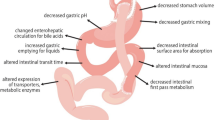
Abstract
Purpose
Bariatric surgery alters the anatomic and physiological structure of the gastrointestinal tract, predisposing patients to the malabsorption of nutrients. The purpose of this study was to determine the prevalence and determinants of secondary hyperparathyroidism (SHPT) in the patients undergoing either one-anastomosis gastric bypass (OAGB) or sleeve gastrectomy (SG).
Materials and Methods
A total of 517 patients (without SHPT at the baseline) who had undergone OAGB or SG were prospectively assessed 1 year after the surgery. Anthropometric parameters, calcium, intact parathyroid hormone (iPTH), and 25(OH)D levels were compared according to the surgery type before and 1 year after surgery. Multiple logistic regression models were used to evaluate possible SHPT predictors after bariatric surgery.
Results
The overall prevalence of SHPT was 12.6% after surgery, significantly different between the OAGB and SG groups (17.1 vs. 9.9%, respectively). The serum levels of albumin-corrected calcium and 25(OH)D were not significantly different between the two groups. The patients undergoing OAGB had significantly higher serum levels of ALP (198.2 vs. 156.6) compared to the subjects undergoing SG. Higher iPTH levels preoperatively, lower 1-year excess weight loss%, and OAGB surgery seemed to be independent predictors for SHPT 1 year after surgery.
Conclusion
Morbidly-obese patients undergoing OAGB had a higher risk of SHPT than their counterparts undergoing SG, whereas 25(OH)D deficiency and calcium levels did not differ between the two groups. The OAGB procedure, preoperative iPTH levels, and 1-year weight loss were predictors of postoperative SHPT development.
Graphical Abstract



Similar content being viewed by others
Data Availability
The datasets used and analyzed in the current study are available from the corresponding author on reasonable request.
References
Jamal SA, Miller PD. Secondary and tertiary hyperparathyroidism. J Clin Densitom: Official J Int Soc Clin Densitom. 2013;16(1):64–8.
Chandran M, Wong J. Secondary and tertiary hyperparathyroidism in chronic kidney disease: an endocrine and renal perspective. Indian J Endocrinol Metab. 2019;23(4):391–9.
Bargagli M, Arena M, Naticchia A, et al. The role of diet in bone and mineral metabolism and secondary hyperparathyroidism. Nutrients. 2021;13(7):2328.
Pourshahidi LK. Vitamin D and obesity: current perspectives and future directions. Proc Nutr Soc. 2015;74(2):115–24.
Kim J, Nimeri A, Khorgami Z, et al. Metabolic bone changes after bariatric surgery: 2020 update, American Society for Metabolic and Bariatric Surgery Clinical Issues Committee position statement. Surg Obes Relat Dis : Off J Am Soc Bariatr Surg. 2021;17(1):1–8.
Chakhtoura MT, Nakhoul NN, Shawwa K, et al. Hypovitaminosis D in bariatric surgery: a systematic review of observational studies. Metab Clin Exp. 2016;65(4):574–85.
Tian Z, Fan XT, Li SZ, et al. Changes in bone metabolism after sleeve gastrectomy versus gastric bypass: a meta-analysis. Obes Surg. 2020;30(1):77–86.
Coupaye M, Rivière P, Breuil MC, et al. Comparison of nutritional status during the first year after sleeve gastrectomy and Roux-en-Y gastric bypass. Obes Surg. 2014;24(2):276–83.
Vix M, Liu KH, Diana M, et al. Impact of Roux-en-Y gastric bypass versus sleeve gastrectomy on vitamin D metabolism: short-term results from a prospective randomized clinical trial. Surg Endosc. 2014;28(3):821–6.
Salman MA, Salman A, Elewa A, et al. Secondary hyperparathyroidism before and after bariatric surgery: a prospective study with 2-year follow-up. Obes Surg. 2022;32(4):1141–8.
Wei J-H, Lee W-J, Chong K, et al. High incidence of secondary hyperparathyroidism in bariatric patients: comparing different procedures. Obes Surg. 2018;28(3):798–804.
Alexandrou A, Tsoka E, Armeni E, et al. Determinants of secondary hyperparathyroidism in bariatric patients after Roux-en-Y gastric bypass or sleeve gastrectomy: a pilot study. Int J Endocrinol. 2015;2015:984935.
Hewitt S, Aasheim ET, Søvik TT, et al. Relationships of serum 25-hydroxyvitamin D, ionized calcium and parathyroid hormone after obesity surgery. Clin Endocrinol. 2018;88(3):372–9.
Toelle P, Peterli R, Zobel I, et al. Risk factors for secondary hyperparathyroidism after bariatric surgery: a comparison of 4 different operations and of vitamin D-receptor-polymorphism. Exp Clin Endocrinol Diabetes : Off J Ger Soc Endocrinol Ger Diabetes Assoc. 2012;120(10):629–34.
Mendonça FM, Neves JS, Silva MM, et al. Secondary hyperparathyroidism among bariatric patients: unraveling the prevalence of an overlooked foe. Obes Surg. 2021;31(8):3768–75.
