Abstract
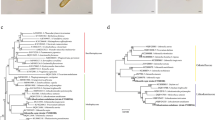
The mitochondrial genomes possess genes of five complexes involved in respiration. Mitochondrial gene content greatly varies across eukaryotes. Frequent gene losses have been reported in the mitogenomes of halophytes. In this study, we report, assemble, and annotate complete mitochondrial genome of a halophyte Pandanus odorifer (Forssk.) Kuntze (Pandanaceae). It is a commercially important species for its essential oil. It grows naturally in coastal regions and can withstand winds as well as salt sprays. The mitogenome of P. odorifer is circular with length of 330,962 bp. The genome contains 33 protein coding genes, 3 rRNAs, and 6 tRNAs. The analysis revealed that the important respiratory genes ccmC, cox1, nad1, nad4, and nad9 were pseudogenized in mitogenome but nuclear encoded copies of cox1, nad1, and nad9 were recovered. The phylogenetic analysis placed P. odorifer sister to P. tectorius. Further, mitogenome was compared with the other monocot mitogenomes for their gene contents. Thus, the study provides first complete mitochondrial genome for the family Pandanaceae and order Pandanales.








Similar content being viewed by others
Data availability
The mitogenome sequence can be accessed via accession number ON055386 on NCBI website https://www.ncbi.nlm.nih.gov. The associated transcriptome data is available in NCBI repository as SRA BioProject ID: PRJNA389238.
References
Adams KL, Palmer JD (2003) Evolution of mitochondrial gene content: gene loss and transfer to the nucleus. Mol Phylogenet Evol 29:380–395. https://doi.org/10.1016/S1055-7903(03)00194-5
Adams KL, Qiu Y-L, Stoutemyer M, Palmer JD (2000) Punctuated evolution of mitochondrial gene content: High and variable rates of mitochondrial gene loss and transfer to the nucleus during angiosperm evolution. Proc Natl Acad Sci 99:9905–9912. https://doi.org/10.1073/pnas.04269489
Aljohi HA, Liu W, Lin Q, Zhao Y, Zeng J, Alamer A, Alanazi IO, Alawad AO, Al-Sadi AM, Hu S, Yu J (2016) Complete sequence and analysis of coconut palm (Cocos nucifera) mitochondrial genome. PLoS One 11(10):e0163990. https://doi.org/10.1371/journal.pone.0163990
Alverson AJ, Wei X, Rice DW, Stern DB, Barry K, Palmer JD (2010) Insights into the evolution of mitochondrial genome size from complete sequences of Citrullus lanatus and Cucurbita pepo (Cucurbitaceae). Mol Biol Evol 27:1436–1448. https://doi.org/10.1093/molbev/msq029
Bendich AJ (2007) The size and form of chromosomes are constant in the nucleus, but highly variable in bacteria, mitochondria and chloroplasts. BioEssays 29:474–483. https://doi.org/10.1002/bies.20576
Bi C, Paterson AH, Wang X, Xu Y, Wu D, Qu Y, Jiang A, Ye Q, Ye N (2016) Analysis of the complete mitochondrial genome sequence of the diploid cotton Gossypium raimondii by comparative genomics approaches. Biomed Res Int 2016:1–18. https://doi.org/10.1155/2016/5040598
Burger G, Jackson CJ, Waller RF (2012) Unusual mitochondrial genomes and genes. Organelle genetics: evolution of organelle genomes and gene expression. Springer-Verlag, Berlin Heidelberg, pp 41–77
Cai X, Jiao C, Sun H, Wang X, Xu C, Fei Z, Wang Q (2017) The complete mitochondrial genome sequence of spinach, Spinacia oleracea L. Mitochondrial DNA B Resour 2:339–340. https://doi.org/10.1080/23802359.2017.1334518
Chase MW, Christenhusz MJ, Fay MF, Byng JW, Judd WS, Soltis DE, Mabberley DJ, Sennikov AN, Soltis PS, Stevens PF (2016) An update of the angiosperm phylogeny group classification for the orders and families of flowering plants: APG IV. Bot J Linn Soc 181:1–20. https://doi.org/10.1111/boj.12385
