Abstract
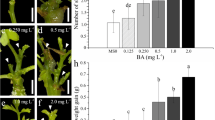
This study demonstrated that somatic embryogenesis and de novo shoot organogenesis-based systems of root-derived Lisianthus (Eustoma grandiflorum) explants can be alternatively induced by exogenous supply of auxin or cytokinin. Somatic embryogenesis was observed when root explants were cultured in the dark on Murashige and Skoog-based medium supplemented with 10 μM 2,4-dichlorophenoxyacetic acid (2,4-D). Somatic embryos were differentiated by transferring embryonic calluses to an embryo conversion phase medium containing 2 μM 6-benzyladenine (BA) to promote full plantlet development. Regarding de novo shoot organogenesis, the addition of 4 μM of either BA or zeatin was the most effective treatment for inducing adventitious shoot buds. A detailed histological characterization of somatic embryogenesis and de novo shoot organogenesis showed that both morphogenetic processes shared the same cellular origin. The formation of somatic embryos and adventitious shoot buds occurred through the reactivation of pericycle and vascular parenchyma cells into proembryos and meristemoids, respectively, which consisted of meristematic cells with similar characteristics. These results provide further evidence of optimization of in vitro propagation as a useful approach to improve this important ornamental species.





Similar content being viewed by others
References
Akashi R, Hoffmann-Tsay S-S, Hoffmann F (1998) Selection of a super-growing legume root culture that permits controlled switching between root cloning and direct embryogenesis. Theor Appl Genet 96:758–764
Arnhold E (2014) Easyanova: Analysis of variance and other important complementary analyzes. R package version 4
Atta R, Laurens L, Boucheron-Dubuisson E, Guivarc'h A, Carnero E, Giraudat-Pautot V, Rech P, Chriqui D (2009) Pluripotency of Arabidopsis xylem pericycle underlies shoot regeneration from root and hypocotyl explants grown in vitro. Plant J 57:626–644
Barrueto Cid LP, Teixeira JB (2006) Indução de organogênese em Lisianthus, Eustoma grandiflorum, a partir de fragmentos foliares cultivados in vitro. Boletim de Pesquisa e Desenvolvimento/Embrapa Recursos Genéticos e Biotecnologia, Brasilia-DF
Blakesley D (1991) Uptake and metabolism of 6-benzyladenine in shoot cultures of Musa and Rhododendron. Plant Cell Tissue Organ Cult 25:69–74
Bozzola JJ, Russell LD (1992) Electron Microscopy: Principles and Techniques for Biologists, 2nd edn. Jones and Barlett, Boston
Cruz ACF, Rocha DI, Iarema L, Ventrella MC, Costa MGC, Paiva Neto VB, Otoni WC (2014) In vitro organogenesis from root culture segments of Bixa orellana L. (Bixaceae). In Vitro Cell Dev Biol - Plant 50:76–83
De Smet I, Vanneste S, Inzé D, Beeckman T (2006) Lateral root initiation or the birth of a new meristem. Plant Mol Biol 60:871–887
Dodsworth S (2009) A diverse and intricate signalling network regulates stem cell fate in the shoot apical meristem. Dev Biol 336:1–9
Esizad SG, Kaviani B, Tarang A, Zanjani SB (2012) Micropropagation of Lisianthus (Eustoma grandiflorum), an ornamental plant. Plant Omics J 5:314–319
Fehér A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult 74:201–228
Ferreira EB, Cavalcanti PP, Nogueira DA (2013) ExpDes: Experimental Designs package versão:1.1.2
Fiuk A, Rybczyński JJ (2007a) Morphogenic capability of Gentiana kurroo Royle seedling and leaf explants. Acta Physiol Plant 30:157–166
