Abstract
Purpose of Review
New therapies are needed to potentiate the effects of current immunotherapies and overcome resistance. The stimulator of interferon genes genes (STING) pathway is an innate immune activating cascade that may enhance current cancer immunotherapies.
Recent Findings
Preclinical data has shown that the addition of a STING agonist enhances the effect of current treatments such as immune checkpoint inhibitor antibodies and radiation therapy. Early phase trials have demonstrated modest efficacy of STING agonists and revealed new mechanistic and technical challenges.
Summary
STING agonists are a new class of agents that activate the immune response to improve tumor control. A wide range of preclinical experiments, translational data, and ongoing clinical trials support the therapeutic use of STING agonists in patients. Trials to determine optimal drug combinations and novel delivery mechanisms are continuing in development.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Syn NL, Teng MWL, Mok TSK, Soo RA. De-novo and acquired resistance to immune checkpoint targeting. Lancet Oncol. 2017;18(12):e731-e41. https://doi.org/10.1016/S1470-2045(17)30607-1.
Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell. 2017; 168(4):707–23. https://doi.org/10.1016/j.cell.2017.01.017.
Bashash D, Zandi Z, Kashani B, Pourbagheri-Sigaroodi A, Salari S, Ghaffari SH. Resistance to immunotherapy in human malignancies: mechanisms, research progresses, challenges, and opportunities. J Cell Physiol. 2022;237(1):346–72. https://doi.org/10.1002/jcp.30575.
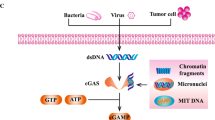
Hopfner K-P, Hornung V. Molecular mechanisms and cellular functions of cGAS–STING signalling. Nat Rev Mol Cell Biol. 2020;21(9):501–21. https://doi.org/10.1038/s41580-020-0244-x.
Sun L, Wu J, Du F, Chen X, Chen ZJ. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science. 2013;339(6121):786–91. https://doi.org/10.1126/science.1232458.
Zhang X, Bai X-c, Chen ZJ. Structures and mechanisms in the cGAS-STING innate immunity pathway. Immunity. 2020;53(1). https://doi.org/10.1016/j.immuni.2020.05.013.
•• Woo SR, Fuertes MB, Corrales L, Spranger S, Furdyna MJ, Leung MY, et al. STING-dependent cytosolic DNA sensing mediates innate immune recognition of immunogenic tumors. Immunity. 2014;41(5):830–42. https://doi.org/10.1016/j.immuni.2014.10.017. This manuscript describes preclinical data supporting STING as an important mechanism of anti-tumor immune reponse.
Diamond MS, Kinder M, Matsushita H, Mashayekhi M, Dunn GP, Archambault JM, et al. Type I interferon is selectively required by dendritic cells for immune rejection of tumors. J Exp Med. 2011;208(10):1989–2003. https://doi.org/10.1084/jem.20101158
Marcus A, Mao AJ, Lensink-Vasan M, Wang L, Vance RE, Raulet DH. Tumor-derived cGAMP triggers a STING-mediated interferon response in non-tumor cells to activate the NK cell response. Immunity. 2018;49(4):754–63. https://doi.org/10.1016/j.immuni.2018.09.016.
•• Corrales L, Glickman LH, McWhirter SM, Kanne DB, Sivick KE, Katibah GE, et al. Direct activation of STING in the tumor microenvironment leads to potent and systemic tumor regression and immunity. Cell Rep. 2015;11(7):1018–30. https://doi.org/10.1016/j.celrep.2015.04.031. This preclinical manuscript provides evidence that activation of STING in the tumor microenviroment can be employed therapeutically to eradicate cancers.
Ohkuri T, Kosaka A, Ishibashi K, Kumai T, Hirata Y, Ohara K, et al. Intratumoral administration of cGAMP transiently accumulates potent macrophages for anti-tumor immunity at a mouse tumor site. Cancer Immunol Immunother. 2017;66(6):705–16. https://doi.org/10.1007/s00262-017-1975-1.
Demaria O, De Gassart A, Coso S, Gestermann N, Di Domizio J, Flatz L, et al. STING activation of tumor endothelial cells initiates spontaneous and therapeutic antitumor immunity. Proc Natl Acad Sci U S A. 2015; 112(50):15408–13. https://doi.org/10.1073/pnas.1512832112
Shi F, Su J, Wang J, Liu Z, Wang T. Activation of STING inhibits cervical cancer tumor growth through enhancing the anti-tumor immune response. Mol Cell Biochem. 2020;476(2):1015–24. https://doi.org/10.1007/s11010-020-03967-5.
