Abstract
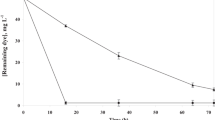
The salt-tolerant Staphylococcus cohnii strain, isolated from textile wastewater, has been found effective on decolorizing several kinds of azo dyes with different structures. The optimal conditions for azo dye acid red B (ARB) decolorization by S. cohnii were determined to be pH = 7.0 and 30°C. The decolorization efficiency increased with the increase of the salinity concentration, and around 90% of ARB (100 mg·L−1) could be decolorized in 24 h when the salinity concentration was up to 50 g·L−1. Moreover, the strain could still decolorize 19% of ARB in 24 h even when the NaCl concentration was increased to 150 g·L−1. Meanwhile, the dependence of the specific decolorization rate by S. cohnii on the ARB concentration could be described with Michaelis-Menten kinetics (K m = 585.7mg·L−1, V max = 109.8 mg·g cell−1·h−1). The addition of quinone redox mediator, named 2-hydroxy-1,4-naphthoquinone and anthraquinone-2,6-disulfonate, significantly accelerated the decolorization performance of S. cohnii. Furtherly, the activities of azoreductase (0.55 μmol·mg protein−1·min−1) and Nicotineamide adenine dinucleotide-dichlorophenol indophenol (NADH-DCIP) reductase (8.9 μmol·mg protein−1·min−1) have been observed in the crude cell extracts of S. cohnii. The decolorization products of ARB were analyzed by HPLC-MS, and the results indicated the reductive pathway was responsible for azo dye decolorization by S. cohnii.
Similar content being viewed by others
References
Stolz A. Basic and applied aspects in the microbial degradation of azo dyes. Applied Microbiology and Biotechnology, 2001, 56(1–2): 69–80
Wang X, Cheng X, Sun D, Qi H. Biodecolorization and partial mineralization of Reactive Black 5 by a strain of Rhodopseudomonas palustris. Journal of Environmental Sciences (China), 2008, 20 (10): 1218–1225
Hu T L. Kinetics of azoreductase and assessment of toxicity of metabolic products from azo dyes by Pseudomonas luteola. Water Science and Technology, 2001, 43(2): 261–269
O’Neill C, Lopez A, Esteves S, Hawkes F R, Hawkes D L, Wilcox S. Azo-dye degradation in an anaerobic-aerobic treatment system operating on simulated textile effluent. Applied Microbiology and Biotechnology, 2000, 53(2): 249–254
Ogugbue C J, Sawidis T, Oranusi N A. Evaluation of colour removal in synthetic saline wastewater containing azo dyes using an immobilized halotolerant cell system. Ecological Engineering, 2010, 37(12): 2056–2060
EPA. Profile of the textile industry. Environmental Protection Agency, Washington, DC. 1997
Dubrow S F, Boardman G D, Michelsen D L. Chemical pretreatment and aerobic-anaerobic degradation of textile dye wastewater. In: Reife A, Freeman H S, eds. Environmental Chemistry of Dyes and Pigments. New York: Wiley, 1996, 75–102
dos Santos A B, Cervantes F J, van Lier J B. Review paper on current technologies for decolourisation of textile wastewaters: perspectives for anaerobic biotechnology. Bioresource Technology, 2007, 98(12): 2369–2385
Jothimani P, Kalaichelvan G, Bhaskaran A, Selvaseelan D A, Ramasamy K. Anaerobic biodegradation of aromatic compounds. Indian Journal of Experimental Biology, 2003, 41(9): 1046–1067
Carmona M, Zamarro M T, Blázquez B, Durante-Rodríguez G, Juárez J F, Valderrama J A, Barragán M J, García J L, Díaz E. Anaerobic catabolism of aromatic compounds: a genetic and genomic view. Microbiology and Molecular Biology Reviews, 2009, 73(1): 71–133
Liu G, Zhou J, Wang J, Wang X, Jin R, Lv H. Decolorization of azo dyes by Shewanella oneidensis MR-1 in the presence of humic acids. Applied Microbiology and Biotechnology, 2011, 91(2): 417–424
van der Zee F P, Cervantes F J. Impact and application of electron shuttles on the redox (bio)transformation of contaminants: a review. Biotechnology Advances, 2009, 27(3): 256–277
Saratale R G, Saratale G D, Chang J S, Govindwar S P. Bacterial decolorization and degradation of azo dyes: a review. Journal of Taiwan Institute of Chemical Engineers, 2011, 42(1): 138–157
Harris L G, Foster S J, Richards R G. An introduction to Staphylococcus aureus, and techniques for identifying and quantifying S. aureus adhesins in relation to adhesion to biomaterials: review. European Cells & Materials, 2002, 4: 39–60
Börjesson S, Matussek A, Melin S, Löfgren S, Lindgren P E. Methicillin-resistant Staphylococcus aureus (MRSA) in municipal wastewater: an uncharted threat? Journal of Applied Microbiology, 2010, 108(4): 1244–1251
Faria C, Vaz-Moreira I, Serapicos E, Nunes O C, Manaia C M. Antibiotic resistance in coagulase negative staphylococci isolated from wastewater and drinking water. Science of the Total Environment, 2009, 407(12): 3876–3882
