Abstract
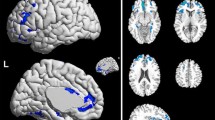
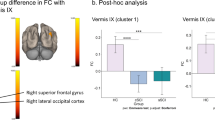
Brachial plexus injury (BPI) is a type of severe peripheral nerve trauma that leads to central remodeling in the brain, as revealed by functional MRI analysis. However, previously reported remodeling is mostly restricted to sensorimotor areas of the brain. Whether this disturbance in the sensorimotor network leads to larger-scale functional remodeling remains unknown. We sought to explore the higher-level brain functional abnormality pattern of BPI patients from a large-scale network function connectivity dimension in 15 right-handed BPI patients. Resting-state functional MRI data were collected and analyzed using independent component analysis methods. Five components of interest were recognized and compared between patients and healthy subjects. Patients showed significantly altered brain local functional activities in the bilateral fronto-parietal network (FPN), sensorimotor network (SMN), and executive-control network (ECN) compared with healthy subjects. Moreover, functional connectivity between SMN and ECN were significantly less in patients compared with healthy subjects, and connectivity strength between ECN and SMN was negatively correlated with patients’ residual function of the affected limb. Functional connectivity between SMN and right FPN were also significantly less than in controls, although connectivity between ECN and default mode network (DMN) was greater than in controls. These data suggested that brain functional disturbance in BPI patients extends beyond the sensorimotor network and cascades serial remodeling in the brain, which significantly correlates with residual hand function of the paralyzed limb. Furthermore, functional remodeling in these higher-level functional networks may lead to cognitive alterations in complex tasks.




Similar content being viewed by others
References
Allen, E. A., Erhardt, E. B., Wei, Y., Eichele, T., & Calhoun, V. D. (2012). Capturing inter-subject variability with group independent component analysis of fMRI data: a simulation study. NeuroImage, 59, 4141–4159.
Auer, T., Pinter, S., Kovacs, N., Kalmar, Z., Nagy, F., Horvath, R. A., Koszo, B., Kotek, G., Perlaki, G., Koves, M., Kalman, B., Komoly, S., Schwarcz, A., Woermann, F. G., & Janszky, J. (2009). Does obstetric brachial plexus injury influence speech dominance? Annals of Neurology, 65, 57–66.
Bell, A. J., & Sejnowski, T. J. (1995). An information-maximization approach to blind separation and blind deconvolution. Neural Computation, 7, 1129–1159.
Borst, J. P., & Anderson, J. R. (2013). Using model-based functional MRI to locate working memory updates and declarative memory retrievals in the fronto-parietal network. Proceedings of the National Academy of Sciences of the United States of America, 110, 1628–1633.
Braver, T. S., Cohen, J. D., Nystrom, L. E., Jonides, J., Smith, E. E., & Noll, D. C. (1997). A parametric study of prefrontal cortex involvement in human working memory. NeuroImage, 5, 49–62.
Calhoun, V. D., Adali, T., Pearlson, G. D., & Pekar, J. J. (2001). A method for making group inferences from functional MRI data using independent component analysis. Human Brain Mapping, 14, 140–151.
Calhoun, V. D., Kiehl, K. A., Liddle, P. F., & Pearlson, G. D. (2004). Aberrant localization of synchronous hemodynamic activity in auditory cortex reliably characterizes schizophrenia. Biological Psychiatry, 55, 842–849.
Damoiseaux, J. S., Rombouts, S. A., Barkhof, F., Scheltens, P., Stam, C. J., Smith, S. M., & Beckmann, C. F. (2006). Consistent resting-state networks across healthy subjects. Proceedings of the National Academy of Sciences of the United States of America, 103, 13848–13853.
Dick, B. D., & Rashiq, S. (2007). Disruption of attention and working memory traces in individuals with chronic pain. Anesth Analg, 104, 1223–1229 tables of contents.
Feng, J. T., Liu, H. Q., Xu, J. G., Gu, Y. D., & Shen, Y. D. (2015). Differences in brain adaptive functional reorganization in right and left total brachial plexus injury patients. World Neurosurgery, 84, 702–708.
Ford, J. M., Johnson, M. B., Whitfield, S. L., Faustman, W. O., & Mathalon, D. H. (2005). Delayed hemodynamic responses in schizophrenia. NeuroImage, 26, 922–931.
Fox, M. D., Corbetta, M., Snyder, A. Z., Vincent, J. L., & Raichle, M. E. (2006). Spontaneous neuronal activity distinguishes human dorsal and ventral attention systems. Proceedings of the National Academy of Sciences of the United States of America, 103, 10046–10051.
Franco, A. R., Pritchard, A., Calhoun, V. D., & Mayer, A. R. (2009). Interrater and intermethod reliability of default mode network selection. Human Brain Mapping, 30, 2293–2303.
