Abstract
Introduction
Imatinib is a first-line therapy to treat chronic myeloid leukemia (CML) patients.
Materials and methods
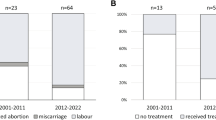
We selected 22 CML cases of pregnancies and reported 9 accidental and 13 planned pregnancies that were on imatinib therapy. Willing female patients remaining in complete hematological, cytogenetic and major molecular responses for at least 2 years planned for pregnancy and were advised to stop imatinib for 1 month prior to conception and 3 months after conception (first trimester). Willing male patients stopped therapy 1 month prior to conception of their wives. In unplanned cases, all patients were in exposure to imatinib during conception. Twenty-two pregnancies resulted in seven male children and eight female children. There were three spontaneous abortions and four elective abortions along with one case of hypospadia and another one of mild hydrocephalus.
Conclusion
We may suggest that planned pregnancy during therapy may be encouraged but imatinib therapy in unplanned pregnancy can cause spontaneous abortion or congenital anomaly.
Similar content being viewed by others
References
Braziel RM, Launder TM, Druker BJ et al (2002) Hematopathologic and cytogenetic findings in imatinib mesylate treated CML patients: 14 months’ experience. Blood 100:435–441
Druker BJ, Guilhot F, O’Brien SG et al (2006) Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med 355(23):2408–2417
Druker BJ, Tamura S, Buchdunger E et al (1996) Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat Med 2:561–566. doi:10.1038/nm0596-561
Quintas-Cardiama A, Kantarjian H, Cortes J (2009) Imatinib and beyond-exploring the full potential of targeted therapy for CML. Nat Rev Clin Oncol 6(9):535–543. doi:10.1038/nrclinonc.2009.112
National Cancer Institute Washington DC: Annual cancer statistics review. USA Department of Health and Human Services. www.cancer.gov
Fey MF, Surbek D (2008) Leukaemia and pregnancy. Recent Results Cancer Res 178:87–110
Hensley ML, Ford JM (2003) Imatinib treatment: specific issues related to safety, fertility and pregnancy. Semin Hematol 40(2 Suppl 2):21–25
Cole S, Kantarjian H, Ault P, Cortes JE (2009) Successful completion of pregnancy in a patient with chronic myeloid leukemia without active intervention: a case report and review of literature. Clin Lymphoma Myeloma 9(4):324–327
Pye SM, Cortes J, Ault P et al (2008) The effects of imatinib on pregnancy outcome. Blood 111(12):5505–5508
Faderal S, Talpaz Estrov E (1999) The biology of chronic myeloid leukemia. N Engl J Med 341(3):164–172
Ali R, Ozkalemkas F, Ozkocaman V et al (2004) Successful pregnancy and delivery in a patient with chronic myelogenous leukemia (CML), and management of CML with Leukapheresis during pregnancy: a case report and review of the literature. J Clin Oncol 34(4):215–217
Mukhopadhyay A, Dasgupta S, Mukhopadhyay S et al (2012) Imatinib mesylate therapy in patients of chronic myeloid leukemia with Philadelphia chromosome positive: an experience from eastern India. Indian J Haematol Blood Transfus 28:82–88
Kantarjian HM, Cortes JE, O’Brien S (2003) Imatinib mesylate therapy in newly diagnosed patients with Philadelphia chromosome-positive chronic myelogenous leukemia: high incidence of early complete and major cytogenetic responses. Blood 101(1):97–100
Jacob LA, Bapsy PP, BabuK Govind et al (2007) Lokanatha imatinib mesylate in newly diagnosed patients of chronic myeloid leukemia. Indian J Med Paediatr Oncol 28:1
Deshmukh C, Saikia T, Bakshi A et al (2005) Imatinib mesylate in chronic myeloid leukemia: a prospective, single arm, non-randomized study. J Assoc Phy India 53:291–295
Guoqing W, Shamudheen R, Delong L (2010) First line treatment for chronic myeloid leukemia: dasatinib, nilotinib or imatinib. J Hematol Oncol 3(47):3–10
Russell MA, Carpenter MW, Akhtar MS et al (2007) Imatinib mesylate and metabolite concentrations in maternal blood, umbilical cord blood, placenta and breast milk. J Perinatol 27:241–243
Ali R, Ozkalemkas F, Ozcelik T et al (2006) Imatinib and pregnancy. J Clin Oncol 24(23):3812–3813
Ault P, Cortes J (2010) Planned pregnancy for a patient with chronic myeloid leukemia. Inter J Oncol 7(2):1528–8331
Risch HA, Weiss NS, Clarke EA et al (1988) Miller AB. Risk factors for spontaneous abortion and its recurrence. Am J Epidemiol 128:420–430
Ault P, Kantarjian H, O’Brien S et al (2006) Pregnancy among patients with chronic myeloid leukemia treated with imatinib. J Clin Oncol 24:1204–1208
Schultheis Beate, Nijmeijer BA, Yin H et al (2012) Imatinib mesylate at therapeutic doses has no impact on folliculogenesis or spermatogenesis in a leukaemic mouse model. Leuk Res 36(3):271–274
Seshadri T, Seymour JF, McArthur GA (2004) Oligospermia in a patient receiving imatinib therapy for the hypereosinophilic syndrome. N Engl J Med 351:2134–2135
Heim C, Minniear K, Dann CT (2011) Imatinib has deleterious effects on differentiating spermatogonia while sparing spermatogonial stem cell self renewal. Reprod Toxicol 31(4):454–463
Kuwabara A, Babb A, Ibrahim A et al (2010) Poor outcome after reintroduction of imatinib in patients with chronic myeloid leukemia who interrupt therapy on account of pregnancy without having achieved an optimal response. Blood 116:1014–1016
Acknowledgment
We owe our sincere acknowledgement to Max Foundation, USA and Novartis, India with thanks for providing the patients with imatinib free of cost. We also acknowledge all of our patients and other fellows of Pathology and Molecular Biology department for their constant help and support. We are also grateful to West Bengal University of Health Sciences under which our Institute is affiliated.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mukhopadhyay, A., Dasgupta, S., Kanti Ray, U. et al. Pregnancy outcome in chronic myeloid leukemia patients on imatinib therapy. Ir J Med Sci 184, 183–188 (2015). https://doi.org/10.1007/s11845-014-1084-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-014-1084-5