Abstract
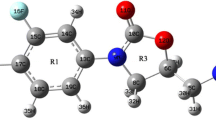
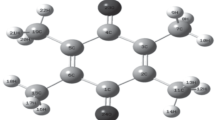
Fourier transform infrared (FT-IR) and Raman (FT-Ra) spectra of 1-(4-methoxyphenyl)-1H-imidazole were recorded and analyzed. The vibrational wavenumbers of the structure were computed by using the density functional theory (DFT)/Becke three Lee–Yang–Parr (B3LYP)/6–311 ++ g(d,p) basis set. The highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) energy values were determined to be − 1 eV and − 6.27 eV, respectively. The HOMO–LUMO energy gap value was found to be 5.27 eV. A large HOMO–LUMO gap means low reactivity in chemical reactions, indicating the high stability of the molecule. The HOMO–LUMO energy gap for the existing structure is relatively high, as a result the title molecule is hard. 1H, 13C NMR, and UV–Vis experimental and theoretical spectral analyses were given. Molecular electrostatic potential, Fukui functions, and charge analysis were performed to predict electrophilic and nucleophilic regions or atoms. The nonlinear optical properties of the structure were investigated, and it was found that the structure showed an important nonlinear optical property. Basic thermodynamic properties such as entropy (S), enthalpy changes (ΔH), heat capacity (Cp), Gibbs free energy (G), and zero-point vibration energy were calculated under constant pressure in the gas phase for different temperature values. It was determined that this compound has a significant antibacterial effect against some bacterial strains. Also, to support the antibacterial activity studies of the title molecule, molecular docking studies were carried out with protein structures of some microorganisms used in the antibacterial activity studies.













Similar content being viewed by others
References
Amul B, Muthu S, Raja M, Sevvanthi S (2019) Spectral, DFT and molecular docking investigations on Etodolac. J Mol Struc 1195:747–761. https://doi.org/10.1016/j.molstruc.2019.06.047
Asif M, Imran M (2020) Effect of Quorum sensing inhibitor agents against Pseudomonas aeruginosa. Russ J Bioorgan Chem 46:194–164. https://doi.org/10.1134/S1068162020020041
Ayers PW, Parr RG (2000) Variational principles for describing chemical reactions: the fukui function and chemical hardness revisited. J Am Chem Soc 122:2010–2018. https://doi.org/10.1021/ja9924039
Biovia (2021) Visualization. https://www.3ds.com/products-services/biovia/products/molecular-modeling-simulation/biovia-discovery-studio/visualization/. Accessed 01 October 2021.
Boiani M, González M (2005) Imidazole and benzimidazole derivatives as chemotherapeutic agents. Mini-Reviews Med Chem 5:409–424. https://doi.org/10.2174/1389557053544047
Brogden RN, Heel RC, Speight TM, Avery GS (1978) Metronidazole in anaerobic infections: a review of its activity. Pharmacokinet Ther Use Drugs 16(5):387–417. https://doi.org/10.2165/00003495-197816050-00002
Büyükmurat Y, Akyüz S (2001) Theoretical and experimental IR spectra and assignments of 3-aminopyridine. J Mol Struct 563:545–550. https://doi.org/10.1016/S0022-2860(00)00801-2
CLSI Document M100-S17 (2007) Performance standards for antimicrobial susceptibility testing 17th Informational Supplement, Clinical and Laboratory Standards Institute, Wayne, Pennsylvania: CLSI, Table 1, p. 26
Çelik S, Alp M, Yurdakul S (2020) A combined experimental and theoretical study on vibrational spectra of 3-pyridyl methyl ketone. Spectrosc Lett 53(4):234–248. https://doi.org/10.1080/00387010.2020.1734840
Çelik S, Badoğlu S, Yurdakul S (2017) Vibrational spectroscopic and density functional study on 1,2,4- triazolo-[1,5-a]pyrimidine. Vibrat Spect 92:20–26. https://doi.org/10.1016/j.vibspec.2017.08.011
