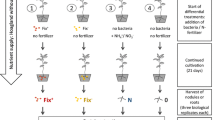
Abstract
The common bean (Phaseolus vulgaris L.)–Rhizobium etli symbiosis and crop productivity are highly affected by adverse environmental conditions that cause oxidative stress. Based on the improved symbiosis of common bean inoculated with engineered R. etli expressing the Vitreoscilla hemoglobin (VHb) (Ramírez et al., Mol Plant Microbe Interact 12:1008–1015, 1999), in this work we analyzed the effect of this strain in plants exposed to the herbicide paraquat (PQ) which generates oxidative stress. PQ-treated plants inoculated with the engineered (VHb) R. etli strain showed higher nitrogenase activity and ureide content than plants inoculated with the wild type strain. We performed microarray transcriptomic analysis to identify PQ-responsive genes in nodules elicited by engineered vs wild type strains. An evident reprogramming of the transcriptional profile was observed in PQ-treated nodules, and the global changes in gene expression were different between nodules elicited with each strain. The most relevant difference was the increased number of up-regulated PQ-responsive genes in wild type strain nodules as compared to VHb-expressing nodules. The majority of these genes were classified into biological processes/functional categories related to defense, response to abiotic stress or signaling, as revealed by Gene Ontology and MapMan analysis. Taken together our analysis suggests that the expression of VHb in R. etli bacteroids contributes to buffering the damage caused by increased reactive oxygen species, and this is reflected in nodule cells that showed decreased sensitivity to oxidative stress and response of stress-related genes. Biotechnological applications of VHb-expressing rhizobia inoculants could be further explored.





Similar content being viewed by others
References
Anand A, Duk BT, Singh S, Akbas MY, Webster DA, Stark BC, Dikshit KL (2010) Redox-mediated interactions of VHb (Vitreoscilla haemoglobin) with OxyR: novel regulation of VHb biosynthesis under oxidative stress. Biochem J 426:271–280. doi:10.1042/BJ20091417
Aparicio-Fabre R, Guillén G, Loredo M, Arellano J, Valdés-López O, Ramírez M, Íñiguez LP, Panzeri D, Castiglioni B, Cremonesi P, Strozzi F, Stella A, Girard L, Sparvoli F, Hernández G (2013) Common bean (Phaseolus vulgaris L.) PvTIFY orchestrates global changes in transcript profile response to jasmonate and phosphorus deficiency. BMC Plant Biol 13:1. doi:10.1186/1471-2229-13-26
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, Kasarskis A, Lewis S, Matese JC, Richardson JE, Ringwald M, Rubin GM, Sherlock G (2000) Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nature Genet 25:25–29. doi:10.1038/75556
Becana M, Matamoros MA, Udvardi M, Dalton DA (2010) Recent insights into antioxidant defenses of legume root nodules. New Phytol 188:960–976. doi:10.1111/j.14698137.2010.03512.x
Boldt R, Zrenner R (2003) Purine pyrimidine biosynthesis in higher plants. Physiol Plant 117:297–304
Broughton WJ, Hernández G, Blair M, Beebe S, Gepts P, Vanderleyden J (2003) Beans (Phaseolus spp): model food legume. Plant Soil 252:55–128. doi:10.1023/A:1024146710611
Brunetti C, Di Ferdinando M, Fini A, Pollastri S, Tattini M (2013) Flavonoids as antioxidants and developmental regulators: relative significance in plants and humans. Int J Mol Sci 14:3540–3555. doi:10.3390/ijms14023540