Greicius, M. D., Krasnow, B., Reiss, A. L., & Menon, V. (2003). Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proceedings of the National Academy of Sciences of the United States of America, 100, 253–258.
Greicius, M. D., Flores, B. H., Menon, V., Glover, G. H., Solvason, H. B., Kenna, H., Reiss, A. L., & Schatzberg, A. F. (2007). Resting-state functional connectivity in major depression: abnormally increased contributions from subgenual cingulate cortex and thalamus. Biological Psychiatry, 62, 429–437.
Hua, X. Y., Li, Z. Y., Xu, W. D., Zheng, M. X., Xu, J. G., & Gu, Y. D. (2012). Interhemispheric functional reorganization after cross nerve transfer: via cortical or subcortical connectivity? Brain Research, 1471, 93–101.
Jafri, M. J., Pearlson, G. D., Stevens, M., & Calhoun, V. D. (2008). A method for functional network connectivity among spatially independent resting-state components in schizophrenia. NeuroImage, 39, 1666–1681.
Kuiken, T. A., Miller, L. A., Lipschutz, R. D., Lock, B. A., Stubblefield, K., Marasco, P. D., Zhou, P., & Dumanian, G. A. (2007). Targeted reinnervation for enhanced prosthetic arm function in a woman with a proximal amputation: a case study. Lancet, 369, 371–380.
Laird, A. R., Fox, P. M., Eickhoff, S. B., Turner, J. A., Ray, K. L., McKay, D. R., Glahn, D. C., Beckmann, C. F., Smith, S. M., & Fox, P. T. (2011). Behavioral interpretations of intrinsic connectivity networks. Journal of Cognitive Neuroscience, 23, 4022–4037.
Liu, B., Li, T., Tang, W. J., Zhang, J. H., Sun, H. P., Xu, W. D., Liu, H. Q., & Feng, X. Y. (2013). Changes of inter-hemispheric functional connectivity between motor cortices after brachial plexuses injury: a resting-state fMRI study. Neuroscience, 243, 33–39.
Lundborg, G. (2000). Brain plasticity and hand surgery: an overview. Journal of Hand Surgery (British), 25, 242–252.
Makin, T. R., Filippini, N., Duff, E. P., Henderson Slater, D., Tracey, I., & Johansen-Berg, H. (2015). Network-level reorganisation of functional connectivity following arm amputation. NeuroImage, 114, 217–225.
Markov, N. T., Ercsey-Ravasz, M., Van Essen, D. C., Knoblauch, K., Toroczkai, Z., & Kennedy, H. (2013). Cortical high-density counterstream architectures. Science, 342, 1238406.
Ren, W. J., Liu, Y., Zhou, L. J., Li, W., Zhong, Y., Pang, R. P., Xin, W. J., Wei, X. H., Wang, J., Zhu, H. Q., Wu, C. Y., Qin, Z. H., Liu, G., & Liu, X. G. (2011). Peripheral nerve injury leads to working memory deficits and dysfunction of the hippocampus by upregulation of TNF-alpha in rodents. Neuropsychopharmacology, 36, 979–992.
Rizzolatti, G., & Matelli, M. (2003). Two different streams form the dorsal visual system: anatomy and functions. Experimental Brain Research, 153, 146–157.
Seeley, W. W., Menon, V., Schatzberg, A. F., Keller, J., Glover, G. H., Kenna, H., Reiss, A. L., & Greicius, M. D. (2007). Dissociable intrinsic connectivity networks for salience processing and executive control. The Journal of Neuroscience, 27, 2349–2356.
Smith, S. M., Fox, P. T., Miller, K. L., Glahn, D. C., Fox, P. M., Mackay, C. E., Filippini, N., Watkins, K. E., Toro, R., Laird, A. R., & Beckmann, C. F. (2009). Correspondence of the brain's functional architecture during activation and rest. Proceedings of the National Academy of Sciences of the United States of America, 106, 13040–13045.
Sridharan, D., Levitin, D. J., & Menon, V. (2008). A critical role for the right fronto-insular cortex in switching between central-executive and default-mode networks. Proceedings of the National Academy of Sciences of the United States of America, 105, 12569–12574.
Taylor, K. S., Anastakis, D. J., & Davis, K. D. (2009). Cutting your nerve changes your brain. Brain, 132, 3122–3133.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
New Cutting Edge Technology Joint Development Program of Shanghai Municipal Hospitals.
Conflict of Interest
Jun-Tao Feng, Han-Qiu Liu, Xu-Yun Hua, Yu-Dong Gu, Jian-Guang Xu, and Wen-Dong Xu declare no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Jun-Tao Feng and Han-Qiu Liu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Feng, JT., Liu, HQ., Hua, XY. et al. Brain functional network abnormality extends beyond the sensorimotor network in brachial plexus injury patients. Brain Imaging and Behavior 10, 1198–1205 (2016). https://doi.org/10.1007/s11682-015-9484-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-015-9484-3