Demircioglu Z, Kastas CA, Büyükgüngör O (2015) The spectroscopic (FT-IR, UV-Vis), Fukui Function, NLO, NBO, NPA and Tautomerism Effect Analysis of (E)-2-[(2-hydroxy-6- methoxybenzylidene)amino]benzonitrile. Spectrochim Acta A 139:539–548. https://doi.org/10.1016/j.saa.2014.11.078
Ebrahimi H, Hadi JS, Al-Ansari HS (2013) A new series of Schiff bases derived from sulfa drugs and indole-3-carboxaldehyde: synthesis, characterization, spectral and DFT computational studies. J Mol Struct 1039:37–45. https://doi.org/10.1016/j.molstruc.2013.01.063
Erdogdu Y, Manimaran D, Güllüoglu MT, Amalanathan M, Joeb IH, Yurdakul S (2013) FT-IR, FT-Raman, NMR Spectra and DFT Simulations of 4-(4-Fluoro-phenyl)-1H-imidazole. Opt Spect 114(4):525–536. https://doi.org/10.7868/S0030403413040077
Erdoğdu Y, Unsalan O, Sajan D, Gulluoglu MT (2010) Structural conformations and vibrational spectral study of chloroflavone with density functional theoretical simulations. Spectrochim Acta A 76:130–136. https://doi.org/10.1016/j.saa.2010.02.043
Eryılmaz S, Akdemir N, İnkaya E (2016) The examination of molecular structure properties of 4,4′-oxydiphthalonitrile compound: combined spectral and computational analysis approaches. Spectrosc Lett 52:28–42. https://doi.org/10.1080/00387010.2018.1544569
Fınkelstein W, Isselbacher KJ (1978) Medical Intelligence. New England J Med 2:992–996
Gece G, Bilgiç S (2012) Molecular-level understanding of the inhibition efficiency of some inhibitors of zinc corrosion by quantum chemical approach. Ind Eng Chem Res 51:14115–14120. https://doi.org/10.1021/ie302324b
Govindarajan M, Ganasan K, Periandy S, Karabacak M, Mohan S (2015) Vibrational spectroscopic analysis of 2-chlorotoluene and 2-bromotoluene: A combined experimental and theoretical study. Spectrochim Acta A Mol Biomol Spect. https://doi.org/10.1016/j.saa.2010.08.038
Gunduz SK, Bicak B, Celik S, Akyuz S, Ozel AE (2017) Structural and spectroscopic investigation on antioxidant dipeptide, l-Methionyl-l-Serine: A combined experimental and DFT study. J Mol Struct. https://doi.org/10.1016/j.molstruc.2017.02.075
Güllüoglu MT, Erdogdu Y, Karpagam J, Sundaraganesan N, Yurdakul S (2011) DFT, FT-Raman, FT-IR and FT-NMR studies of 4-phenylimidazole. J Mol Struct 990(1–3):14–20. https://doi.org/10.1016/j.molstruc.2011.01.001
Hakiria R, Ameurb I, Abidb S, Derbela N (2018) Synthesis, X-Ray Structural, Hirshfeld surface analysis, FTIR, MEP and NBO analysis using DFT study of a 4-chlorobenzylammonium nitrate (C7ClH9N)+(NO3)-. J Mol Struct 1164:486–492. https://doi.org/10.1016/j.molstruc.2018.03.068
Haruna K, Kumar VS, Maray YS, Popoola SA, Thomas R, Roxy MS, Al-Saadi AA (2019) Conformational profile, vibrational assignments, NLO properties and molecular docking of biologically active herbicide1,1-dimethyl-3-phenylurea. Heliyon 5:e01987. https://doi.org/10.1016/j.heliyon.2019.e01987
Jain AK, Ravichandran V, Sisodiya M, Agrawal RK (2010) Syntthesis and antibacterial evaluation of 2-substituted-4,5-diphenylN-alkyl imidazole derivatives. Asian Pac J Trop Med. https://doi.org/10.1016/S1995-7645(10)60113-7
Junchao L, Han H, Zhang X, Li S, Ge S, Zhang G, Gao T (2018) First-principles study the structural, electronic, vibrational and thermodynamic properties of Zr1-xHfxCoH3. Int J Hydrog Energy 43(41):19152–19163. https://doi.org/10.1016/j.ijhydene.2018.08.130
Kumar S, Radha A, Kour M, Kumar R, Chouaih A, Pandey SK (2019) DFT studies of disubstituted diphenyldithiophosphates of nickel(II): Structural and some spectral parameters. J Mol Struct 1185:212–218. https://doi.org/10.1016/j.molstruc.2019.02.105