Dalton DA, Boniface C, Turner Z, Lindahl A, Kim HJ, Jelinek L, Govindarajulu M, Finger RE, Taylor CG (2009) Physiological roles of glutathione S-transferases in soybean root nodules. Plant Physiol 150:521–530. doi:10.1104/pp.109.136630
Danquah A, de Zelicourt A, Colcombet J, Hirt H (2014) The role of ABA and MAPK signaling pathways in plant abiotic stress responses. Biotechnol Adv 32:40–52. doi:10.1016/j.biotechadv.2013.09.006
Edgar R, Domrachev M, Lash AE (2002) Gene Expression Omnibus: NCBI gene expression hybridization array data repository. Nucleic Acids Res 30:207–210. doi:10.1093/nar/30.1.207
ElSayed AI, Rafudeen MS, Golldack D (2014) Physiological aspects of raffinose family oligosaccharides in plants: protection against abiotic stress. Plant Biol 16:1–8. doi:10.1111/plb.12053
Ferrer JL, Austin MB, Stewart C Jr, Noel JP (2008) Structure and function of enzymes involved in the biosynthesis of phenylpropanoids. Plant Physiol Biochem 46:356–370. doi:10.1016/j.plaphy.2007.12.009
Geckil H, Stark BC, Webster DA (2001) Cell growth and oxygen uptake of Escherichia coli and Pseudomonas aeruginosa are differently effected by the genetically engineered Vitreoscilla hemoglobin gene. J Biotechnol 85:57–66. doi:10.1016/S0168-1656(00)00384-9
Geckil H, Gencer S, Kahraman H, Erenler SO (2003) Genetic engineering of Enterobacter aerogenes with the Vitreoscilla hemoglobin gene: cell growth, survival, and antioxidant enzyme status under oxidative stress. Res Microbiol 154:425–431. doi:10.1016/S0923-2508(03)00083-4
Graham PH, Vance CP (2003) Legumes: importance and constraints to greater use. Plant Physiol 131:872–877. doi:10.1104/pp.017004
Granados-Baeza MJ, Gómez-Hernández N, Mora Y, Delgado MJ, Romero D, Girard L (2007) Novel reiterated Fnr-type proteins control the production of the symbiotic terminal oxidase cbb 3 in Rhizobium etli CFN42. Mol Plant Microbe Interact 20:1241–1249. doi:10.1094/MPMI-20-10-1241
Hardison R (1998) Hemoglobins from bacteria to man: evolution of different patterns of gene expression. J Exp Biol 201:1099–1117
Hardy RW, Holsten RD, Jackson EK, Burns RC (1968) The acetylene–ethylene assay for N2 fixation: laboratory and field evaluation. Plant Physiol 43:1185–1207
Hernández G, Ramírez M, Valdes-López O, Tesfaye M, Graham MA, Czechowski T, Schlereth A, Wandrey M, Erban A, Cheung F, Wu H, Lara M, Town CD, Kopka J, Udvardi MK, Vance CP (2007) Phosphorus stress in common bean: root transcript and metabolic responses. Plant Physiol 144:752–767. doi:10.1104/pp.107.096958
Holmberg N, Lilius G, Bailey JE, Bülow L (1997) Transgenic tobacco expressing Vitreoscilla hemoglobin exhibits enhanced growth and altered metabolite production. Nat Biotechnol 15:244–247. doi:10.1038/nbt0397-244
Joshi M, Mande S, Dikshi KL (1998) Hemoglobin biosynthesis in Vitreoscilla stercoraria DW: cloning, expression, and characterization of a new homolog of a bacterial globin gene. Appl Environ Microbiol 64:2220–2228
Kaur R, Pathania R, Sharma V, Mande SC, Dikshit KL (2002) Chimeric Vitreoscilla hemoglobin (VHb) carrying a flavoreductase domain relieves nitrosative stress in Escherichia coli: new insight into the functional role of VHb. Appl Environ Microbiol 68:152–160. doi:10.1128/AEM.68.1.152-160.2002
Khosla C, Bailey JE (1988) Heterologous expression of a bacterial haemoglobin improves the growth properties of recombinant Escherichia coli. Nature 18:633–635. doi:10.1038/331633a0
Klie S, Nikoloski Z (2012) The choice between MapMan and Gene Ontology for automated gene function prediction in plant science. Front Genet 3:115. doi:10.3389/fgene.2012.00115
