Abstract
Objective
To observe the efficacy and safety of total glucosides of paeony capsule (TGPC) in patients with mild and moderate alopecia areata.
Methods
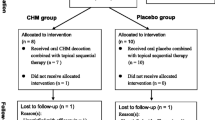
A total of 86 outpatients were randomly allocated into two groups of TGPC (treatment, 44 cases) and compound glycyrrhizin tablet (control, 42 cases). The treatment group was given oral TGPC, three times daily and 600 mg per time; the control group was given oral compound glycyrrhizin tablets, three times daily and 50 mg per time. In addition, both groups were given 10 mg of vitamin B2 and tapped the bold patches with massage. The treatment course was three months for both groups. Peripheral blood T-cell subsets (CD3+CD4+, CD3+CD8+, Th, Ts, Th/Ts) of 10 patients randomly selected from each group respectively were tested before and after three months of treatment. The effectiveness and adverse reaction of all cases were observed each month. The safety was evaluated according to the incidence rate of adverse reaction.
Results
In the treatment group, the cured and markedly effective rate was 36.36% (16/44), 50.00% (22/44) and 68.18% (30/44) at the end of first, second and third month of treatment, respectively, and the incidence rate of adverse reaction was 13.64% (6/44). In the control group, the cured and markedly effective rate was 38.10% (16/42), 57.14% (24/42) and 71.43% (30/42), respectively, and the incidence rate of adverse reaction was 16.67% (7/42). The cured and markedly effective rate and the incidence rate of adverse reaction were similar in both groups (P>0.05). TGPC and compound glycyrrhizin tablet can inhibit CD3+CD4+ and CD3+CD8+, and decrease the ratio of Th/Ts (P<0.05).
Conclusion
TGPC is effective and safe in the treatment of alopecia areata.
Similar content being viewed by others
References
Gilhar A, Shalaginov R, Assy B, Serafimovich S, Kalish RS. Alopecia areata is a T-lymphocyte mediated autoimmune disease: lesional human T-lymphocytes transfer alopecia areata to human skin grafts on SCID mice. J Investig Dermatol Symp Proc 1999;4:207–210.
Zhou J, Yang QP, Xu F, Lou W, Sheng YY. Observation on compound glycyrrhizin in the treatment of 107 patients with alopecia areata. Chin J Dermatovenereol (Chin) 2008;2:446–447.
Zhang JX. Pharmacological action, clinical application and adverse reaction of compound glycyrrhizin tablet for alopecia areata. Guide China Med (Chin) 2011;9:198–199.
Zheng H. Pharmacological anti-rheumatic action of total glucosides of paeony. Zhejiang J Integr Tradit Chin West Med (Chin) 2008;18:520.
Ye X. Effect of total glucosides of paeony capsule combined with plum-blossom needle for alopecia areata in 50 cases. Chin J Pract Med (Chin) 2011;38:119–120.
Zhao B, ed. Clinical dermatology. 3rd ed. Nanjing: Jiangsu Science and Technology Press;2006:947–948.
Olsen EA, Hordinsky MK, Price VH, Roberts JL, Shapiro J, Canfield D, et al. Alopecia areata investigational assessment guidelines-part II. National Alopecia Areata Foundation. J Am Acad Dermatol 2004;51:440–447.
Special Committee of Dermatovenereology, Chinese Association of Integrated Traditional and Western Medicine. Standards for diagnosis and treatment of five skin diseases (Draft). Chin J Integr Tradit West Med (Chin) 1992;12:56.
McElwee KJ, Freyschmidt-Paul P, Hoffmann R, Kissling S, Hummel S, Vitacolonna M, et al. Transfer of CD8+ cells induces localized hair loss whereas CD4+/CD25− cells promote systemic alopecia areata and CD4+/CD25+ cells blockade disease onset in the C3H/HeJ mouse model. J Invest Dermatol 2005;124:947–957.
Gilhar A, Landau M, Assy B, Shalaginov R, Serafimovich S, Kalish RS. Mediation of alopecia areata by cooperation between CD4+ and CD8+ T lymphocytes: transfer to human scalp explants on Prkdc(scid) mice. Arch Dermatol 2002;138:916–922.
Huang WN, Hou XZ, Lu HQ, Lu HM, Xu X. Studies on the relationship between T lymphocyte subtypes and alopecia areata. Soutern China J Dermato-venereol (Chin) 2007;14:208–210.
Song FW, Bai YM, Chen YX, Cao Y, Li XJ, et al. The molecular configuration and clinical effectiveness of compound glycyrrhizin tablets (β-compound glycyrrhizin preparation). China Pharm (Chin) 2003;14:304.
Zhang ZHM. Clinical observation of compound glycyrrhizin tablets combined with cystinuria for alopecia areata. China J Leprosy Skin Dis (Chin) 2007;23:349.
Zhang MQ, Ma M. Clinical application and improvement of the total glucosides of peony. J Pediatr Pharm (Chin) 2008;12:48–50.
Wang YY, Xiong XL, Long JW. Influence of total glucosides of paeony on TNF-α and IL-8 in patients with psoriasis vulgaris stationary phase. Modern J Integr Chin Tradit West Med (Chin) 2009;18:2011–2012.
Guan XH, Wang NL. Effect of TGP on peripheral blood T cell subsets of plaque psoriasis vulgaris. Henan Tradit Chin Med (Chin) 2010;30:455–456.
Wang YW, Wang YJ. Pharmacological study and clinical application of total glucosides of peony in autoimmune diseases. J Zhejiang Univ Tradit Chin Med (Chin) 2007;31:240–241.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, Dq., You, Lp., Song, Ph. et al. A randomized controlled trial comparing total glucosides of paeony capsule and compound glycyrrhizin tablet for alopecia areata. Chin. J. Integr. Med. 18, 621–625 (2012). https://doi.org/10.1007/s11655-012-1173-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-012-1173-0