Abstract
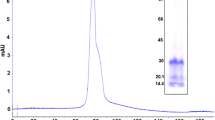
Biophysical and carbohydrate binding studies have been carried out on a lectin of Spatholobus parviflorus (SPL) seeds isolated by affinity chromatography on cross-linked guar gum. It agglutinated erythrocytes of all ABO blood groups. SDS-PAGE, both in reducing and non-reducing conditions, showed two bands with MW of 29 and 31 kDa. MALDI TOF analysis revealed two peaks at 60 and 120 kDa, indicating that SPL is a hetero-dimeric tetramer. Temperature and pH stability studies revealed that SPL is a thermostable protein and its lectin activity is unaffected in the temperature range of 0–70 °C. Its activity was maximal in the pH range of 7–8. Unfolding studies with different denaturants like urea and guanidine hydrochloride indicated its globular nature and the presence of tryptophan in the highly hydrophobic area, which could be correlated to the results of fluorescence spectroscopic studies. The effect of carbohydrate binding on the lectin, shown by circular dichroism spectra, indicated the changes in its secondary and tertiary structures. SPL exerted anti-fungal activity against Aspergillus sp.










Similar content being viewed by others
References
Liener, I. E., Sharon, N., & Goldstein, I. J. (1986). The lectins: properties, functions, and applications in biology and medicine. Orlando: Academic.
Loris, R., Hamelryck, T., Bouckaert, J., & Wyns, L. (1998). Biochimica et Biophysica Acta, 1383, 9–36.
Vijayan, M., & Chandra, N. (1999). Current Opinion in Structural Biology, 9, 707–714.
Nishi, N., Shoji, H., Seki, M., Itoh, A., Miyanaka, H., Yuube, K., Hirashima, M., & Nakamura, T. (2003). Glycobiology, 13, 755–763.
Sharon, N., & Lis, H. (2004). Glycobiology, 14, 53–62R.
Bies, C., Lehr, C.-M., & Woodley, J. F. (2004). Advanced Drug Delivery Reviews, 56, 425–435.
Jepson, M. A., Clark, M. A., & Hirst, B. H. (2004). Advanced Drug Delivery Reviews, 56, 511–525.
Chandra, N. R., Kumar, N., Jeyakani, J., Singh, D. D., Gowda, S. B., & Prathima, M. N. (2006). Glycobiology, 16, 938–946.
Rini, J. M. (1995). Annual Review of Biophysics and Biomolecular Structure, 24, 551–577.
Weis, W. I., & Drickamer, K. (1996). Annual Review of Biochemistry, 65, 441–473.
Lis, H., & Sharon, N. (1998). Chemical Reviews, 98, 637–674.
Rüdiger, H., & Gabius, H. J. (2001). Glycoconjugate Journal, 18, 589–613.
Van Damme, E. J., Roy, S., Barre, A., Citores, L., Mostafapous, K., Rougé, P., Van Leuven, F., Girbés, T., Goldstein, I. J., & Peumans, W. J. (1997). European Journal of Biochemistry, 245, 648–655.
Abdullaev, F. I., & Gonzalez de Mejia, E. (1997). Natural Toxins, 5, 157–163.
Rubinstein, N., Ilarregui, J. M., Toscano, M. A., & Rabinovich, G. A. (2004). Tissue Antigens, 64, 1–12.
Herre, J., Willment, J. A., Gordon, S., & Brown, G. D. (2004). Critical Reviews in Immunology, 24, 193–203.
Macedo, M. L. R., Damico, D. C. S., Freire, M., Das, G. M., Toyama, M. H., Marangoni, S., & Novello, J. C. (2003). J. Agric. Food Chemistry, 51, 2980–2986.
Etzler, M. E. (1985). Annual Review of Plant Physiology, 36, 209–234.
Sharon, N., & Lis, H. (2004). Glycobiology, 14, 53–62.
Lis, H., & Sharon, N. (1984). Biology of Carbohydrates. (John Wiley and Sons). New York, 2, 1–85.
Loganathan, D., Osborne, S. E., Glick, G. D., & Goldstein, I. J. (1992). Archives of Biochemistry and Biophysics, 299, 268–274.
Ourth, D. D., Narra, M. B., & Chung, K. T. (2005). Biochemical and Biophysical Research Communications, 335, 1085–1089.
Fenton-Navarro, B., García-Hernández, E., Heimer, E., Aguilar, M. B., Rodríguez-A, C., Arreguín-Espinosa, R., & Falcón, A. (2003). Toxicon, 42, 525–532.