Li X, Peng RH, Fan HQ, Xiong AS, Yao QH, Cheng ZM (2005) Vitreoscilla hemoglobin overexpression increases submergence tolerance in cabbage. Plant Cell Rep 23:710–715. doi:10.1186/s12870-016-0728-1
Malhotra S, Sowdhamini R (2014) Interactions among plant transcription factors regulating expression of stress-responsive genes. Bioinform Biol Insights 8:193–198. doi:10.4137/BBI.S16313
Mandon K, Pauly N, Boscari A, Brouquisse R, Frendo P, Demple B, Puppo A (2009) ROS in the legume-Rhizobium symbiosis. In: Del Río LA, Puppo A (eds) Reactive oxygen species in plant signaling. Springer, Berlin, pp 135–147
Mendoza-Soto AB, Naya L, Leija A, Hernández G (2015) Responses of symbiotic nitrogen-fixing common bean to aluminum toxicity and delineation of nodule responsive microRNAs. Front Plant Sci 6. doi:10.3389/fpls.2015.00587
Moon S, Jung KH (2014) Genome-wide expression analysis of rice ABC transporter family across spatio-temporal samples and in response to abiotic stresses. J Plant Physiol 171:1276–1288. doi:10.1016/j.jplph.2014.05.006
Naya L, Paul S, Valdés-López O, Mendoza-Soto AB, Nova-Franco B, Sosa-Valencia G, Reyes JL, Hernández G (2014) Regulation of copper homeostasis and biotic interactions by microRNA 398b in common bean. PLoS One 9:e84416. doi:10.1371/journal.pone.0084416
Peralta H, Mora Y, Salazar E, Encarnación S, Palacios R, Mora J (2004) Engineering the nifH promoter region and abolishing poly-β-hydroxybutyrate accumulation in Rhizobium etli enhance nitrogen fixation in symbiosis with Phaseolus vulgaris. Appl Environ Microbiol 70:3272–3281. doi:10.1128/AEM.70.6.3272-3281.2004
Peshev D, Vergauwen R, Moglia A, Hideg É, Van den Ende W (2013) Towards understanding vacuolar antioxidant mechanisms: a role for fructans? J Exp Bot 64:1025–1038. doi:10.1093/jxb/ers377
Pieterse CM, Van der Does D, Zamioudis C, Leon-Reyes A, Van Wees SC (2012) Hormonal modulation of plant immunity. Annu Rev Cell Dev Biol 28:489–521. doi:10.1146/annurev-cellbio-092910-154055
Ramírez M, Valderrama B, Arrendondo-Peter R, Sobéron M, Mora J, Hernández G (1999) Rhizobium etli genetically engineered for the heterologous expression of Vitreoscilla sp. hemoglobin: effect on free-living symbiosis. Mol Plant Microbe Interact 12:1008–1015. doi:10.1094/MPMI.1999.12.11.1008
Ramírez M, Guillén G, Fuentes SI, Iñiguez LP, Aparicio-Fabre R, Zamorano-Sánchez D, Encarnación-Guevara S, Panzeri D, Castiglioni B, Cremonesi P, Strozzi F, Stella A, Girard L, Sparvoli F, Hernández G (2013) Transcript profiling of common bean nodules subjected to oxidative stress. Physiol Plant 149:389–407. doi:10.1111/ppl.12040
Routledge R (1998) Fisher’s exact test. In: Armitage P, Colton T (eds) Encyclopedia of biostatistics, vol 2. Wiley, New York, pp 1519–1523
Schmutz J, McClean PE, Mamidi S, Wu GA, Cannon SB, Grimwood J, Jenkins J, Shu S, Song Q, Chavarro C, Torres-Torres M, Geffroy V, Moghaddam SM, Gao D, Abernathy B, Barry K, Blair M, Brick MA, Chovatia M, Gepts P, Goodstein DM, Gonzales M, Hellsten U, Hyten DL, Jia G, Kelly JD, Kudrna D, Lee R, Richard MM, Miklas PN, Osorno JM, Rodrigues J, Thareau V, Urrea CA, Wang M, Yu Y, Zhang M, Wing RA, Cregan PB, Rokhsar DS, Jackson SA (2014) A reference genome for common bean and genome-wide analysis of dual domestications. Nat Genet 46:707–713. doi:10.1038/ng.3008
Segovia L, Young JPW, Martínez-Romero E (1993) Reclassification of American Rhizobium leguminosarum biovar phaseoli type I strains as Rhizobium etli sp. nov. Int J Syst Evol Microbiol 43:374–377. doi:10.1099/00207713-43-2-374
