Abstract
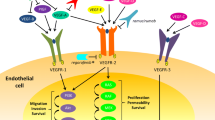
Angiogenesis is a multistep process that plays a key role in cancer growth and metastases. It is mediated through multiple vascular endothelial growth factor receptors (VEGFRs) and their ligands. The expression of VEGFR-2 is upregulated in tumor endothelial cells, and it is considered to be the primary receptor driving malignant angiogenesis. Ramucirumab (IMC-1121B, LY3009806) is a fully human monoclonal antibody that directly binds to VEGFR-2 with high affinity and specificity. It is also the most recent addition to our armamentarium of anti-angiogenic drugs approved for the treatment of metastatic colorectal cancer (CRC). The aim of this review is to summarize the pre-clinical and clinical development of ramucirumab and discuss its place in the current treatment paradigm for metastatic CRC.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66(1):7–30. doi:10.3322/caac.21332.
Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z. Vascular endothelial growth factor (VEGF) and its receptors. FASEB J. 1999;13(1):9–22.
Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9(6):669–76. doi:10.1038/nm0603-669.
Folkman J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med. 1995;1(1):27–31.
Hicklin DJ, Ellis LM. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol Off J Am Soc Clin Oncol. 2005;23(5):1011–27. doi:10.1200/jco.2005.06.081.
Sawano A, Iwai S, Sakurai Y, Ito M, Shitara K, Nakahata T, et al. Flt-1, vascular endothelial growth factor receptor 1, is a novel cell surface marker for the lineage of monocyte-macrophages in humans. Blood. 2001;97(3):785–91.
Gabrilovich DI, Chen HL, Girgis KR, Cunningham HT, Meny GM, Nadaf S, et al. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat Med. 1996;2(10):1096–103.
Barleon B, Sozzani S, Zhou D, Weich HA, Mantovani A, Marme D. Migration of human monocytes in response to vascular endothelial growth factor (VEGF) is mediated via the VEGF receptor flt-1. Blood. 1996;87(8):3336–43.
Zhang HT, Craft P, Scott PA, Ziche M, Weich HA, Harris AL, et al. Enhancement of tumor growth and vascular density by transfection of vascular endothelial cell growth factor into MCF-7 human breast carcinoma cells. J Natl Cancer Inst. 1995;87(3):213–9.
Amini A, Masoumi Moghaddam S, Morris DL, Pourgholami MH. The critical role of vascular endothelial growth factor in tumor angiogenesis. Curr Cancer Drug Targets. 2012;12(1):23–43.
Tugues S, Koch S, Gualandi L, Li X, Claesson-Welsh L. Vascular endothelial growth factors and receptors: anti-angiogenic therapy in the treatment of cancer. Mol Asp Med. 2011;32(2):88–111. doi:10.1016/j.mam.2011.04.004.
Gille H, Kowalski J, Li B, LeCouter J, Moffat B, Zioncheck TF, et al. Analysis of biological effects and signaling properties of Flt-1 (VEGFR-1) and KDR (VEGFR-2). A reassessment using novel receptor-specific vascular endothelial growth factor mutants. J Biol Chem. 2001;276(5):3222–30. doi:10.1074/jbc.M002016200.
Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N. Vascular endothelial growth factor is a secreted angiogenic mitogen. Science. 1989;246(4935):1306–9.
Witte L, Hicklin DJ, Zhu Z, Pytowski B, Kotanides H, Rockwell P, et al. Monoclonal antibodies targeting the VEGF receptor-2 (Flk1/KDR) as an anti-angiogenic therapeutic strategy. Cancer Metastasis Rev. 1998;17(2):155–61.
Takahashi Y, Kitadai Y, Bucana CD, Cleary KR, Ellis LM. Expression of vascular endothelial growth factor and its receptor, KDR, correlates with vascularity, metastasis, and proliferation of human colon cancer. Cancer Res. 1995;55(18):3964–8.
Ellis LM, Staley CA, Liu W, Fleming RY, Parikh NU, Bucana CD, et al. Down-regulation of vascular endothelial growth factor in a human colon carcinoma cell line transfected with an antisense expression vector specific for c-src. J Biol Chem. 1998;273(2):1052–7.
Warren RS, Yuan H, Matli MR, Gillett NA, Ferrara N. Regulation by vascular endothelial growth factor of human colon cancer tumorigenesis in a mouse model of experimental liver metastasis. J Clin Invest. 1995;95(4):1789–97. doi:10.1172/jci117857.
Prewett M, Huber J, Li Y, Santiago A, O’Connor W, King K, et al. Antivascular endothelial growth factor receptor (fetal liver kinase 1) monoclonal antibody inhibits tumor angiogenesis and growth of several mouse and human tumors. Cancer Res. 1999;59(20):5209–18.
Bruns CJ, Liu W, Davis DW, Shaheen RM, McConkey DJ, Wilson MR, et al. Vascular endothelial growth factor is an in vivo survival factor for tumor endothelium in a murine model of colorectal carcinoma liver metastases. Cancer. 2000;89(3):488–99.