Vonderhaar EP, Barnekow NS, McAllister D, McOlash L, Eid MA, Riese MJ, et al. STING activated tumor-intrinsic type i interferon signaling promotes CXCR3 dependent antitumor immunity in pancreatic cancer. Cell MolGastroenterol Hepatol. 2021;12(1):41–58. https://doi.org/10.1016/j.jcmgh.2021.01.018.
Deng L, Liang H, Xu M, Yang X, Burnette B, Arina A, et al. STING-dependent cytosolic DNA sensing promotes radiation-induced type I interferon-dependent antitumor immunity in immunogenic tumors. Immunity. 2014;41(5):843–52. https://doi.org/10.1016/j.immuni.2014.10.019.
Liu Y, Crowe WN, Wang L, Lu Y, Petty WJ, Habib AA, et al. An inhalable nanoparticulate STING agonist synergizes with radiotherapy to confer long-term control of lung metastases. Nat Commun. 2019;10(1):1–15. https://doi.org/10.1038/s41467-019-13094-5.
Zaidi AH, Kelly RJ, Gorbunova A, Omstead AN, Salvitti MS, Zheng P, et al. Intratumoral immunotherapy with STING agonist, ADU-S100, induces CD8+ Tcell mediated anti-tumor immunity in an esophageal adenocarcinoma model. Oncotarget. 2021;12(4):292–303. https://doi.org/10.18632/oncotarget.27886.
• Xue A, Shang Y, Jiao P, Zhang S, Zhu C, He X, et al. Increased activation of cGAS-STING pathway enhances radiosensitivity of non-small cell lung cancer cells. Thorac Cancer. 2022;13(9):1361–8. https://doi.org/10.1111/1759-7714.14400. This manuscript provides preclinical data supporting the combination of radiotherapy and STING activation as a synergistic immunotherapy combination.
Hu M, Zhou M, Bao X, Pan D, Jiao M, Liu X, et al. ATM inhibition enhances cancer immunotherapy by promoting mtDNA leakage and cGAS/STING activation. J Clin Invest. 2021;131(3). https://doi.org/10.1172/JCI139333.
• Nakamura T, Sato T, Endo R, Sasaki S, Takahashi N, Sato Y, et al. STING agonist loaded lipid nanoparticles overcome anti-PD-1 resistance in melanoma lung metastasis via NK cell activation. J Immunother Cancer. 2021;9(7). https://doi.org/10.1136/jitc-2021-002852. This manuscript describes the efficacy in prelinical melanoma models for a STING agonist loaded in a lipid nanoparticle.
Wang-Bishop L, Wehbe M, Shae D, James J, Hacker BC, Garland K, et al. Potent STING activation stimulates immunogenic cell death to enhance antitumor immunity in neuroblastoma. J Immunother Cancer. 2020;8(1). https://doi.org/10.1136/jitc-2019-000282.
Lee SJ, Yang H, Kim WR, Lee YS, Lee WS, Kong SJ, et al. STING activation normalizes the intraperitoneal vascular-immune microenvironment and suppresses peritoneal carcinomatosis of colon cancer. J Immunother Cancer.. 2021;9(6). https://doi.org/10.1136/jitc-2020-002195.
Yi M, Niu M, Zhang J, Li S, Zhu S, Yan Y, et al. Combine and conquer: manganese synergizing anti-TGF-β/PD-L1 bispecific antibody YM101 to overcome immunotherapy resistance in non-inflamed cancers. J Hematol Oncol. 2021;14(1):1–21. https://doi.org/10.1186/s13045-021-01155-6.
Wang Z, Celis E. STING activator c-di-GMP enhances the anti-tumor effects of peptide vaccines in melanoma-bearing mice. Cancer Immunol Immunother. 2015;64(8):1057–66. https://doi.org/10.1007/s00262-015-1713-5.
Rossi M, Carboni S, DiBerardino-Besson W, Riva E, Santiago-Raber ML, Belnoue E, et al. STING agonist combined to a protein-based cancer vaccine potentiates peripheral and intra-tumoral T cell immunity. Front Immunol. 2021;12:695056. https://doi.org/10.3389/fimmu.2021.695056.
Chandra D, Quispe-Tintaya W, Jahangir A, Asafu-Adjei D, Ramos I, Sintim HO, et al. STING ligand c-di-GMP improves cancer vaccination against metastatic breast cancer. Cancer Immunol Res. 2014;2(9):901–10. https://doi.org/10.1158/2326-6066.CIR-13-0123.