Cheng Y, He X, Priyadarshani SV, Wang Y, Ye L, Shi C, Ye K, Zhou Q, Luo Z, Deng F, Cao L (2021) Assembly and comparative analysis of the complete mitochondrial genome of Suaeda glauca. BMC Genomics 22:1–15. https://doi.org/10.1186/s12864-021-07490-9
Cho Y, Mower JP, Qiu Y-L, Palmer JD (2004) Mitochondrial substitution rates are extraordinarily elevated and variable in a genus of flowering plants. Proc Natl Acad Sci U S A 101:17741–17746. https://doi.org/10.1073/pnas.040830210
Cole LW, Guo W, Mower JP, Palmer JD (2018) High and variable rates of repeat-mediated mitochondrial genome rearrangement in a genus of plants. Mol Biol Evol 35:2773–2785. https://doi.org/10.1093/molbev/msy176
Darshetkar AM, Patil SS, Pable AA, Nadaf AB, Barvkar VT (2022) Chloroplast genome sequence of Pandanus odorifer (Forssk.) Kuntze: genome features, mutational hotspots and phylogenetic analyses. Biologia (bratisl). https://doi.org/10.1007/s11756-022-01155-y
Deng W, Nickle DC, Learn GH, Maust B, Mullins JI (2007) ViroBLAST: a stand-alone BLAST web server for flexible queries of multiple databases and user’s datasets. Bioinformatics 23:2334–2336. https://doi.org/10.1093/bioinformatics/btm331
Gerke P, Szövényi P, Neubauer A, Lenz H, Gutmann B, McDowell R, Small I, Schallenberg-Rüdinger M, Knoop V (2020) Towards a plant model for enigmatic U-to-C RNA editing: the organelle genomes, transcriptomes, editomes and candidate RNA editing factors in the hornwort Anthoceros agrestis. New Phytol 225:1974–1992. https://doi.org/10.1111/nph.16297
Gray MW (2014) The pre-endosymbiont hypothesis: a new perspective on the origin and evolution of mitochondria. Cold Spring Harb Perspect Biol 6:a016097. https://doi.org/10.1101/cshperspect.a016097
Gray MW, Burger G, Lang BF (1999) Mitochondrial evolution. Science 283(5407):1476–1481
Gray MW, Lang BF, Burger G (2004) Mitochondria of protists. Annu Rev Genet 38:477–524. https://doi.org/10.1146/annurev.genet.37.110801.142526
Greiner S, Lehwark P, Bock R (2019) OrganellarGenomeDRAW (OGDRAW) version 1.3.1: expanded toolkit for the graphical visualization of organellar genomes. Nucleic Acids Res 47:W59–W64. https://doi.org/10.1093/nar/gkz238
Grewe F, Zhu A, Mower JP (2016) Loss of a trans-splicing nad1 intron from Geraniaceae and transfer of the maturase gene matR to the nucleus in Pelargonium. Genome Biol Evol 8:3193–3201. https://doi.org/10.1093/gbe/evw233
Gualberto JM, Mileshina D, Wallet C, Niazi AK, Weber-Lotfi F, Dietrich A (2014) The plant mitochondrial genome: dynamics and maintenance. Biochimie 100:107–120. https://doi.org/10.1016/j.biochi.2013.09.016
Guo W, Mower JP (2013) Evolution of plant mitochondrial intron-encoded maturases: frequent lineage-specific loss and recurrent intracellular transfer to the nucleus. J Mol Evol 77:43–54. https://doi.org/10.1007/s00239-013-9579-7
Guo W, Zhu A, Fan W, Mower JP (2017) Complete mitochondrial genomes from the ferns Ophioglossum californicum and Psilotum nudum are highly repetitive with the largest organellar introns. New Phytol 213:391–403. https://doi.org/10.1111/nph.14135
Kamala S, Karthikeyan R, Sengottuvelu S (2016) Pandanus odoratissimus: a potential cardioprotective herb against isoproterenol induced myocardial infarction in rats. Sch Acad J Biosci 4:684–687
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780. https://doi.org/10.1093/molbev/mst010