Fullmer, J. M., Riedl, M. S., Higgins, L., & Elde, R. (2004). NeuroReport, 15, 1705–1709.
Gerlach, D., Schlott, B., Zähringer, U., & Schmidt, K.-H. (2005). FEMS Immunol. Med. Microbiol., 43, 223–232.
Iwama, M., Ogawa, Y., Sasaki, N., Nitta, K., Takayanagi, Y., Ohgi, K., Tsuji, T., & Irie, M. (2001). Biological and Pharmaceutical Bulletin, 24, 978–981.
De Mejía, E. G., & Prisecaru, V. I. (2005). Critical Reviews in Food Science and Nutrition, 45, 425–445.
Singh, T., Wu, J. H., Peumans, W. J., Rougé, P., Van Damme, E. J. M., Alvarez, R. A., Blixt, O., & Wu, A. M. (2006). The Biochemical Journal, 393, 331–341.
Ciopraga, J., Gozia, O., Tudor, R., Brezuica, L., & Doyle, R. J. (1999). Biochimica et Biophysica Acta, 1428, 424–432.
Ye, X. Y., Ng, T. B., Tsang, P. W., & Wang, J. (2001). Journal of Protein Chemistry, 20, 367–375.
Hatakeyama, T., Suenaga, T., Eto, S., Niidome, T., & Aoyagi, H. (2004). Journal of Biochemistry, 135, 65–70.
Balzarini, J., Neyts, J., Schols, D., Hosoya, M., Van Damme, E., Peumans, W., & De Clercq, E. (1992). Antiviral Research, 18, 191–207.
Wong, J. H., & Ng, T. B. (2005). Archives of Biochemistry and Biophysics, 439, 91–98.
Dalla Pellegrina, C., Rizzi, C., Mosconi, S., Zoccatelli, G., Peruffo, A., & Chignola, R. (2005). Toxicology and Applied Pharmacology, 207, 170–178.
Komath, S. S., & Swamy, M. J. (1998). Current Science, 75, 608–611.
Geethanandan, K., Abhilash, J., Bharath, S. R., Sadasivan, C., & Haridas, M. (2011). Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun., 67, 700–702.
Geethanandan, K., Abhilash, J., Bharath, S. R., Sadasivan, C., & Haridas, M. (2011). International Journal of Biological Macromolecules, 49, 992–998.
Appukuttan, P. S., Surolia, A., & Bachawat, B. K. (1977). Indian J. Biochem. Biophys., 14, 382–384.
Silva, J. A., Damico, D. C. S., Baldasso, P. A., Mattioli, M. A., Winck, F. V., Fraceto, L. F., Novello, J. C., & Marangoni, S. (2007). The Protein Journal, 26, 193–201.
Laemmli, U. K. (1970). Nature, 227, 680–685.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). The Journal of Biological Chemistry, 193, 265–275.
Morris, D. L. (1948). Science, 107, 254–255.
Gozia, O., Ciopraga, J., Bentia, T., Lungu, M., Zamfirescu, I., Tudor, R., Roseanu, A., & Nitu, F. (1993). C. R. Acad. Sci. III. Sci. Vie., 316, 788–792.
Ngai, P. H. K., & Ng, T. B. (2007). Applied Microbiology and Biotechnology, 74, 366–371.
Sultan, N. A. M., Rao, R. N., Nadimpalli, S. K., & Swamy, M. J. (2006). Biochimica et Biophysica Acta, 1760, 1001–1008.
Wongkham, S., Wongkham, C., Trisonthi, C., Boonsiri, P., Simasathiansophon, S., & Atisook, K. (1994). Plant Science, 103, 121–126.
Sinha, S., Mitra, N., Kumar, G., Bajaj, K., & Surolia, A. (2005). Biophysical Journal, 88, 1300–1310.
Ye, X. Y., Ng, T. B., Tsang, P. W., & Wang, J. (2001). Journal of Protein Chemistry, 20, 367–375.
Acknowledgments
The authors would like to acknowledge SARD, KSCSTE for granting the chromatographic facility and Molecular Biophysics Unit, IISc, Bangalore for fluorescence, CD, X-ray diffraction, and MALDI TOF data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
K., G., Joseph, A., C., S. et al. Carbohydrate Binding and Unfolding of Spatholobus parviflorus Lectin: Fluorescence and Circular Dichroism Spectroscopic Study. Appl Biochem Biotechnol 171, 80–92 (2013). https://doi.org/10.1007/s12010-013-0340-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0340-x