Mallick S, Dutta T K. Kinetics of phenanthrene degradation by Staphylococcus sp. strain PN/Y involving 2-hydroxy-1-naphthoic acid in a novel metabolic pathway. Process Biochemistry, 2008, 43(9): 1004–1008
Mallick S, Chatterjee S, Dutta T K. A novel degradation pathway in the assimilation of phenanthrene by Staphylococcus sp. strain PN/Y via meta-cleavage of 2-hydroxy-1-naphthoic acid: formation of trans-2,3-dioxo-5-(2′-hydroxyphenyl)-pent-4-enoic acid. Microbiology, 2007, 153(Pt 7): 2104–2115
Monna L, Omori T, Kodama T. Microbial degradation of dibenzofuran, fluorene, and dibenzo-p-dioxin by Staphylococcus auriculans DBF63. Applied and Environmental Microbiology, 1993, 59(1): 285–289
Chen H, Hopper S L, Cerniglia C E. Biochemical and molecular characterization of an azoreductase from Staphylococcus aureus, a tetrameric NADPH-dependent flavoprotein. Microbiology, 2005, 151(Pt 5): 1433–1441
Elisangela F, Andrea Z, Fabio D G, Cristiano R M, Regina D L, Artur C P. Biodegradation of textile azo dyes by a facultative Staphylococcus arlettae strain VN-11 using a sequential microaerophilic/aerobic process. International Biodeterioration and Biodegradation, 2009, 63(3): 280–288
Ayed L, Chaieb K, Cheref A, Bakhrouf A. Biodegradation and decolorization of triphenylmethane dyes by Staphylococcus epidermidis. Desalination, 2010, 260(1–3): 137–146
Uddin M S, Zhou J, Qu Y, Guo J, Wang P, Zhao L. Biodecolorization of azo dye acid red B under high salinity condition. Bulletin of Environmental Contamination and Toxicology, 2007, 79(4): 440–444
Thompson J D, Gibson T J, Plewniak F, Jeanmougin F, Higgins D G. The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Research, 1997, 25(24): 4876–4882
Salokhe M D, Govindwar S P. Effect of carbon source on the biotransformation enzymes in Serratia marcescens. World Journal of Microbiology and Biotechnology, 1999, 15(2): 229–232
Bradford M M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 1976, 72(1–2): 248–254
Liu G, Wang J, Lu H, Jin R, Zhou J, Zhang L. Effects of reduction products of ortho-hydroxyl substituted azo dyes on biodecolorization of azo dyes. Journal of Hazardous Materials, 2009, 171(1–3): 222–229
Wang X, Cheng X, Sun D. Autocatalysis in reactive black 5 biodecolorization by Rhodopseudomonas palustris W1. Applied Microbiology and Biotechnology, 2008, 80(5): 907–915
Kostick D S. The material flow concept for materials. Natural Resources Research, 1996, 5(4): 211–233
Khalid A, Arshad M, Crowley D E. Decolorization of azo dyes by Shewanella sp. under saline conditions. Applied Microbiology and Biotechnology, 2008, 79(6): 1053–1059
Amoozegar M A, Hajighasemi M, Hamedi J, Asad S, Ventosa A. Azo dye decolorization by halophilic and halotolerant microorganisms. Annals of Microbiology, 2010, 61(2): 217–230
Asad S, Amoozegar M A, Pourbabaee A A, Sarbolouki M N, Dastgheib S M M. Decolorization of textile azo dyes by newly isolated halophilic and halotolerant bacteria. Bioresource Technology, 2007, 98(11): 2082–2088
Guo J, Zhou J, Wang D, Tian C, Wang P, Uddin M S. A novel moderately halophilic bacterium for decolorizing azo dye under high salt condition. Biodegradation, 2008, 19(1): 15–19
Dawkar V V, Jadhav U U, Ghodake G S, Govindwar S P. Effect of inducers on the decolorization and biodegradation of textile azo dye Navy blue 2GL by Bacillus sp. VUS. Biodegradation, 2009, 20(6): 777–787
Gomare S S, Govindwar S P. Brevibacillus laterosporus MTCC 2298: a potential azo dye degrader. Journal of Applied Microbiology, 2009, 106(3): 993–1004
Yang Y Y, Du L N, Wang G, Jia XM, Zhao Y H. The decolorisation capacity and mechanism of Shewanella oneidensis MR-1 for methyl orange and acid yellow 199 under microaerophilic conditions.Water Science and Technology, 2011, 63(5): 956–963
Saratale R G, Saratale G D, Chang J S, Govindwar S P. Decolorization and biodegradation of reactive dyes and dye wastewater by a developed bacterial consortium. Biodegradation, 2010, 21(6): 999–1015
dos Santos A B, Cervantes F J, van Lier J B. Review paper on current technologies for decolourisation of textile wastewaters: perspectives for anaerobic biotechnology. Bioresource Technology, 2007, 98(12): 2369–2385
Saratale R G, Saratale G D, Chang J S, Govindwar S P. Bacterial decolorization and degradation of azo dyes: a review. Journal of the Taiwan Institute of Chemical Engineers, 2011, 42(1): 138–157
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yan, B., Du, C., Xu, M. et al. Decolorization of azo dyes by a salt-tolerant Staphylococcus cohnii strain isolated from textile wastewater. Front. Environ. Sci. Eng. 6, 806–814 (2012). https://doi.org/10.1007/s11783-012-0453-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11783-012-0453-4