Tonra JR, Deevi DS, Corcoran E, Li H, Wang S, Carrick FE, et al. Synergistic antitumor effects of combined epidermal growth factor receptor and vascular endothelial growth factor receptor-2 targeted therapy. Clin Cancer Res. 2006;12(7 Pt 1):2197–207. doi:10.1158/1078-0432.ccr-05-1682.
Posey JA, Ng TC, Yang B, Khazaeli MB, Carpenter MD, Fox F, et al. A phase I study of anti-kinase insert domain-containing receptor antibody, IMC-1C11, in patients with liver metastases from colorectal carcinoma. Clin Cancer Res. 2003;9(4):1323–32.
Lu D, Jimenez X, Zhang H, Bohlen P, Witte L, Zhu Z. Selection of high affinity human neutralizing antibodies to VEGFR2 from a large antibody phage display library for antiangiogenesis therapy. Int J Cancer. 2002;97(3):393–9.
Lu D, Shen J, Vil MD, Zhang H, Jimenez X, Bohlen P, et al. Tailoring in vitro selection for a picomolar affinity human antibody directed against vascular endothelial growth factor receptor 2 for enhanced neutralizing activity. J Biol Chem. 2003;278(44):43496–507. doi:10.1074/jbc.M307742200.
Miao HQ, Hu K, Jimenez X, Navarro E, Zhang H, Lu D, et al. Potent neutralization of VEGF biological activities with a fully human antibody Fab fragment directed against VEGF receptor 2. Biochem Biophys Res Commun. 2006;345(1):438–45. doi:10.1016/j.bbrc.2006.04.119.
Spratlin JL, Cohen RB, Eadens M, Gore L, Camidge DR, Diab S, et al. Phase I pharmacologic and biologic study of ramucirumab (IMC-1121B), a fully human immunoglobulin G1 monoclonal antibody targeting the vascular endothelial growth factor receptor-2. J Clin Oncol Off J Am Soc Clin Oncol. 2010;28(5):780–7. doi:10.1200/jco.2009.23.7537.
Chiorean EG, Hurwitz HI, Cohen RB, Schwartz JD, Dalal RP, Fox FE, et al. Phase I study of every 2- or 3-week dosing of ramucirumab, a human immunoglobulin G1 monoclonal antibody targeting the vascular endothelial growth factor receptor-2 in patients with advanced solid tumors. Ann Oncol. 2015;26(6):1230–7. doi:10.1093/annonc/mdv144. Phase 1 study evaluating ramucirumab in two-weekly and three-weekly dosing regimen.
Yoshino T, Yamazaki K, Gotoh M, Nasroulah F, Gao L, Yoshizuka N, et al. Safety and pharmacokinetics of second-line ramucirumab plus FOLFIRI in Japanese patients with metastatic colorectal carcinoma. Anticancer Res. 2015;35(7):4003–7. Phase 1B study demonstrating safety of ramucirumab and FOLFIRI combination in metastatic colorectal cancer.
Deng R, Jin F, Prabhu S, Iyer S. Monoclonal antibodies: what are the pharmacokinetic and pharmacodynamic considerations for drug development? Expert Opin Drug Metab Toxicol. 2012;8(2):141–60. doi:10.1517/17425255.2012.643868.
Chow LQM, Smith DC, Tan AR, Denlinger CS, Wang D, Shepard DR et al. Phase II study evaluating the effect of concomitant ramucirumab (IMC-1121B; RAM) on the pharmacokinetics (PK) of paclitaxel (PTX) and the PK of RAM as monotherapy in patients (pts) with advanced malignant solid tumors. ASCO Meeting Abstracts. 2014;32(15_suppl):e13554.
Wang D, Braiteh FS, Lee JJ, Denlinger CS, Shepard DR, Chaudhary A et al. Phase II study evaluating the effect of concomitant ramucirumab (RAM) on the pharmacokinetics (PK) of irinotecan (IRI) and its metabolite SN-38 when coadministered with folinic acid (FA) and 5-fluorouracil (5-FU) (FOLFIRI) in patients (pts) with advanced malignant solid tumors. ASCO Meeting Abstracts. 2015;33(3_suppl):691.
Garcia-Carbonero R, Rivera F, Maurel J, Ayoub JP, Moore MJ, Cervantes A, et al. An open-label phase II study evaluating the safety and efficacy of ramucirumab combined with mFOLFOX-6 as first-line therapy for metastatic colorectal cancer. Oncologist. 2014;19(4):350–1. doi:10.1634/theoncologist.2014-0028.
Hochster HS, Catalano PJ, Mitchell EP, Cohen DJ, O’Dwyer PJ, Benson AB et al. E7208: A randomized phase II trial of irinotecan and cetuximab (IC) versus IC plus ramucirumab (ICR) in second-line therapy of KRAS wild-type colorectal cancer (CRC). ASCO Meeting Abstracts. 2014;32(15_suppl):TPS3665.