Kumar S, Saini V, Maurya IK, Sindhu J, Kumari M, Kataria R, Kumar V (2018) Design, synthesis, DFT, docking studies and ADME prediction of some new coumarinyl linked pyrazolylthiazoles: Potential standalone or adjuvant antimicrobial agents. Ploes One 13(4):0196016. https://doi.org/10.1371/journal.pone.0196016
Leszczynski J, Papadopoulos MG, Sadlej AJ (2006) Non-linear optical properties of matter. Springer, Dordrecht, The Netherlands
Lu X, Liu X, Wan B, Franzblau SG, Chen L, Zhou C, You, (2012) Synthesis and evaluation of anti-tubercular and antibacterial activities of new 4-(2,6-dichlorobenzyloxy)phenyl thiazole, oxazole and imidazole derivatives, Part 2. Eur J Med Chem 49:164–171. https://doi.org/10.1016/j.ejmech.2012.01.007
McLean KH, Winson MK, Fish L, Taylor A, Chhabra SR, Camara M, Daykin M, Lamb JH, Swift S, Bycroft BW, Stewart GS, Williams P (1997) Quorum sensing and Chromobacterium violaceum: exploitation of violacein production and inhibition for the detection of N-acyl homoserine lactones. Microbiology 143(12):3703–3711. https://doi.org/10.1099/00221287-143-12-3703
Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ (2009) AutoDock4 and AutoDockTools4: automated docking with selective receptorflex-ibility. J Comput Chem 30:2785–2791. https://doi.org/10.1002/jcc.21256
Mumita MA, Pala TK, Alam MA, Islama AAM, Paulb S, Sheikh MC (2020) DFT studies on vibrational and electronic spectra, HOMO–LUMO, MEP, HOMA, NBO and molecular docking analysis of benzyl-3N-(2,4,5-trimethoxyphenylmethylene) hydrazinecarbodithioate. J Mol Struct 1220:128715. https://doi.org/10.1016/j.molstruc.2020.128715
Narasimhan B, Sharma D, Kumar P (2011) Biological importance of imidazole nucleus in the new millennium. Med Chem Res 20:1119–1140. https://doi.org/10.1007/s00044-010-9472-5
Ozkay Y, Isıkdag I, Incesu Z, Akalın G (2010) Synthesis of Synthesis of 2-subsituted-N-[4-(1-methyl-4,5-diphenyl-1H-imidazole-2-yl)phenyl]acetamide derivatives and evalutaion of their anticancer activity. Eur J Med Chem 45:3320–3328. https://doi.org/10.1016/j.ejmech.2010.04.015
Padmavathi V, Kumari CP, Venkatesh BC, Padmaja A (2011) Synthesis and antimicrobial activity of amido linked pyrrolyl and pyrazolyl-oxazoles, thiazoles and imidazoles. Eur J Med Chem 46:5317–5326. https://doi.org/10.1016/j.ejmech.2011.08.032
Pandey J, Tiwari VK, Verma SS, Chaturvedi V, Bhatnagar S, Sinha S, Gaikwad AN, Tripathi RP (2009) Synthesis and antitubercular screening of imidazole derivatives. Eur J Med Chem 44:3350–3355. https://doi.org/10.1016/j.ejmech.2009.02.013
Petrus EM, Tinakumari S, Chai LC, Ubong A, Tunung R, Elexson N, Chai LF, Son R (2011) A study on the minimum inhibitory concentration and minimum bactericidal concentration of Nano Colloidal Silver on food-borne pathogens. Int Food Res 18:55–66
Pilepic V, Urslic S (2001) Nucleophilic reactivity of the nitroso group. Fukui function DFT calculations for nitrosobenzene and 2-methyl-2-nitrosopropane. J Mol Struct (theochem) 539:41–49. https://doi.org/10.1016/S0166-1280(00)00642-4
Ramanathan P (2017) Synthesis, Spectral Characterization and Biological Studies of 2-(4-Methoxynaphthalen-1-Yl)-1-(4-Methoxyphenyl)-1H-Phenanthro[9,10-d] imidazole. Mod Chem Appl. https://doi.org/10.4172/2329-6798.1000242
Ravindranath L, Reddy BV (2020) Theoretical and experimental study of torsional potentials, molecular structure (monomer and dimer), vibrational analysis and molecular characteristics of some dimethyl bipyridines. J Mol Struct 1200(36):127089. https://doi.org/10.1016/j.molstruc.2019.127089
RSCB PDB Protein Data Bank. https://www.rcsb.org/. Accessed 01 October 2021.