Fiuk A, Rybczyński JJ (2007b) The effect of several factors on somatic embryogenesis and plant regeneration in protoplast cultures of Gentiana kurroo (Royle). Plant Cell Tissue Organ Cult 91:263–271
Fiuk A, Rybczyński JJ (2008) Factors influencing efficiency of somatic embryogenesis of Gentiana kurroo (Royle) cell suspension. Plant Biotechnol Rep 2:33–39
Furukawa H, Matsubara C, Shigematsu N (1990) Shoot regeneration from the roots of prairie gentian (Eustoma grandiflorum (Griseb.) Schinners). Plant Tissue Cult Lett 7:11–13
Harbaugh BK (2007) Lisianthus Eustoma grandiflorum. In: Anderson NO (ed) Flower breeding and genetics issues, challenges and opportunities for the 21st century, 1st edn. Springer, Netherlands, pp 645–663
Ikeuchi M, Ogawa Y, Iwase A, Sugimoto K (2016) Plant regeneration: cellular origins and molecular mechanisms. Development 143:1442–1451
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol 27:137–138
Kaminek M, Motyka V, Vankova R (1997) Regulation of cytokinin content in plant cells. Physiol Plant 101:689–700
Kaviani B (2014) Micropropagation of ten weeks (Matthiola incana) and Lisianthus (Eustoma grandiflorum) (two ornamental plants) by using kinetin (kin), naphthalene acetic acid (NAA) and 2,4-dichlorophenoxyacetic acid (2,4-D). Acta Sci Pol Hortorum Cultus 13:141–154
Komai F, Okuse I, Harada T (1995) Histological identification of somatic embryogenesis from excised root tissues of spinach (Spinacia oleracea L.). Plant Tissue Cult Lett 12:313–315
Kraut M, Wójcikowska B, Ledwoń A, Gaj MD (2011) Immature zygotic embryo cultures of Arabidopsis. A model system for molecular studies on morphogenic pathways induced in vitro. Acta Biol Crac Ser Bot 53:59–67
Krishna Kumar G, Thomas TD (2012) High frequency somatic embryogenesis and synthetic seed production in Clitoria ternatea Linn. Plant Cell Tissue Organ Cult 110:141–151
Kurczyńska EU, Potocka I, Dobrowolska I, Kulinska-Lukaszek K, Sala K, Wrobel J (2012) Cellular Markers for Somatic Embryogenesis. In: Sato K-I (ed) Embryogenesis. InTech, Rijeka, pp 307–332
Lombardi SP, Ribeiro I, Nogueira Stolf MC, Appezzato-da-Glória B (2007) In vitro shoot regeneration from roots and leaf discs of Passiflora cincinnata Mast. Braz Arch Biol Technol 50:239–247
Mikuła A, Rybczyński JJ (2001) Somatic embryogenesis of Gentiana genus I. The effect of the preculture treatment and primary explant origin on somatic embryogenesis of Gentiana cruciata (L.), G. pannonica (Scop.), and G. tibetica (King). Acta Physiol Plant 23:15–25
Motte H, Vereecke D, Geelen D, Werbrouck S (2014) The molecular path to in vitro shoot regeneration. Biotechnol Adv 32:107–121
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nhut DT, Tuan NS, Ngoc HM, Uyen PN, Don NT, Mai NT, Teixeira da Silva JA (2006) Somatic embryogenesis induction from in vitro leaf cultures of Lisianthus (Eustoma grandiflorum (Raf.) Shinn.). Propag Ornam Plant 6:121–127
O’Brien T, McCully M (1981) The study of plant structure principles and selected methods, 1st ed. Termarcarphi Pty Ltda., Melbourne, Australia
Ogas J (1997) Cellular differentiation regulated by gibberellin in the Arabidopsis thaliana pickle mutant. Science 277:91–94
Ördögh M, Jámbor-Benczúr E, Tilly-Mándy A (2006) Micropropagation of “Echo” cultivars of Eustoma grandilorum. Acta Hortic 725:457–460