Smyth GK (2004) Linear models empirical Bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol 3:1–26
Soberón M, López O, Morera C, Girard ML, Tabche M, Miranda J (1999) Enhanced nitrogen fixation in a Rhizobium etli ntrC mutant that overproduces the Bradyrhizobium japonicum symbiotic terminal oxidase cbb 3 . Appl Environ Microbiol 65:2015–2019
Stark BC, Pagilla KR, Dikshit KL (2015) Recent applications of Vitreoscilla hemoglobin technology in bioproduct synthesis and bioremediation. Appl Microbiol Biotechnol 99:1627–1636. doi:10.1007/s00253-014-6350-y
Suárez R, Wong A, Ramírez M, Barraza A, Orozco MDC, Cevallos MA, Lara M, Hernández G, Iturriaga G (2008) Improvement of drought tolerance and grain yield in common bean by overexpressing trehalose 6-phosphate synthase in rhizobia. Mol Plant Microbe Interact 21:958–966. doi:10.1094/MPMI-21-7-0958
Summerfield RJ, Huxley PA, Minchin FR (1977) Plant husbandry management techniques for growing grain legumes under simulated tropical conditions in controlled environments. Exp Agric 13:113–121. doi:10.1017/S0014479700007638
Talbi C, Sánchez C, Hidalgo-Garcia A, González EM, Arrese-Igor C, Girard L, Delgado MJ (2012) Enhanced expression of Rhizobium etli cbb 3 oxidase improves drought tolerance of common bean symbiotic nitrogen fixation. J Exp Bot 63:5035–5043. doi:10.1093/jxb/ers101
Thimm O, Bläsing O, Gibon Y, Nagel A, Meyer S, Krüger P, Selbig J, Müller LA, Rhee SY, Stitt M (2004) MapMan: a user-driven tool to display genomic data sets onto diagrams of metabolic pathways other biological processes. Plant J 37:914–939. doi:10.1111/j.1365-313X.2004.02016.x
Vadez V, Berger JD, Warkentin T, Asseng S, Kumar PR, Rao KPC (2012) Adaptation of grain legumes to climate change: a review. Agron Sustain Dev 32:31–44. doi:10.1007/s13593-011-0020-6
Verma V, Ravindran P, Kumar PP (2016) Plant hormone-mediated regulation of stress responses. BMC Plant Biol 16:1. doi:10.1186/s12870-016-0771-y
Vogels GD, Van Der Drift C (1970) Differential analyses of glyoxylate derivatives. Anal Biochem 33:143–157
Wakabayashi S, Matsubara H, Webster DA (1986) Primary sequence of a dimeric bacterial haemoglobin from Vitreoscilla. Nature 322:481–483. doi:10.1038/322481a0
Wang Z, Xiao Y, Chen W, Tang K, Zhang L (2009) Functional expression of Vitreoscilla hemoglobin (VHb) in Arabidopsis relieves submergence, nitrosative, photo-oxidative stress and enhances antioxidants metabolism. Plant Sci 176:66–77. doi:10.1186/s12870-016-0728-1
Wittenberg JB (2012) Reactivity and function of leghemoglobin. In: Caughey W (ed) Biochemical and clinical aspects of oxygen. Academic Press, London, pp 53–68
Acknowledgements
We thank Dr. Michael Dunn for critical review of this manuscript and Biol.Víctor Bustos for technical assistance. This work was supported partially by PAPIIT grant IN214308 from Dirección General de Asuntos del Personal Académico – UNAM. LP Íñiguez is a student from Doctorado en Ciencias Biomédicas – UNAM and a recipient of a studentship from CONACyT, México (No. 340334).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ramírez, M., Íñiguez, L.P., Guerrero, G. et al. Rhizobium etli bacteroids engineered for Vitreoscilla hemoglobin expression alleviate oxidative stress in common bean nodules that reprogramme global gene expression. Plant Biotechnol Rep 10, 463–474 (2016). https://doi.org/10.1007/s11816-016-0422-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-016-0422-7