Saalem H, Krishnan AR, Erdogdu Y, Subashchandrabose S, Thanikachalam V, Manikandan G (2011) Density functional theory studies on 2,5-bis(4-hydroxy-3-methoxybenzylidene)-cyclopentanone. J Mol Struct 999:2–9. https://doi.org/10.1016/j.molstruc.2011.02.039
Saravanan RR, Seshadri S, Gunasekaran S, Mendoza RM, Granda SG (2015) Conformational analysis, X-ray crystallographic, FT-IR, FT-Raman, DFT, MEP and molecular docking studies on 1-(1-(3-methoxyphenyl) ethylidene) thiosemicarbazide. Spectrochim Acta a: Mol Biomol Spect 139:321–328. https://doi.org/10.1016/j.saa.2014.12.026
Sarıkaya EK, Dereli O (2013) Molecular structure and vibrational spectra of 7-Methoxy- 4-methylcoumarin by density functional method. J Mol Struct 1052:214–220. https://doi.org/10.1016/j.molstruc.2013.08.024
Senge MO, Fazekas M, Notaras EGA, Blau WJ, Zawadzka M, Locos OB, Mhuircheartaigh EMN (2007) Nonlinear optical properties of porphyrins. Adv Mater 19:2737–2774. https://doi.org/10.1002/adma.200601850
Sethi A, Singh RP, Shukla D, Singh P (2016) Synthesis of novel pregnane-diosgenin prodrugs via ring a and ring a connection: a combined experimental and theoretical studies. J Mol Struct 1125:616–623. https://doi.org/10.1016/j.molstruc.2016.07.020
Shalini K, Sharma PK, Kumar N (2010) Imidazole and its biological activities: a review. Pelagia Res Lib Der Chem Sinica 1(3):36–47
Sheeba BQ, Mary MSM, Amalanathan M, Job CB (2021) Structural and vibrational spectral investigation on the identification of Non-Linear Optical properties and wave function analyses (electrostatic potential, electron localisation function, localised orbital locator) of 3-Ethoxy Salicilaldehyde. Mol Simulat 47(15):1217–1233. https://doi.org/10.1080/08927022.2021.1962862
Sigma Aldrich Company (2006) Safety data sheet 1907. https://www.sigmaaldrich.com/TR/en/sds/aldrich/457612. Accessed 10 July 2021.
