Abstract
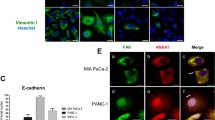
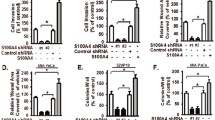
Previous studies implicated ATX/LPA axis as a potential driver of tumorigenesis and progression in pancreatic cancer. This study aimed to determine the existence of the autocrine pathway of ATX/LPA action in pancreatic cancer cells, and to elucidate its influence on focal adhesion kinase (FAK) activation, cellular proliferation, apoptosis, and migration. Firstly, we identified the lysophosphatidic acid (LPA) concentrations in cultured cell supernatant by ELISA and observed the effect of the autotaxin (ATX)-specific inhibitor S32826 on LPA concentrations. We found the existence of a certain concentration of LPA in cellular supernatant, which was significantly decreased by S32826 in a dose- and time-dependent manner. A maximum response was observed at 50 μM for 72 h. Secondly, the effect of S32826 on the protein expression and intracellular sublocalization of total FAK and phosphorylated FAK (pY397 FAK) was determined by Western blot and immunofluorescence staining. It was found that the expression of total FAK and pY397 FAK and their distribution along the cell membrane where adhesion structures are located were significantly decreased by S32826. Finally, we observed the influence of S32826 on cell proliferation, apoptosis, and migration by CCK-8 assay, flow cytometric analysis, and transwell migration assay. Results showed that cell viability and migration were significantly declined, and the proportions of apoptotic cells were significantly increased by S32826. This study verified the existence of autocrine regulation of LPA secretion via producing ATX by pancreatic cancer cells in vitro and the important role of LPA/ATX axis on FAK activation, cell proliferation, apoptosis, and motility.





Similar content being viewed by others
References
Auciello FR, Bulusu V, Oon C, Tait-Mulder J, Berry M, Bhattacharyya S, Tumanov S, Allen-Petersen BL, Link J, Kendsersky ND, Vringer E, Schug M, Novo D, Hwang RF, Evans RM, Nixon C, Dorrell C, Morton JP, Norman JC, Sears RC, Kamphorst JJ, Sherman MH (2019) A stromal lysolipid-autotaxin signaling axis promotes pancreatic tumor progression. Cancer Discov 9:617–627
Benesch MG, Zhao YY, Curtis JM, McMullen TP, Brindley DN (2015) Regulation of autotaxin expression and secretion by lysophosphatidate and sphingosine 1-phosphate. J Lipid Res 56:1134–1144
Bian D, Mahanivong C, Yu J, Frisch SM, Pan ZK, Ye RD, Huang S (2006) The G12/13-RhoA signaling pathway contributes to efficient lysophosphatidic acid-stimulated cell migration. Oncogene 25:2234–2244
Biffi G, Tuveson DA (2019) A FATal combination: fibroblast-derived lipids and cancer-derived autotaxin promote pancreatic cancer growth. Cancer Discov 9:578–580
Burridge K (2017) Focal adhesions: a personal perspective on a half century of progress. FEBS J 284:3355–3361
Chen J, Li H, Xu W, Guo X (2021) Evaluation of serum ATX and LPA as potential diagnostic biomarkers in patients with pancreatic cancer. BMC Gastroenterol 21:58
Eder AM, Sasagawa T, Mao M, Aoki J, Mills GB (2000) Constitutive and lysophosphatidic acid (LPA)-induced LPA production: role of phospholipase D and phospholipase A2. Clin Cancer Res 6:2482–2491