Kim B, Kim K, Yang TJ, Kim S (2016) Completion of the mitochondrial genome sequence of onion (Allium cepa L.) containing the CMS-S male-sterile cytoplasm and identification of an independent event of the ccmFN gene split. Curr Genet 62:873–885. https://doi.org/10.1007/s00294-016-0595-1
Kozik A, Rowan BA, Lavelle D, Berke L, Schranz ME, Michelmore RW, Christensen AC (2019) The alternative reality of plant mitochondrial DNA: One ring does not rule them all. PLoS Genet 15:e1008373. https://doi.org/10.1371/journal.pgen.1008373
Kubo T, Nishizawa S, Sugawara A, Itchoda N, Estiati A, Mikami T (2000) The complete nucleotide sequence of the mitochondrial genome of sugar beet (Beta vulgaris L.) reveals a novel gene for tRNA Cys (GCA). Nucleic Acids Res 28:2571–2576. https://doi.org/10.1093/nar/28.13.2571
Kumari S, Kumar P, Wanjari M, Palani S (2012) Antidiabetic activity of Pandanus fascicularis Lamk - aerial roots in alloxan-induced hyperglycemic rats. Int J Nutr Pharmacol Neurol Dis 2:105. https://doi.org/10.4103/2231-0738.95943
Lam VKY, Gomez MS, Graham SW (2015) The highly reduced plastome of mycoheterotrophic Sciaphila (Triuridaceae) is colinear with its green relatives and is under strong purifying selection. Genome Biol Evol 7:2220–2236. https://doi.org/10.1093/gbe/evv134
Lam VK, Darby H, Merckx VS, Lim G, Yukawa T, Neubig KM, Abbott JR, Beatty GE, Provan J, Soto Gomez M, Graham SW (2018) Phylogenomic inference in extremis: a case study with mycoheterotroph plastomes. Am J Bot 105:480–494. https://doi.org/10.1002/ajb2.1070
Lang BF, Gray MW, Burger G (1999) Mitochondrial genome evolution and the origin of eukaryotes. Annu Rev Genet 33:351–397
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359. https://doi.org/10.1038/nmeth.1923
Liu J, Jiang M, Chen H, Liu Y, Liu C, Wu W (2021) Comparative genome analysis revealed gene inversions, boundary expansions and contractions, and gene loss in the Stemona sessilifolia (Miq.) Miq. chloroplast genome. PLoS One 16:e0247736. https://doi.org/10.1371/journal.pone.0247736
Lu Q, Ye W, Lu R, Xu W, Qiu Y (2018) Phylogenomic and comparative analyses of complete plastomes of Croomia and Stemona (Stemonaceae). Int J Mol Sci 19:2383. https://doi.org/10.3390/ijms19082383
Maréchal A, Brisson N (2010) Recombination and the maintenance of plant organelle genome stability. New Phytol 186:299–317. https://doi.org/10.1111/j.1469-8137.2010.03195.x
Maughan PJ, Chaney L, Lightfoot DJ, Cox BJ, Tester M, Jellen EN, Jarvis DE (2019) Mitochondrial and chloroplast genomes provide insights into the evolutionary origins of quinoa (Chenopodium quinoa Willd.). Sci Rep 9:1–11. https://doi.org/10.1038/s41598-018-36693-6
Minh BQ, Schmidt HA, Chernomor O, Schrempf D, Woodhams MD, Von Haeseler A, Lanfear R (2020) IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol 37:1530–1534. https://doi.org/10.1093/molbev/msaa015
Mower JP (2020) Variation in protein gene and intron content among land plant mitogenomes. Mitochondrion 53:203–213. https://doi.org/10.1016/j.mito.2020.06.002
Mower JP, Sloan DB, Alverson AJ (2012) Plant mitochondrial genome diversity: the genomics revolution. Plant genome diversity volume 1: plant genomes, their residents, and their evolutionary dynamics. Springer-Verlag, Vienna, pp 123–144. https://doi.org/10.1007/978-3-7091-1130-7_9
Nadaf A, Zanan R (2012) Indian Pandanaceae-an overview. Springer, New Delhi, pp 1–163. https://doi.org/10.1007/978-81-322-0753-5
Nasim N, Behera JK, Sandeep IS, RamaRao VV, Kar B, Mishra A, Nayak S, Mohanty S (2018) Phytochemical analysis of flower from Pandanus odorifer (Forssk.) Kuntze for industrial application. Nat Prod Res 32:2494–2497. https://doi.org/10.1080/14786419.2017.1422184
