Abstract
Objective
To explore the effects of bufalin on inhibiting proliferation, up-regulating methylation of Wilm’ tumor 1 gene (WT1) as well as its possible mechanisms in human erythroid leukemic (HEL) cells.
Methods
The HEL cells were treated with bufalin at various concentrations to observe cellular morphology, proliferation assay and cell cycle. The mRNA and protein expression levels of WT1 were detected by reverse transcription polymerase chain reaction (RT-PCR), Western blot and immunocytochemistry, DNA methylation of WT1 and protein expression levels of DNA methyltransferase 3a (DNMT3a) and DNMT3b were analyzed by methylation-specific PCR, and Western blot respectively.
Results
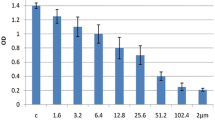
The bufalin was effective to inhibit proliferation of HEL cells in a dose-dependent manner, their suppression rates were from 23.4%±2.1% to 87.2%±5.4% with an half maximal inhibit concentration (IC50) of 0.046 μmol/L. Typical apoptosis morphology was observed in bufalin-treated HEL cells. The proliferation index of cell cycle decreased from 76.4%±1.9% to 49.7%±1.3%. The expression levels of WT1 mRNA and its protein reduced gradually with increasing doses of bufalin, meanwhile, the methylation status of WT1 gene changed from unmethylated into partially or totally methylated. While, the expression levels of DNMT3a and DNMT3b protein gradually increased by bufalin treatment in a dose-dependent manner.
Conclusions
Bufalin can not only significantly inhibit the proliferation of HEL cells and arrest cell cycle at G0/G1 phase, but also induce cellular apoptosis and down-regulate the expression level of WT1. Our results provide the evidence of bufalin for anti-leukemia, its mechanism may involve in increasing WT1 methylation status which is related to the up-regulation of DNMT3a and DNMT3b proteins in erythroid leukemic HEL cells.
Similar content being viewed by others
References
Zhang L, Nakaya K, Yoshida T, Kuroiwa Y. Induction by bufalin of differentiation of human leukemia cells HL60, U937, and ML1 toward macrophage/monocyte-like cells and its potent synergistic effect on the differentiation of human leukemia cells in combination with other inducers. Cancer Res 1992;52:4634–4641.
Ke QH, Guang H, Wei G, Yong HS, Xue QH, Chang QL. Anti-tumor activities and apoptosis-regulated mechanisms of bufalin on the orthotopic transplantation tumor model of human hepatocellular carcinoma in nude mice. World J Gastroenterol 2007;13:3374–3379.
Hong C, Shibayama-Imazu T, Masudai Y, Shinki T, Nakajo S, Nakaya K. Involvement of Tiam1 in apoptosis induced by bufalin in HeLa cells. Anticancer Res 2007;27:245–250.
Heppe RK, Beckwith JB. Nephrogenic rests in Wilms tumor patients with the Drash syndrome. J Urol 1991;145:1225–1228.
Beckwith JB, Kiviat NB, Bonadio JF. Nephrogenic rests, nephroblastomatosis, and the pathogenesis of Wilms’ tumor. Pediatr Pathol 1990;10(1–2):1–36.
Liu Y, Qu X, Wang P, Tian X, Luo Y, Liu S, et al. WT1 downregulation during K562 cell differentiation and apoptosis induced by bufalin. Chin J Hematol (Chin) 2002;23:356–359.
Gao RL, Chen XH, Lin XJ, Qian XD, Xu WH, Chong BH. Effects of notoginosides on proliferation and up-regulation of GR nuclear tanscription factor in hematopoietic cells. Acta Pharmacol Sin 2007;28:703–707.
Sun X, Gao RL, Lin XJ, Xu WH, Chen XH. Panax notoginseng saponins induced up-regulation, phosphorylation and binding activity of MEK, ERK, AKT, PI-3K protein kinases and GATA transcription factors in hematopoietic cells. Chin J Integr Med 2013;19:112–118.