Esteves AM, Papaevangelou E, Dasgupta P, Galustian C. Combination of interleukin-15 with a STING agonist, ADU-S100 analog: a potential immunotherapy for prostate cancer. Front Oncol. 2021;11:621550. https://doi.org/10.3389/fonc.2021.621550.
Xu N, Palmer DC, Robeson AC, Shou P, Bommiasamy H, Laurie SJ, et al. STING agonist promotes CAR T cell trafficking and persistence in breast cancer. J Exp Med. 2021;218(2). https://doi.org/10.1084/jem.20200844.
Siim BG, Lee AE, Shalal-Zwain S, Pruijn FB, McKeage MJ, Wilson WR. Marked potentiation of the antitumour activity of chemotherapeutic drugs by the antivascular agent 5,6-dimethylxanthenone-4-acetic acid (DMXAA). Cancer Chemother Pharmacol. 2003;51(1):43–52. https://doi.org/10.1007/s00280-002-0529-0.
Prantner D, Perkins DJ, Lai W, Williams MS, Sharma S, Fitzgerald KA, et al. 5,6-Dimethylxanthenone-4-acetic acid (DMXAA) activates stimulator of interferon gene (STING)-dependent innate immune pathways and is regulated by mitochondrial membrane potential. J Biol Chem. 2012;287(47):39776–88. https://doi.org/10.1074/jbc.M112.382986.
McKeage MJ, Von Pawel J, Reck M, Jameson MB, Rosenthal MA, Sullivan R, et al. Randomised phase II study of ASA404 combined with carboplatin and paclitaxel in previously untreated advanced non-small cell lung cancer. Br J Cancer. 2008;99(12):2006–12. https://doi.org/10.1038/sj.bjc.6604808.
McKeage MJ, Jameson MB, Investigators ASSG. Comparative outcomes of squamous and non-squamous non-small cell lung cancer (NSCLC) patients in phase II studies of ASA404 (DMXAA) - retrospective analysis of pooled data. J Thorac Dis. 2010;2(4):199–204. https://doi.org/10.3978/j.issn.2072-1439.2010.02.04.1.
Johnson DH, Fehrenbacher L, Novotny WF, Herbst RS, Nemunaitis JJ, Jablons DM, et al. Randomized phase II trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol. 2004;22(11):2184–91. https://doi.org/10.1200/JCO.2004.11.022.
Lara PN Jr, Douillard JY, Nakagawa K, von Pawel J, McKeage MJ, Albert I, et al. Randomized phase III placebo-controlled trial of carboplatin and paclitaxel with or without the vascular disrupting agent vadimezan (ASA404) in advanced non-small-cell lung cancer. J Clin Oncol. 2011;29(22):2965–71. https://doi.org/10.1200/JCO.2011.35.0660.
A Phase III, randomized, double-blind, placebo-controlled, multi-center study of ASA404 in combination with paclitaxel and carboplatin as first-line treatment for locally advanced or met- astatic (stage IIIb/IV) non-small cell lung cancer (NSCLC). Novartis; 2010 [cited 2022]. Available from: https://www.novctrd.com/ctrdweb/trialresult/trialresults/pdf?trialResultId=4609.
A phase III, randomized, double-blind, placebo-controlled, multi-center study of Vadimezan in combination with docetaxel in second-line treatment of patients with locally advanced or metastatic (stage IIIb/IV) non-small-cell lung cancer (NSCLC) 2011 [cited 2022]. Available from: https://www.novctrd.com/ctrdweb/trialresult/trialresults/pdf?trialResultId=4927.
Pili R, Rosenthal MA, Mainwaring PN, Van Hazel G, Srinivas S, Dreicer R, et al. Phase II study on the addition of ASA404 (vadimezan; 5,6-dimethylxanthenone-4-acetic acid) to docetaxel in CRMPC. Clin Cancer Res. 2010;16(10):2906–14. https://doi.org/10.1158/1078-0432.CCR-09-3026.
Conlon J, Burdette DL, Sharma S, Bhat N, Thompson M, Jiang Z, et al. Mouse, but not human STING, binds and signals in response to the vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid. J Immunol. 2013;190(10):5216–25. https://doi.org/10.4049/jimmunol.1300097.
Yi G, Brendel VP, Shu C, Li P, Palanathan S, Cheng Kao C. Single nucleotide polymorphisms of human STING can affect innate immune response to cyclic dinucleotides. PLoS One. 2013;8(10):e77846. https://doi.org/10.1371/journal.pone.0077846.