Singh P, Islam SS, Ahmad H, Prabaharan A (2018) Spectroscopic investigation (FT-IR, FT-Raman), HOMO-LUMO, NBO, and molecular docking analysis of N-ethyl-N-nitrosourea, a potential anticancer agent. J Mol Struct 1154:39–50. https://doi.org/10.1016/j.molstruc.2017.10.012
Spasov AR, Iezhitsa IN, Bugaeva LI, Anisimova VA (1999) Benzimidazole derivatives: Spectrum of pharmacological activity and toxicological properties (a review). Khim Farm Zhurn 33:6–17. https://doi.org/10.1007/BF02510042
Srivastava S, Gupta P, Sethi A, Pratap SR (2016) One pot synthesis of Curcumin-NSAIDs prodrug, spectroscopic characterization, conformational analysis, chemical reactivity, intramolecular interactions and first order hyperpolarizability by DFT method. J Mol Struct 1117:173–180. https://doi.org/10.1016/j.molstruc.2016.03.033
Stigliani JL, Genisson VB, Bernadou J, Pratviel G (2012) Cross-docking study on InhA inhibitors: a combination of Autodock Vina and PM6-DH2 simulations to retrieve bio-active conformations. Org Biomol Chem 10:6341–6349. https://doi.org/10.1039/c2ob25602a
Subashini K, Periandy S (2016) Spectroscopic (FT-IR, FT-Raman, UV, NMR, NBO) investigation and molecular docking study of (R)-2-Amino-1-PhenylEthanol. J Mol Struct 1117:240–256. https://doi.org/10.1016/j.molstruc.2016.03.063
Thomas R, Hossain M, Mary YS, Resmi KS, Armakovic S, Armakovic SJ, Nanda AK, Ranjan VK, Vijayakumar G, Alsenoy CV (2018) Spectroscopic analysis and molecular docking of imidazole derivatives and investigation of its reactive properties by DFT and molecular dynamics simulations. J Mol Struct 1158:156–175. https://doi.org/10.1016/j.molstruc.2018.01.021
Trott O, Olson AJ (2009) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31(2):455–461. https://doi.org/10.1002/jcc.21334
Uzun S, Esen Z, Koç E, Usta NC, Ceylan M (2019) Experimental and density functional theory (MEP, FMO, NLO, Fukui functions) and antibacterial activity studies on 2-amino-4-(4-nitrophenyl) -5,6-dihydrobenzo [h] quinoline-3-carbonitrile. J Mol Struct 1178:450–457. https://doi.org/10.1016/j.molstruc.2018.10.001
Verma A, Joshi S, Singh D (2013) Imidazole: Having Versatile Biological Activities. J Chemist. https://doi.org/10.1155/2013/329412
Vijesh AM, Isloor AM, Telkar S, Peethambar SK, Rai S, Isloor N (2011) Synthesis, characterization and antimicrobial studies of some new pyrazole incorporated imidazole derivatives. Eur J Med Chem 46:3531–3556. https://doi.org/10.1016/j.ejmech.2011.05.005
Yamijala SSRKC, Mukhopadhyay M, Pati SK (2015) Linear and nonlinear optical properties of graphene quantum dots: a computational study. J Phys Chem C 119:12079–12087. https://doi.org/10.1021/acs.jpcc.5b03531
Yazıcı S, Albayrak Ç, Gümrükçüoğlu İ, Şenel İ, Büyükgüngör O (2011) Experimental and density functional theory (DFT) studies on (E)-2-Acetyl-4-(4-nitrophenyldiazenyl) phenol. J Mol Struct 985(2–3):292–298. https://doi.org/10.1016/j.molstruc.2010.11.009
Yurdakul S, Temel E, Buyukgungor O (2019) Crystal structure, spectroscopic characterization, thermal properties and theoretical investigations on [Ag(methyl 4-pyridylketone)2NO3]. J Mol Struct 1191:301–313. https://doi.org/10.1016/j.molstruc.2019.04.071
Zampieri D, Mamolo MG, Laurini E, Vioa SG, L, (2008) Antifungal and antimycobacterial activity of 1-(3,5-diaryl-4,5- dihydro-1H-pyrazol-4-yl)-1H-imidazole derivatives. Bioorgan Med Chem 16:4516–4522. https://doi.org/10.1016/j.bmc.2008.02.055
Acknowledgements
We would like to thank to our reviewers for their comments that help improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kucuk, C., Yurdakul, S. & Erdem, B. Experimental and theoretical Fourier transform infrared and Raman spectroscopy, density functional theory, antibacterial activity and molecular docking studies on 1-(4-methoxyphenyl)-1H-imidazole. Chem. Pap. 76, 2833–2854 (2022). https://doi.org/10.1007/s11696-021-02017-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-021-02017-8