Barzin M, Khalaj A, Motamedi MA, et al. Safety and effectiveness of sleeve gastrectomy versus gastric bypass: one-year results of Tehran Obesity Treatment Study (TOTS). Gastroenterol Hepatol Bed Bench. 2016;9(Suppl1):S62–9.
Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911–30.
Brethauer SA, Kim J, el Chaar M, et al. Standardized outcomes reporting in metabolic and bariatric surgery. Surg Obes Relat Dis : Off J Am Soc Bariatr Surg. 2015;11(3):489–506.
Liu C, Wu D, Zhang JF, et al. Changes in Bone metabolism in morbidly obese patients after bariatric surgery: a meta-analysis. Obes Surg. 2016;26(1):91–7.
Li Z, Zhou X, Fu W. Vitamin D supplementation for the prevention of vitamin D deficiency after bariatric surgery: a systematic review and meta-analysis. Eur J Clin Nutr. 2018;72(8):1061–70.
Jl C, Picot J, Loveman E, et al. Surgery for obesity. Cochrane Database Syst Rev. 2009;15(2).
Billeter AT, Fischer L, Wekerle AL, et al. Malabsorption as a therapeutic approach in bariatric surgery. Viszeralmedizin. 2014;30(3):198–204.
Schafer AL, Weaver CM, Black DM, et al. Intestinal calcium absorption decreases dramatically after gastric bypass surgery despite optimization of vitamin D status. J Bone Miner Res : Off J Am Soc Bone Miner Res. 2015;30(8):1377–85.
de Holanda NCP, Baad VMA, Bezerra LR, et al. Secondary hyperparathyroidism, bone density, and bone turnover after bariatric surgery: differences between Roux-en-Y gastric bypass and sleeve gastrectomy. Obes Surg. 2021;31(12):5367–75.
Salazar DA, Ferreira MJS, Neves JS, et al. Variable thresholds of vitamin D plasma levels to suppress PTH: the effect of weight and bariatric surgery. Obes Surg. 2020;30(4):1551-9.
Barzin M, Ebadinejad A, Vahidi F, et al. The mediating role of bariatric surgery in the metabolic relationship between parathyroid hormone and 25-hydroxyvitamin D. Osteoporos Int. 2022.
Spatz JM, Ellman R, Cloutier AM, et al. Sclerostin antibody inhibits skeletal deterioration due to reduced mechanical loading. J Bone Miner Res : Off J Am Soc Bone Miner Res. 2013;28(4):865–74.
Zerwekh JE, Ruml LA, Gottschalk F, et al. The effects of twelve weeks of bed rest on bone histology, biochemical markers of bone turnover, and calcium homeostasis in eleven normal subjects. J Bone Miner Res : Off J Am Soc Bone Miner Res. 1998;13(10):1594–601.
Paccou J, Thuillier D, Courtalin M, et al. A comparison of changes in bone turnover markers after gastric bypass and sleeve gastrectomy, and their association with markers of interest. Surg Obes Relat Dis : Off J Am Soc Bariatr Surg. 2022;18(3):373–83.
Acknowledgements
The authors would like to thank the hospital staff, assistants, and coordinators who took part in this research. Also, special thanks to Mohammadreza Golsibi for his assistance and support of the electronic data collection system. This article was derived from the disease registry entitled “Registration of patients in Tehran Obesity Treatment Center” and approved under the ethical code of “IR.SBMU.ENDOCRINE.REC1397.059” (date: 2018-05-08) by the local ethics committee. The study was supported by the deputy of research and technology of Shahid Beheshti University of Medical Sciences (http://dregistry.sbmu.ac.ir).
Author information
Authors and Affiliations
Contributions
MB: study design, data collection, the coordination of patients’ issues and manuscript preparation, and the final approval of the manuscript. AE: study design, data collection, literature review, and manuscript preparation. AK: study design, performing surgical operations, and the final approval of the manuscript. MM: data analysis, interpretation, and manuscript preparation, MV: final approval of the manuscript. FH: study design, revising, and the final approval of the manuscript: All authors reviewed and approved the final draft of the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
All the procedures performed in the study were approved by Research Ethics Committee of the Research Institute for Endocrine Sciences of Shahid Beheshti University of Medical Sciences (IR.SBMU.ENDOCRINE.REC.1401.055) and were in accordance with the ethical standards of the institutional Human Research Review Committee and the 1964 Helsinki Declaration and its later amendments.
Consent to Participate
Informed written consent was obtained from all individual participants included in the study.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Points
• Postoperative SHPT was 9.9% and 17.1% in the SG and OAGB.
• The patients undergoing OAGB had significantly higher serum levels of alkaline phosphatase.
• Calcium and 25(OH)D levels did not differ between the two groups.
• Individuals with a higher preoperative iPTH level and lower 1-year EWL% undergoing OAGB were more likely to develop SHPT.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Barzin, M., Ebadinejad, A., Khalaj, A. et al. Determinants of Secondary Hyperparathyroidism 1 Year After One-Anastomosis Gastric Bypass or Sleeve Gastrectomy. OBES SURG 33, 156–163 (2023). https://doi.org/10.1007/s11695-022-06337-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-022-06337-w