Gao P, Ascano M, Wu Y, Barchet W, Gaffney BL, Zillinger T, et al. Cyclic [G(2’,5’)pA(3’,5’)p] is the metazoan second messenger produced by DNA-activated cyclic GMP-AMP synthase. Cell. 2013;153(5):1094–107. https://doi.org/10.1016/j.cell.2013.04.046.
Kato K, Nishimasu H, Oikawa D, Hirano S, Hirano H, Kasuya G, et al. Structural insights into cGAMP degradation by Ecto-nucleotide pyrophosphatase phosphodiesterase 1. Nat Commun. 2018;9(1):4424. https://doi.org/10.1038/s41467-018-06922-7.
•• Meric-Bernstam F, Sweis RF, Hodi FS, Messersmith WA, Andtbacka RHI, Ingham M, et al. Phase I dose-escalation trial of MIW815 (ADU-S100), an intratumoral STING agonist, in patients with advanced/metastatic solid tumors or lymphomas. Clin Cancer Res. 2022;28(4):677–88. https://doi.org/10.1158/1078-0432.CCR-21-1963. This manuscript is the first published clinical trial on therapeutic STING activation. It shows that intratumoral injection with a STING agonist can lead to tumnor regression in patients.
•• Meric-Bernstam F, Sweis RF, Kasper S, Hamid O, Bhatia S, Dummer R, et al. Combination of the STING agonist MIW815 and PD-1 inhibitor spartalizumab in advanced/metastatic solid tumors or lymphomas: an open-label, multicenter, phase Ib study. Clin Cancer Res. 2022; in press . This manuscript reports a clinical trial with an anti-PD-1 antibody combined with with intratumoral STING activation in patients.
• Harrington K, Brody J, Ingham M, Strauss J, Cemerski S, Wang M, et al. Preliminary results of the first-in-human (FIH) study of MK-1454, an agonist of stimulator of interferon genes (STING), as monotherapy or in combination with pembrolizumab (pembro) in patients with advanced solid tumors or lymphomas. Devel Ther. 2018;29:vii712. This abstract reported with an anti-PD-1 antibody combined with with STING activation can lead to tumnor regression in patients.
Merck Sharp & Dohme LLC. Annual report for the fiscal year ended December 31, 2020.
Kim DS, Endo A, Fang FG, Huang KC, Bao X, Choi HW, et al. E7766, a macrocycle-bridged stimulator of interferon genes (STING) agonist with potent pan-genotypic activity. ChemMedChem. 2021;16(11):1740–3. https://doi.org/10.1002/cmdc.202100068.
Inc. E. Study of intratumorally administered stimulator of interferon genes (STING) agonist E7766 in participants with advanced solid tumors or lymphomas - INSTAL-101 2022 [updated April 25, 2022; cited 2022 May 23]. Available from: https://clinicaltrials.gov/ct2/show/NCT04144140.
Inc. E. A Study of Stimulator of Interferon Genes (STING) Agonist E7766 in non-muscle invasive bladder cancer (NMIBC) including participants unresponsive to Bacillus Calmette-Guerin (BCG) therapy, INPUT-102 2020 [updated December 14, 2020; cited 2022 May 23]. Available from: https://clinicaltrials.gov/ct2/show/NCT04109092.
Ramanjulu JM, Pesiridis GS, Yang J, Concha N, Singhaus R, Zhang SY, et al. Design of amidobenzimidazole STING receptor agonists with systemic activity. Nature. 2018;564(7736):439–43. https://doi.org/10.1038/s41586-018-0705-y.
Luke JJ, Janku F, Strauss J, Olszanski AJ, Leach K, Radhakrishnan I, et al. 598TiP A phase I/Ib dose-escalation study of intravenously administered SB 11285 alone and in combination with nivolumab in patients with advanced solid tumours. Ann Oncol. 2020;31. https://doi.org/10.1016/j.annonc.2020.08.712.
Jang SC, Economides KD, Moniz RJ, Sia CL, Lewis N, McCoy C, et al. ExoSTING, an extracellular vesicle loaded with STING agonists, promotes tumor immune surveillance. Commun Biol. 2021;4(1):497. https://doi.org/10.1038/s42003-021-02004-5.
Codiak Biosciences. A First-in-Human Study of CDK-002 (exoSTING) in Subjects With Advanced/Metastatic, Recurrent, Injectable Solid Tumors 2022 [updated May 20, 2022; cited 2022 May 29]. Available from: https://clinicaltrials.gov/ct2/show/NCT04592484.