Li LJ, Xu NW, Gao RL, Lin XJ, Qiu HY, Liu WH, et al. Effects of Danshen Injection on inhibiting proliferation and inducing apoptosis through down-regulation of mutant JAK2 gene and its protein phosphorylation in human erythroid leukemic cells. Chin J Integr Med 2014;20:381–386.
Yamada K, Hino K, Tomoyasu S, Honma Y, Tsuruoka N. Enhancement by bufalin of retinoic acid-induced differentiation of acute promyelocytic leukemia cells in primary culture. Leuk Res 1998;22:589–595.
Menssen HD, Renkl HJ, Rodeck U, Kari C, Schwartz S, Thiel E. Detection by monoclonal antibodies of the Wilms tumor (WT1) nuclear protein in patients with acute leukemia. Int J Cancer 1997;70:518–523.
Baird PN, Simmons PJ. Expression of the Wilms tumor gene (WT1) in normal hemopoiesis. Exp Hematol 1997;25:312–320.
Yamagami T, Sugiyama H, Inoue K, Ogawa H, Tatekawa T, Hirata M, et al. Growth inhibition of human leukemic cells by WT1 (Wilms tumor gene) antisense oligodeoxynucleotides: implications for the involvement of WT1 in leukemogenesis. Blood 1996;87:2878–2884.
Sugiyama H. WT1 (Wilms tumor gene 1): biology and cancer immunotherapy. Jpn J Clin Oncol 2010;40:377–387.
Van Driessche A, Berneman ZN, Van Tendeloo VF. Active specific immunotherapy targeting the Wilms tumor protein 1 (WT1) for patients with hematological malignancies and solid tumors: lessons from early clinical trials. Oncologist 2012;17:250–259.
Nomdedéu JF, Hoyos M, Carricondo M, Bussaglia E, Estiviu C, Esteve J, et al. Bone marrow WT1 levels at diagnosis, post-induction and post-intensification in adult de novo AML. Leukemia 2013;27:2157–2164.
Scheibenbogen C, Letsch A, Thiel E, Schmittel A, Mailaender V, Baerwolf S, et al. CD8 T-cell responses to Wilms tumor gene product WT1 and proteinase 3 in patients with acute myeloid leukemia. Blood 2002;100:2132–2137.
Rezvani K, Brenchley JM, Price DA, Kilical Y, Gostick E, Sewell AK, et al. T-cell responses directed against multiple HLA-A*0201-restricted epitopes derived from Wilms’ tumor 1 protein in patients with leukemia and healthy donors: identification, quantification, and characterization. Clin Cancer Res 2005;11:8799–8807.
Andersson C, Li X, Lorenz F, Golovleva I, Wahlin A, Li A. Reduction in WT1 gene expression during early treatment predicts the outcome in patients with acute myeloid leukemia. Diagn Mol Pathol 2012;21:225–233.
Kwon YR, Son MJ, Kim HJ, Kim YJ. Reactivation of silenced WT1 transgene by hypomethylating agents: implications for in vitro modeling of chemoimmunotherapy. Immune Netw 2012;12:58–65.
Ho PA, Kutny MA, Alonzo TA, Gerbing RB, Joaquin J, Raimondi SC, et al. Leukemic mutations in the methylationassociated genes DNMT3A and IDH2 are rare events in pediatric AML: a report from the Children’s Oncology Group. Pediatr Blood Cancer 2011;57:204–209.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by National Natural Science Foundation of China (No. 81403223), Zhejiang Provincial Natural Science Foundation of China (No. LQ14H290003) and Science and Technology Foundation of Zhejiang Province (No. 2015C33173)
Rights and permissions
About this article
Cite this article
Wang, Lp., Zhao, Yn., Sun, X. et al. Effects of bufalin on up-regulating methylation of Wilm’s tumor 1 gene in human erythroid leukemic cells. Chin. J. Integr. Med. 23, 288–294 (2017). https://doi.org/10.1007/s11655-017-2404-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-017-2404-1