Paim-Pinto LD, Rocha AAM, Monteiro RM, Lemes SM, Jardim OE, Otoni WC (2011) Somatic embryogenesis from mature zygotic embryos of commercial passionfruit (Passiflora edulis Sims) commercial populations. Plant Cell Tissue Organ Cult 107:521–530
Parimalan R, Venugopalan A, Giridhar P, Ravishankar GA (2011) Somatic embryogenesis and Agrobacterium-mediated transformation in Bixa orellana L. Plant Cell Tissue Organ Cult 105:317–328
Parveen S, Shahzad A (2011) A micropropagation protocol for Cassia angustifolia Vahl. from root explants. Acta Physiol Plant 33:789–796
Pathak S, Mishra BK, Misra P, Misra P, Joshi VK, Shukla S, Trivedi PK (2012) High frequency somatic embryogenesis, regeneration and correlation of alkaloid biosynthesis with gene expression in Papaver somniferum. Plant Growth Regul 68:17–25
Pulianmackal AJ, Kareem AVK, Durgaprasad K, Trivedi ZB, Prasad K (2014) Competence and regulatory interactions during regeneration in plants. Front Plant Sci 5:142
R Core Team (2014) Software R: A language and environment for statistical computing version:3.0.3
Rabobank (2015) Floriculture Map 2015. Rabobank Industry Note 475
Rocha DI, Monte-Bello CC, Dornelas MC (2015) Alternative induction of de novo shoot organogenesis or somatic embryogenesis from in vitro cultures of mature zygotic embryos of passion fruit (Passiflora edulis Sims) is modulated by the ratio between auxin and cytokinin in the medium. Plant Cell Tissue Organ Cult 120:1087–1098
Rocha DI, Vieira LM, Tanaka FAO, Silva LC, Otoni WC (2012) Anatomical and ultrastructural analyses of in vitro organogenesis from root explants of commercial passion fruit (Passiflora edulis Sims). Plant Cell Tissue Organ Cult 111:69–78
Ruffoni B, Bassolino L (2016) Somatic Embryogenesis in Lisianthus (Eustoma russellianum Griseb.). In: Maria AG, Maurizio L (eds) In Vitro Embryogenesis in Higher Plants. Springer New York, New York, pp 359–370
Ruffoni B, Damiano C, Massabò F, Esposito P (1990) Organogenesis and embryogenesis in Lisianthus russellianus Hook. Acta Hortic 280:83–88
Saldanha CW, Otoni CG, Azevedo JLF, Dias LLC, Rêgo MM, Otoni WC (2012) A low-cost alternative membrane system that promotes growth in nodal cultures of Brazilian ginseng [Pfaffia glomerata (Spreng.) Pedersen]. Plant Cell Tissue Organ Cult 110:413–422
Semeniuk P, Griesbach RJ (1987) In vitro propagation of prairie gentian. Plant Cell Tissue Organ Cult 8:249–253
Sahai A, Shahzad A, Sharma S (2010) Histology of organogenesis and somatic embryogenesis in excised root cultures of an endangered species Tylophora indica (Asclepiadaceae). Aust J Bot 58:198–205
Savona M, Mattioli R, Nigro S, Falasca G, Della RF, Costantino P, De Vries S, Ruffoni B, Trovato M, Altamura MM (2012) Two SERK genes are markers of pluripotency in Cyclamen persicum Mill. J Exp Bot 63:471–488
Shemer O, Landau U, Candela H, Zemach A, Eshed L (2015) Competency for shoot regeneration from Arabidopsis root explants is regulated by DNA methylation. Plant Sci 238:251–261
Stanišić M, Raspor M, Ninković S, Milošević S, Ćalić D, Bohanec B, Trifunović M, Petrić M, Subotić A, Jevremović S (2015) Clonal fidelity of Iris sibirica plants regenerated by somatic embryogenesis and organogenesis in leaf-base culture — RAPD and flow cytometer analyses. S Afr J Bot 96:42–52
Sugimoto K, Meyerowitz EM (2013) Regeneration in Arabidopsis tissue culture. In: De Smet I (ed) Plant Organogenesis: Methods and Protocols. Methods in Molecular Biology. Humana Press, Totowa, New Jersey, pp 265–275