Nasim N, Sandeep IS, Sahoo A, Das S, Panda MK, Acharya L, RamaRao VV, Nayak S, Mohanty S (2020) Population genetic structure and diversity analysis in economically important Pandanus odorifer (Forssk.) Kuntze accessions employing ISSR and SSR markers. Ind Crops Prod 143:111894. https://doi.org/10.1016/j.indcrop.2019.111894
Nasim N, Sandeep IS, Nayak S, Mohanty S (2021) Cultivation and utilization of Pandanus odorifer for industrial application. Medicinal Plants. Sustainable development and biodiversity, vol 28. Springer, Cham, pp 435–456. https://doi.org/10.1007/978-3-030-74779-4_15
Patel RK, Jain M (2012) NGS QC toolkit: a toolkit for quality control of next generation sequencing data. PLoS One 7:e30619. https://doi.org/10.1371/journal.pone.0030619
Petersen G, Cuenca A, Zervas A, Ross GT, Graham SW, Barrett CF, Davis JI, Seberg O (2017) Mitochondrial genome evolution in Alismatales: Size reduction and extensive loss of ribosomal protein genes. PLoS One 12:e0177606. https://doi.org/10.1371/journal.pone.0177606
Rashmi D, Nadaf A (2018) Understanding the mechanism of salt tolerance in Pandanus odorifer L. Proc Natl Acad Sci India Sect B Biol Sci 88:1557–1563. https://doi.org/10.1007/s40011-017-0900-x
Rashmi D, Barvkar VT, Nadaf A, Mundhe S, Kadoo NY (2019) Integrative omics analysis in Pandanus odorifer (Forssk.) Kuntze reveals the role of Asparagine synthetase in salinity tolerance. Sci Rep 9:1–6. https://doi.org/10.1038/s41598-018-37039-y
Rice DW, Alverson AJ, Richardson AO, Young GJ, Sanchez-Puerta MV, Munzinger J, Barry K, Boore JL, Zhang Y, DePamphilis CW, Knox EB (2013) Horizontal transfer of entire genomes via mitochondrial fusion in the angiosperm Amborella. Science (1979) 342:1468–1473. https://doi.org/10.1126/science.1246275
Rozas J, Ferrer-Mata A, Sánchez-DelBarrio JC, Guirao-Rico S, Librado P, Ramos-Onsins SE, Sánchez-Gracia A (2017) DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol Biol Evol 34:3299–3302. https://doi.org/10.1093/molbev/msx248
Satoh M, Kubo T, Nishizawa S, Estiati A, Itchoda N, Mikami T (2004) The cytoplasmic male-sterile type and normal type mitochondrial genomes of sugar beet share the same complement of genes of known function but differ in the content of expressed ORFs. Mol Genet Genom 272:247–256. https://doi.org/10.1007/s00438-004-1058-9
Shearman JR, Sangsrakru D, Ruang-Areerate P, Sonthirod C, Uthaipaisanwong P, Yoocha T, Poopear S, Theerawattanasuk K, Tragoonrung S, Tangphatsornruang S (2014) Assembly and analysis of a male sterile rubber tree mitochondrial genome reveals DNA rearrangement events and a novel transcript. BMC Plant Biol 14:1–11. https://doi.org/10.1186/1471-2229-14-45
Skippingtona E, Barkmanb TJ, Ricea DW, Palmera JD (2015) Miniaturized mitogenome of the parasitic plant viscum scurruloideum is extremely divergent and dynamic and has lost all nad genes. Proc Natl Acad Sci U S A 112:E3515–E3524. https://doi.org/10.1073/pnas.1504491112
Sloan DB, Alverson AJ, Štorchová H, Palmer JD, Taylor DR (2010) Extensive loss of translational genes in the structurally dynamic mitochondrial genome of the angiosperm Silene latifolia. BMC Evol Biol 10:1–5. https://doi.org/10.1186/1471-2148-10-274
Sloan DB, Müller K, McCauley DE, Taylor DR, Štorchová H (2012) Intraspecific variation in mitochondrial genome sequence, structure, and gene content in Silene vulgaris, an angiosperm with pervasive cytoplasmic male sterility. New Phytol 196:1228–1239. https://doi.org/10.1111/j.1469-8137.2012.04340.x