Riese R, Luke J, Lewis K, Janku F, Piha-Paul S, Verschraegen C, et al. 500 SYNB1891, a bacterium engineered to produce a STING agonist, demonstrates target engagement in humans following intratumoral injection. J Immunother Cancer. 2021;9(Suppl 2):A532-A. https://doi.org/10.1136/jitc-2021-SITC2021.500.
Janku F, Luke JJ, Brennan A, Riese R, Varterasian M, Armstrong MB, et al. Abstract CT110: Intratumoral injection of SYNB1891, a synthetic biotic designed to activate the innate immune system, demonstrates target engagement in humans including intratumoral STING activation. Cancer Res. 2021;81(13_Supplement):CT110-CT. https://doi.org/10.1158/1538-7445.Am2021-ct110.
Ramos JC. Safety Trial of STING-dependent Activators and Stimulated Dendritic Cells for Aggressive Relapsed/Refractory Leukemias 2022 [updated April 11, 2022; cited 2022 May 29]. Available from: https://clinicaltrials.gov/ct2/show/NCT05321940.
Gulla A, Morelli E, Samur MK, Botta C, Hideshima T, Bianchi G, et al. Bortezomib induces anti-multiple myeloma immune response mediated by cGAS/STING pathway activation. Blood Cancer Discov. 2021;2(5). https://doi.org/10.1158/2643-3230.BCD-21-0047.
Pantelidou C, Sonzogni O, De Oliveria Taveira M, Mehta AK, Kothari A, Wang D, et al. PARP inhibitor efficacy depends on CD8. Cancer Discov. 2019;9(6):722–37. https://doi.org/10.1158/2159-8290.CD-18-1218.
• Sen T, Rodriguez BL, Chen L, Corte CMD, Morikawa N, Fujimoto J, et al. Targeting DNA damage response promotes antitumor immunity through STING-mediated T-cell activation in small cell lung cancer. Cancer Discov. 2019;9(5):646–61. https://doi.org/10.1158/2159-8290.CD-18-1020. This manuscript presents in vivo preclinical data indicating that inhibition of DNA damage response proteins leads to activation of the STING pathway.
Ji F, Zhang F, Zhang M, Long K, Xia M, Lu F, et al. Targeting the DNA damage response enhances CD70 CAR-T cell therapy for renal carcinoma by activating the cGAS-STING pathway. J Hematol Oncol. 2021;14(1):152. https://doi.org/10.1186/s13045-021-01168-1.
Wang Z, Chen J, Hu J, Zhang H, Xu F, He W, et al. cGAS/STING axis mediates a topoisomerase II inhibitor-induced tumor immunogenicity. J Clin Invest. 2019;129(11):4850–62. https://doi.org/10.1172/JCI127471.
Tian J, Zhang D, Kurbatov V, Wang Q, Wang Y, Fang D, et al. 5-Fluorouracil efficacy requires anti-tumor immunity triggered by cancer-cell-intrinsic STING. EMBO J. 2021;40(7):e106065. https://doi.org/10.15252/embj.2020106065.
Wongthida P, Catcott K, Lancaster K, Bentley K, Dirksen A, Du B, et al. 785 STING-agonist ADCs targeting tumor-associated antigens coordinate immune-mediated killing of antigen-negative cancer cells. J Immunother Cancer. 2021;9(Suppl 2):A820. https://doi.org/10.1136/jitc-2021-SITC2021.785.
•• Pan B-S, Perera SA, Piesvaux JA, Presland JP, Schroeder GK, Cumming JN, et al. An orally available non-nucleotide STING agonist with antitumor activity. Science. 2020;369(6506):eaba6098. https://doi.org/10.1126/science.aba6098. This manuscript describes a novel delivery mechanism with the oral administration of a non-nucleotide STING agonist.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Conflict of Interest
JBH and AJK have nothing to disclose. Randy F. Sweis reports research grants from Astellas, AstraZeneca, Bristol Myers Squibb (BMS), CytomX, Eisai, Eli Lilly, Genentech/Roche, Immunocore, Moderna, Mirati, Novartis, Pfizer, and QED Therapeutics; consulting/advisory fees from Astellas, AstraZeneca, BMS, Eisai, Janssen, Mirati, Pfizer, and Seattle Genetics; and pending patent for Neoantigens in Cancer.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical on Evolving Therapies
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hines, J.B., Kacew, A.J. & Sweis, R.F. The Development of STING Agonists and Emerging Results as a Cancer Immunotherapy. Curr Oncol Rep 25, 189–199 (2023). https://doi.org/10.1007/s11912-023-01361-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11912-023-01361-0