional analysis of three β-glucosidases from bacterium Clostridium cellulovorans, fungus Trichoderma reesei and termite Neotermes koshunensis. Journal of Structural Biology, 173(1), 46–56.
Jeng, W., Wang, N., Lin, C., Chang, W., Liu, C.-I., & Wang, A. H.-J. (2012). High-resolution structures of Neotermes koshunensis β-glucosidase mutants provide insights into the catalytic mechanism and the synthesis of glucoconjugates. Acta Crystallographica, D68, 829–838.
Lee, H. L., Chang, C. K., Jeng, W. Y., Wang, A. H., & Liang, P. H. (2012). Mutations in the substrate entrance region of beta-glucosidase from Trichoderma reesei improve enzyme activity and thermostability. Protein Engineering Design and Selection, 25(11), 733–740.
Franco Cairo, J. P., Leonardo, F. C., Alvarez, T. M., Ribeiro, D. A., Buchli, F., Costa-Leonardo, A. M., Carazzolle, M. F., Costa, F. F., Paes Leme, A. F., Pereira, G. A., & Squina, F. M. (2011). Functional characterization and target discovery of glycoside hydrolases from the digestome of the lower termite Coptotermes gestroi. Biotechnology for Biofuels, 4, 50.
Tokuda, G., Saito, H., & Watanabe, H. (2002). A digestive β-glucosidase from the salivary glands of the termite, Neotermes koshunensis (Shiraki): distribution, characterization and isolation of its precursor cDNA by 5'- and 3'-RACE amplifications with degenerate primers. Insect Biochemistry and Molecular Biology, 32(12), 1681–1689.
Ni, J., Tokuda, G., Takehara, M., & Watanabe, H. (2007). Heterologous expression and enzymatic characterization of β-glucosidase from the drywood-eating termite, Neotermes koshunensis. APPL ENTOMOL ZOOL., 42(3), 457–463.
Marana, S. R., Jacobs-Lorena, M., Terra, W. R., & Ferreira, C. (2001). Amino acid residues involved in substrate binding and catalysis in an insect digestive beta-glycosidase. Biochimica et Biophysica Acta, 1545(1–2), 41–52.
Swalley, S. E., Fulghum, J. R., & Chambers, S. P. (2006). Screening factors effecting a response in soluble protein expression: formalized approach using design of experiments. Analytical Biochemistry, 351(1), 122–127.
Uchima, C. A., Tokuda, G., Watanabe, H., Kitamoto, K., & Arioka, M. (2011). Heterologous expression and characterization of a glucose-stimulated β-glucosidase from the termite Neotermes koshunensis in Aspergillus oryzae. Applied Microbiology and Biotechnology, 89(6), 1761–1771.
Sun, J.-Z., & Scharf, M. (2010). Exploring and integrating cellulolytic systems of insects to advance biofuel technology. Insect Science., 17(3), 163–165.
Sun, W., Sammynaiken, R., Chen, L., Maley, J., Schatte, G., Zhou, Y., & Yang, J. (2011). Sphingobium chlorophenolicum dichlorohydroquinone dioxygenase (PcpA) is alkaline resistant and thermally stable. International Journal of Biological Sciences, 7(8), 1171–1179.
Uchima, C. A., Tokuda, G., Watanabe, H., Kitamoto, K., & Arioka, M. (2013). A novel glucose-tolerant β-glucosidase from the salivary gland of the termite Nasutitermes takasagoensis. Journal of General and Applied Microbiology, 59(2), 141–145.
Scharf, M. E., Karl, Z. J., Sethi, A., & Boucias, D. G. (2011). Multiple levels of synergistic collaboration in termite lignocellulose digestion. PloS One, 6(7), e21709.
Asha, B. M. (2012). Purification and characterization of a thermophilic cellulase from a novel cellulolytic strain, Paenibacillus barcinonensis. Journal of Microbiology and Biotechnology, 22(11), 1501–1509.
Pei, X. Q., Yi, Z. L., Tang, C. G., & Wu, Z. L. (2011). Three amino acid changes contribute markedly to the thermostability of β-glucosidase BglC from Thermobifida fusca. Bioresource Technology, 102(3), 3337–3342.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation (No. 31271272, 31071030), the Scientific Research Foundation for the Returned Overseas Chinese Scholars, the Jiangsu talent in six major fields-biotechnology and new medicine project, the Jiangsu Natural Science Foundation (No. BK20130506), and the Jiangsu University startup fund (10JDG021).
We are also thankful to the anonymous reviewers for their constructive comments.
Compliance with Ethical Standards
This research has no human participants and/or animals.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Feng, T., Liu, H., Xu, Q. et al. Identification and Characterization of Two Endogenous β-Glucosidases from the Termite Coptotermes formosanus . Appl Biochem Biotechnol 176, 2039–2052 (2015). https://doi.org/10.1007/s12010-015-1699-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1699-7