Tabernero J, Yoshino T, Cohn AL, Obermannova R, Bodoky G, Garcia-Carbonero R, et al. Ramucirumab versus placebo in combination with second-line FOLFIRI in patients with metastatic colorectal carcinoma that progressed during or after first-line therapy with bevacizumab, oxaliplatin, and a fluoropyrimidine (RAISE): a randomised, double-blind, multicentre, phase 3 study. Lancet Oncol. 2015;16(5):499–508. doi:10.1016/s1470-2045(15)70127-0. Phase 3 trial demonstrating the efficacy of ramucirumab in the second-line treatment of metastatic colorectal cancer.
Fuchs CS, Tomasek J, Yong CJ, Dumitru F, Passalacqua R, Goswami C, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2014;383(9911):31–9. doi:10.1016/s0140-6736(13)61719-5. Phase 3 trial demonstrating the efficacy of ramucirumab in the second-line treatment of metastatic gastric cancer.
Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol. 2014;15(11):1224–35. doi:10.1016/s1470-2045(14)70420-6. Phase 3 trial demonstrating the efficacy of ramucirumab plus paclitaxel in the second-line treatment of metastatic gastric cancer.
Garon EB, Ciuleanu TE, Arrieta O, Prabhash K, Syrigos KN, Goksel T, et al. Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial. Lancet. 2014;384(9944):665–73. doi:10.1016/s0140-6736(14)60845-x. Phase 3 trial demonstrating the efficacy of ramucirumab plus docetaxel in the second-line treatment of non-small-cell lung cancer.
Hatch AJ, Clarke JM, Nixon AB, Hurwitz HI. Identifying blood-based protein biomarkers for antiangiogenic agents in the clinic: a decade of progress. Cancer J. 2015;21(4):322–6. doi:10.1097/ppo.0000000000000129.
Zhu AX, Finn RS, Mulcahy M, Gurtler J, Sun W, Schwartz JD, et al. A phase II and biomarker study of ramucirumab, a human monoclonal antibody targeting the VEGF receptor-2, as first-line monotherapy in patients with advanced hepatocellular cancer. Clin Cancer Res. 2013;19(23):6614–23. doi:10.1158/1078-0432.ccr-13-1442.
Goyal L, Ancukiewicz M, Jain RK, Zhu AX, Duda DG. A comparative study of circulating biomarkers of anti-VEGF therapy in phase II trials in advanced hepatocellular carcinoma (HCC) patients (pts). ASCO Meeting Abstracts. 2014;32(15_suppl):2543.
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350(23):2335–42. doi:10.1056/NEJMoa032691.
Bennouna J, Sastre J, Arnold D, Osterlund P, Greil R, Van Cutsem E, et al. Continuation of bevacizumab after first progression in metastatic colorectal cancer (ML18147): a randomised phase 3 trial. Lancet Oncol. 2013;14(1):29–37. doi:10.1016/s1470-2045(12)70477-1. Phase 3 trial demonstrating the efficacy of bevacizumab in the second-line treatment of metastatic colorectal cancer.
Holash J, Davis S, Papadopoulos N, Croll SD, Ho L, Russell M, et al. VEGF-Trap: a VEGF blocker with potent antitumor effects. Proc Natl Acad Sci U S A. 2002;99(17):11393–8. doi:10.1073/pnas.172398299.
Van Cutsem E, Tabernero J, Lakomy R, Prenen H, Prausova J, Macarulla T, et al. Addition of aflibercept to fluorouracil, leucovorin, and irinotecan improves survival in a phase III randomized trial in patients with metastatic colorectal cancer previously treated with an oxaliplatin-based regimen. J Clin Oncol Off J Am Soc Clin Oncol. 2012;30(28):3499–506. doi:10.1200/jco.2012.42.8201. Phase 3 trial demonstrating the efficacy of ziv-aflibercept in the second-line treatment of metastatic colorectal cancer.
Goel G, Sun W. Ramucirumab, another anti-angiogenic agent for metastatic colorectal cancer in second-line setting—its impact on clinical practice. J Hematol Oncol. 2015;8:92. doi:10.1186/s13045-015-0183-8.
Goldstein DA, El-Rayes BF. Considering efficacy and cost, where does ramucirumab fit in the management of metastatic colorectal cancer? Oncologist. 2015;20(9):981–2. doi:10.1634/theoncologist.2015-0028. Cost analysis of one month treatment with bevacizumab, ziv-aflibercept and ramucirumab found ramucirumab to be considerably more expensive than others.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Therapeutic Approaches to Metastatic Colorectal Cancers
Rights and permissions
About this article
Cite this article
Goel, G., Chauhan, A. & Hosein, P. Ramucirumab: a Novel Anti-Angiogenic Agent in the Treatment of Metastatic Colorectal Cancer. Curr Colorectal Cancer Rep 12, 232–240 (2016). https://doi.org/10.1007/s11888-016-0329-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11888-016-0329-2