Abstract
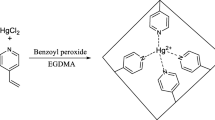
A simple lead ion imprinted sorbent was synthesized by coupling activated carbon with a known metal chelating compound, iminodiacetic acid. The ion imprinted sorbent has been characterized using Fourier transform infrared spectroscopy, elemental analysis and thermogravimetric analysis and subjected for the extraction and determination of trace Pb(II) in environmental water samples. The optimum pH value for sorption of the lead ion was 6.5. The sorption capacity of lead imprinted sorbent was 42.2 mg g−1. The chelating imprinted sorbent can be reused for five cycles of sorption-desorption without any significant change in sorption capacity. Compared with non-imprinted polymer particles, the lead ion imprinted sorbent showed high adsorption capacity, significant selectivity, good site accessibility for Pb(II). The equilibrium adsorption data of Pb(II) by modified resin were analyzed by Langmuir, Freundlich, Temkin and Redlich-Peterson models.
Similar content being viewed by others
References
F. Shah, M. Soylak, T.G. Kazi and H. I. Afridi, J. Anal. At. Spectrom., 8, 601 (2013).
Z. A. ALOthman, E. Yilmaz, M. Habila, A. Shabaka and Soylak, Mikrochim. Acta, 180, 669 (2013).
S. Candir, I. Narin and M. Soylak, Talanta, 77, 289 (2008).
Y. Lee and S.W. Oa, Environ. Eng. Res., 17(3), 145 (2012).
X. A. Li, D. M. Zhou, J. J. Xu and H.Y. Chen, Talanta, 71, 1130 (2007).
M. Tuzen, E. Sesli and M. Soylak, Food Control, 18, 806 (2007).
E. S. Yanovska, V.A. Tertykh, O.Y. Kichkiruk and A. D. Dadashev, Adsorpt. Sci. Technol., 25, 81 (2007).
A. R. Ghiasvand, R. Ghaderi and A. Kakanejadifard, Talanta, 62, 287 (2004).
T. Kiriyoma and R. Kuroda, Fresenius J. Anal. Chem., 332, 338 (1988).
J. Kubova, V. Neveral and V. Stresko, J. Anal. At. Spectrom., 9, 241 (1994).
P. L. Malvankar and V.M. Shinde, Analyst, 116, 1081 (1991).
M. Soylak and L. Elçi, nt. J. Environ. Anal. Chem., 66, 51 (1997).
R. E. Santelli, M. Gallego and M. Valcarcel, Anal. Chem., 61, 1427 (1989).
L. Elçi and S. Saraçoqglu, Talanta, 46, 1305 (1998).
S. Kagaya, Y. Araki and K. Hasegawa, Fresenius J. Anal. Chem., 366, 842 (2000).
S. Kaqava, Z. A. Malek, Y. Araki and K. Haseqawa, Anal. Sci., 18, 923 (2002).
H. Ahmad Panahi,_J. Morshedian, N. Mehmandost, E. Moniri and I. Y. Galaev, J. Chromatogr. A, 1217, 5165 (2010).
L. Elçi, Z. Arslan and J. F. Tyson, Spectrochim. Acta Part B, 55, 1109 (2000).
E.M. Soliman, M. E. Mahmoud and S. A. Ahmed, Talanta, 54, 243 (2001).
L. Elci, D. SŞahan, A. Basaran and M. Soylak, Environ. Monit. Assess., 132, 331 (2007).
X. Chang, X. Yang, X. Wei and K. Wu, Anal. Chim. Acta, 450, 231 (2001).
X. Chang, Q. Su, D. Liang, X. Wei and B. Wang, Talanta, 57, 253 (2002).
M.A. Bezerra, W. N. L. dos Santos, V. A. Lemos, M.G. A. Korn and S. L. C. Ferreira, J. Hazard. Mater., 148, 334 (2007).
K. Pyrzynska, Crit. Rev. Anal. Chem., 29, 313 (1999).
S. A. Ahmed, J. Hazard. Mater., 156, 521 (2008).
P. K. Tewari and A. K. Singh, Talanta, 56, 735 (2002).
D.M. Han, G. Z. Fang and X. P. Yan, J. Chromatogr. A, 1100, 131 (2005).
X. Jiang, C. Zhao, N. Jiang, H. Zhang and M. Liu, Food Chem., 108, 1061 (2008).
H. Ahmad Panahi, J. Morshedian, N. Mehmandost, E. Moniri and Igor Yu. Galaev, J. Chromatogr. A, 1217, 5165 (2010).
H. Ahmad Panahi, E. Mottaghinejad, A. R. Badr and E. Moniri, J. Appl. Polym. Sci., 121, 1127 (2011).
M. Kumar, D. P. S. Rathore and A. K. Singh, Talanta, 51, 1187 (2000).
F. Xie, X. Lin, X. Wu and Z. Xie, Talanta, 74, 836 (2008).
L. Vinícius, A. Gurgel, R. Pereira de Freitas and L. Frédéric Gil, Carbohydr. Polym., 74, 922 (2008).
D. Chen, B. Hu and C. Huang, Talanta, 78, 491 (2009).
B. Filiz Senkal, M. Ince, E. Yavuz and M. Yaman, Talanta, 72, 962 (2007).
E. Melek, M. Tuzen and M. Soylak, Anal. Chim. Acta, 578, 213 (2006).
P. K. Tewari and A. K. Singh, Talanta, 56, 735 (2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Naraghi, K., Panahi, H.A., Hassani, A.H. et al. Ion imprinted activated carbon solid-phase extraction coupled to flame atomic absorption spectrometry for selective determination of lead ions in environmental samples. Korean J. Chem. Eng. 31, 1818–1823 (2014). https://doi.org/10.1007/s11814-014-0104-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-014-0104-5