Smith DR, Keeling PJ (2015) Mitochondrial and plastid genome architecture: reoccurring themes, but significant differences at the extremes. Proc Natl Acad Sci U S A 112:10177–10184. https://doi.org/10.1073/pnas.1422049112
Soto Gomez M, Lin Q, da Silva LE, Gallaher TJ, Scherberich D, Mennes CB, Smith SY, Graham SW (2020) A bi-organellar phylogenomic study of Pandanales: inference of higher-order relationships and unusual rate-variation patterns. Cladistics 36:481–504. https://doi.org/10.1111/cla.12417
Thomson LA, Englberger L, Guarino L, Thaman RR, Elevitch CR (2006) Pandanus tectorius (pandanus) Pandanaceae (screwpine family) Species Profiles for Pacific Island Agroforestry. pp 1–29. www.traditionaltree.org
Tillich M, Lehwark P, Pellizzer T, Ulbricht-Jones ES, Fischer A, Bock R, Greiner S (2017) GeSeq - Versatile and accurate annotation of organelle genomes. Nucleic Acids Res 45:W6–W11. https://doi.org/10.1093/nar/gkx391
Wanga VO, Dong X, Oulo MA, Mkala EM, Yang JX, Onjalalaina GE, Gichua MK, Kirika PM, Gituru RW, Hu GW, Wang QF (2021) Complete chloroplast genomes of Acanthochlamys bracteata (China) and Xerophyta (Africa) (Velloziaceae): comparative genomics and phylogenomic placement. Front Plant Sci 12:1135. https://doi.org/10.3389/fpls.2021.691833
Wynn EL, Christensen AC (2019) Repeats of unusual size in plant mitochondrial genomes: identification, incidence and evolution. G3: Genes. Genomes Genet 9:549–559. https://doi.org/10.1534/g3.118.200948
Yu Y, Ouyang Y, Yao W (2018) ShinyCircos: an R/Shiny application for interactive creation of circos plot. Bioinformatics 34:1229–1231. https://doi.org/10.1093/bioinformatics/btx763
Yu R, Sun C, Liu Y, Zhou R (2021) Shifts from cis-totrans-splicing of five mitochondrial introns in Tolypanthus maclurei. PeerJ 9:e12260. https://doi.org/10.7717/peerj.12260
Acknowledgements
Authors are grateful to Savitribai Phule Pune University for providing facilities and financial support. The work was supported by SERB, Department of Science & Technology, India vide project no. CRG/2021/005730. AMD is thankful to SPPU-PDF [SPPU-PDF/ST/BL/2021/0001] for the post-doctoral fellowship support. VTB acknowledges partial funding support received from IQAC, Savitribai Phule Pune University under ASPIRE- scheme research mentorship program (18TEC000030).
Author information
Authors and Affiliations
Contributions
AMD and VTB: designed the study. SSP: carried out DNA extraction. AMD and VTB: performed analyses. AMD and SSP: wrote the original draft. AAP, ABN, and VTB: reviewed and contributed to the rewriting, editing the manuscript, and were involved in funding acquisition.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11816_2023_841_MOESM2_ESM.xlsx
Supplementary file2 RNA editing sites in the mitogenome of P. odorifer when compared to Arabidopsis thaliana, Oryza sativa Japonica group, Cocos nucifera (XLSX 30 KB)
11816_2023_841_MOESM3_ESM.tif
Supplementary file3 Fig. S1 Secondary structures of tRNAs present in the mitogenome of P. odorifer. The colour code is following the RNAfold webserver. (Green: Stems (canonical helices), Red: Multiloops (junctions), Yellow: Interior Loops, Blue: Hairpin loops, Orange: 5' and 3' unpaired region) (TIF 3039 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Darshetkar, A.M., Patil, S.S., Pable, A.A. et al. Loss of mitogenome-encoded respiratory genes complemented by nuclear transcripts in halophyte Pandanus odorifer (Forssk.) Kuntze. Plant Biotechnol Rep 18, 91–104 (2024). https://doi.org/10.1007/s11816-023-00841-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-023-00841-w