Gabarra-Niecko V, Schaller MD, Dunty JM (2003) FAK regulates biological processes important for the pathogenesis of cancer. Cancer Metastasis Rev 22:359–374
George J, Lim JS, Jang SJ, Cun Y, Ozretić L, Kong G, Leenders F, Lu X, Fernández-Cuesta L, Bosco G, Müller C, Dahmen I, Jahchan NS, Park KS, Yang D, Karnezis AN, Vaka D, Torres A, Wang MS, Korbel JO, Menon R, Chun SM, Kim D, Wilkerson M, Hayes N, Engelmann D, Pützer B, Bos M, Michels S, Vlasic I, Seidel D, Pinther B, Schaub P, Becker C, Altmüller J, Yokota J, Kohno T, Iwakawa R, Tsuta K, Noguchi M, Muley T, Hoffmann H, Schnabel PA, Petersen I, Chen Y, Soltermann A, Tischler V, Choi CM, Kim YH, Massion PP, Zou Y, Jovanovic D, Kontic M, Wright GM, Russell PA, Solomon B, Koch I, Lindner M, Muscarella LA, la Torre A, Field JK, Jakopovic M, Knezevic J, Castaños-Vélez E, Roz L, Pastorino U, Brustugun OT, Lund-Iversen M, Thunnissen E, Köhler J, Schuler M, Botling J, Sandelin M, Sanchez-Cespedes M, Salvesen HB, Achter V, Lang U, Bogus M, Schneider PM, Zander T, Ansén S, Hallek M, Wolf J, Vingron M, Yatabe Y, Travis WD, Nürnberg P, Reinhardt C, Perner S, Heukamp L, Büttner R, Haas SA, Brambilla E, Peifer M, Sage J, Thomas RK (2015) Comprehensive genomic profiles of small cell lung cancer. Nature 524:47–53
Golubovskaya VM (2010) Focal adhesion kinase as a cancer therapy target. Anticancer Agents Med Chem 10:735–741
Hashimoto K, Morishige K, Sawada K, Tahara M, Shimizu S, Sakata M, Tasaka K, Murata Y (2005) Geranylgeranylacetone inhibits lysophosphatidic acid-induced invasion of human ovarian carcinoma cells in vitro. Cancer 103:1529–1536
Haskell H, Natarajan M, Hecker TP, Ding Q, Stewart J Jr, Grammer JR, Gladson CL (2003) Focal adhesion kinase is expressed in the angiogenic blood vessels of malignant astrocytic tumors in vivo and promotes capillary tube formation of brain microvascular endothelial cells. Clin Cancer Res 9:2157–2165
Ho WJ, Jaffee EM, Zheng L (2020) The tumour microenvironment in pancreatic cancer -clinical challenges and opportunities. Nat Rev Clin Oncol 17:527–540
Houben AJ, Moolenaar WH (2011) Autotaxin and LPA receptor signaling in cancer. Cancer Metastasis Rev 30:557–565
Huang W, Navarro-Serer B, Jeong YJ, Chianchiano P, Xia L, Luchini C, Veronese N, Dowiak C, Ng T, Trujillo MA, Huang B, Pflüger MJ, Macgregor-Das AM, Lionheart G, Jones D, Fujikura K, Nguyen-Ngoc KV, Neumann NM, Groot VP, Hasanain A, van Oosten AF, Fischer SE, Gallinger S, Singhi AD, Zureikat AH, Brand RE, Gaida MM, Heinrich S, Burkhart RA, He J, Wolfgang CL, Goggins MG, Thompson ED, Roberts NJ, Ewald AJ, Wood LD (2020) Pattern of invasion in human pancreatic cancer organoids is associated with loss of SMAD4 and clinical outcome. Cancer Res 80:2804–2817
Jiang X, Sinnett-Smith J, Rozengurt E (2007) Differential FAK phosphorylation at Ser-910, Ser-843 and Tyr-397 induced by angiotensin II, LPA and EGF in intestinal epithelial cells. Cell Signal 19:1000–1010
Jung ID, Lee J, Lee KB, Park CG, Kim YK, Seo DW, Park D, Lee HW, Han JW, Lee HY (2004) Activation of p21-activated kinase 1 is required for lysophosphatidic acid-induced focal adhesion kinase phosphorylation and cell motility in human melanoma A2058 cells. Eur J Biochem 271:1557–1565