Sugimoto K, Jiao Y, Meyerowitz EM (2010) Arabidopsis regeneration from multiple tissues occurs via a root development pathway. Dev Cell 18:463–471
Twyford CT, Mantell SH (1996) Production of somatic embryos and plantlets from root cells of the Greater Yam. Plant Cell Tissue Organ Cult 46:17–26
Verdeil J-L, Alemanno L, Niemenak N, Tranbarger TJ (2007) Pluripotent versus totipotent plant stem cells: dependence versus autonomy? Trends Plant Sci 12:245–252
Vieira LM, Rocha DI, Taquetti MF, da Silva LC, de Campos JMS, Viccini LF, Otoni WC (2014) In vitro plant regeneration of Passiflora setacea D.C. (Passifloraceae): the influence of explant type, growth regulators, and incubation conditions. In Vitro Cell Dev Biol - Plant 50:738–745
Vinocur B, Carmi T, Altman A, Ziv M (2000) Enhanced bud regeneration in aspen (Populus tremula L.) roots cultured in liquid media. Plant Cell Rep 19:1146–1154
Wan L, Yang X, Gui M, Qu S, Su Y, Zhou X (2009) In vitro anther culture and plant induction of Eustoma russellianum. Southwest China J Agric Sci 22:1424–1427
Wang XD, Nolan KE, Irwanto RR, Sheahan MB, Rose RJ (2011) Ontogeny of embryogenic callus in Medicago truncatula: The fate of the pluripotent and totipotent stem cells. Ann Bot 107:599–609
Wu HJ, Wang XX, Li Y, Zhang DG, Zhang BW, Xin Y (2011) Propagation of Gentiana macrophylla (Pall) from hairy root explants via indirect somatic embryogenesis and gentiopicroside content in obtained plants. Acta Physiol Plant 33:2229–2237
Xu L, Huang H (2014) Genetic and epigenetic controls of plant regeneration. In: Galliot B (ed) Current Topics in Developmental Biology, 1st edn. Elsevier Inc., USA, pp 1–33
Yang JL, Seong ES, Kim MJ, Ghimire BK, Kang WH, Yu CY, Li CH (2009) Direct somatic embryogenesis from pericycle cells of broccoli (Brassica oleracea L. var. italica) root explants. Plant Cell Tissue Organ Cult 100:49–58
You CR, Fan TJ, Gong XQ, Bian FH, Liang LK, Qu FN (2011) A high-frequency cyclic secondary somatic embryogenesis system for Cyclamen persicum Mill. Plant Cell Tissue Organ Cult 107:233–242
You XL, Tan X, Dai JL, Li YH, Choi YE (2012) Large-scale somatic embryogenesis and regeneration of Panax notoginseng. Plant Cell Tissue Organ Cult 108:333–338
Acknowledgements
The authors would like to thank the Microscopy and Microanalysis Center of the Federal University of Viçosa (Viçosa, MG, Brazil) and the Brazilian funding agencies Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG), and Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior (CAPES) for providing financial support.
Authors’ contributions
MY-O, ACFC, MVMP, DIR, DSB, JGB, ADK, and WCO conceived, designed, and performed the experiments; MYO raised the in vitro plants used in the experiments; MY-O, ACFC, and MVMP carried out histological analysis; MY-O, DIR, DSB, JGB, and WCO interpreted the acquired data and drafted the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Editor: Jayasankar Subramanian
Rights and permissions
About this article
Cite this article
Yumbla-Orbes, M., da Cruz, A.C.F., Pinheiro, M.V.M. et al. Somatic embryogenesis and de novo shoot organogenesis can be alternatively induced by reactivating pericycle cells in Lisianthus (Eustoma grandiflorum (Raf.) Shinners) root explants. In Vitro Cell.Dev.Biol.-Plant 53, 209–218 (2017). https://doi.org/10.1007/s11627-017-9800-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-017-9800-2