Kong K, Guo M, Liu Y, Zheng J (2020) Progress in animal models of pancreatic ductal adenocarcinoma. J Cancer 11:1555–1567
Krempley BD, Yu KH (2017) Preclinical models of pancreatic ductal adenocarcinoma. Chin Clin Oncol 6:25
Leblanc R, Houssin A, Peyruchaud O (2018) Platelets, autotaxin and lysophosphatidic acid signalling: win-win factors for cancer metastasis. Br J Pharmacol 175:3100–3110
Lee SC, Dacheux MA, Norman DD, Balázs L, Torres RM, Augelli-Szafran CE, Tigyi GJ (2020) Regulation of tumor immunity by lysophosphatidic acid. Cancers (Basel) 12:1202
Leve F, Marcondes TG, Bastos LG, Rabello SV, Tanaka MN, Morgado-Díaz JA (2011) Lysophosphatidic acid induces a migratory phenotype through a crosstalk between RhoA-Rock and Src-FAK signalling in colon cancer cells. Eur J Pharmacol 671:7–17
Liao Y, Mu G, Zhang L, Zhou W, Zhang J, Yu H (2013) Lysophosphatidic acid stimulates activation of focal adhesion kinase and paxillin and promotes cell motility, via LPA1-3, in human pancreatic cancer. Dig Dis Sci 58:3524–3533
Linseman DA, Hofmann F, Fisher SK (2000) A role for the small molecular weight GTPases, Rho and Cdc42, in muscarinic receptor signaling to focal adhesion kinase. J Neurochem 74:2010–2020
Nakai Y, Ikeda H, Nakamura K, Kume Y, Fujishiro M, Sasahira N, Hirano K, Isayama H, Tada M, Kawabe T, Komatsu Y, Omata M, Aoki J, Koike K, Yatomi Y (2011) Specific increase in serum autotaxin activity in patients with pancreatic cancer. Clin Biochem 44:576–581
Riquelme-Guzmán C, Contreras O, Brandan E (2018) Expression of CTGF/CCN2 in response to LPA is stimulated by fibrotic extracellular matrix via the integrin/FAK axis. Am J Physiol Cell Physiol 314:C415–C427
Ryder NM, Guha S, Hines OJ, Reber HA, Rozengurt E (2001) G protein-coupled receptor signaling in human ductal pancreatic cancer cells: neurotensin responsiveness and mitogenic stimulation. J Cell Physiol 186:53–64
Shimizu T, Fukuoka K, Takeda M, Iwasa T, Yoshida T, Horobin J, Keegan M, Vaickus L, Chavan A, Padval M, Nakagawa K (2016) A first-in-Asian phase 1 study to evaluate safety, pharmacokinetics and clinical activity of VS-6063, a focal adhesion kinase (FAK) inhibitor in Japanese patients with advanced solid tumors. Cancer Chemother Pharmacol 77:997–1003
Tao J, Yang G, Zhou W, Qiu J, Chen G, Luo W, Zhao F, You L, Zheng L, Zhang T, Zhao Y (2021) Targeting hypoxic tumor microenvironment in pancreatic cancer. J Hematol Oncol 14:14
Tapial Martínez P, López Navajas P, Lietha D (2020) FAK structure and regulation by membrane interactions and force in focal adhesions. Biomolecules 10:179
Xie D, Yu S, Li L, Quan M, Gao Y (2020) The FOXM1/ATX signaling contributes to pancreatic cancer development. Am J Transl Res 12:4478–4487
Acknowledgements
This study was supported by the Wuhan Medical Backbone Talent Project 2017 (2017060201010224) from Wuhan Municipal Health Commission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Editor: Tetsuji Okamoto
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liao, Y., Liu, L., Yang, J. et al. ATX/LPA axis regulates FAK activation, cell proliferation, apoptosis, and motility in human pancreatic cancer cells. In Vitro Cell.Dev.Biol.-Animal 58, 307–315 (2022). https://doi.org/10.1007/s11626-022-00660-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-022-00660-3