Abstract
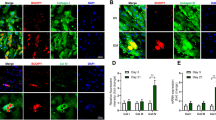
Adipose-derived mesenchymal stem cells (ADSCs) are useful cell model to study adipogenesis and energy metabolism. However, the biological characteristics of bovine ADSCs (bADSCs) remain unclear. This study aimed to isolate and identify bADSCs and further investigate fatty acid (FA)-related gene expression and composition of FAs during adipogenesis. The growth curve showed the bADSCs of P5 cells had rapid proliferation superior to P10–P50. The colony formation assay showed colony number of P5 cells was higher than that of P50 cells (51.67 ± 3.06 vs 35.67 ± 6.43, P < 0.05). The immunofluorescence showed that bADSCs were positive for CD13, CD44, CD49d, CD90, CD105, and Vimentin while negative for CD34. The multipotential towards adipocyte, osteocyte, and chondrocyte was confirmed by specific histological staining and lineage gene expression. During adipogenic induction, the genes related to lipogenesis and lipolysis were assessed by real-time PCR and the FA composition was detected by GC-MS. Expression of lipogenesis-related genes showed coordinated regulation as peaking on day 7 and declining until induction ended, including PPARγ, SREBP1, ACC1, FAS, ELOVL6, SCD1, and FABP4. FA deposition-related genes (DGAT1 and ACAT1) increased until day 14. Lipolysis genes (CPT-1A, VLCAD, and ACO) showed a variant expression pattern. The profile of FAs showed that proportion of the FAs (C4–C15, ≥ C22) increased, but proportion of long-chain fatty acids (C16–C20) reduced after induction. And saturated FAs (SFA) decreased while monounsaturated FAs (MUFA) and polyunsaturated FAs (PUFA) increased during adipogenesis. These data suggest that bADSCs possess the characteristics of mesenchymal stem cells and have active de novo lipogenesis (DNL) and desaturation of FAs during adipogenesis.





Similar content being viewed by others
References
Bauche IB, El Mkadem SA, Pottier AM, Senou M, Many MC, Rezsohazy R, Penicaud L, Maeda N, Funahashi T, Brichard SM (2007) Overexpression of adiponectin targeted to adipose tissue in transgenic mice: impaired adipocyte differentiation. Endocrinology 148:1539–1549
Böhrnsen F, Lindner U, Meier M, Gadallah A, Schlenke P, Lehnert H, Rohwedel J, Kramer J (2009) Murine mesenchymal progenitor cells from different tissues differentiated via mesenchymal microspheres into the mesodermal direction. BMC Cell Biol 10:92
Borgne FL, Demarquoy J (2012) Interaction between peroxisomes and mitochondria in fatty acid metabolism. Open J Mol Integr Physiol 2:27–33
Chandran K, Goswami S, Sharma-Walia N (2016) Implications of a peroxisome proliferator-activated receptor alpha (PPARalpha) ligand clofibrate in breast cancer. Oncotarget 7:15577–15599
Coelho M, Oliveira T, Fernandes R (2013) Biochemistry of adipose tissue: an endocrine organ. Arch Med Sci: AMS 9:191–200
Collins JM, Neville MJ, Hoppa MB, Frayn KN (2010) De novo lipogenesis and stearoyl-CoA desaturase are coordinately regulated in the human adipocyte and protect against palmitate-induced cell injury. J Biol Chem 285:6044–6052
Collins JM, Neville MJ, Pinnick KE, Hodson L, Ruyter B, van Dijk TH, Reijngoud DJ, Fielding MD, Frayn KN (2011) De novo lipogenesis in the differentiating human adipocyte can provide all fatty acids necessary for maturation. J Lipid Res 52:1683–1692
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. Int Soc Cell Therap Position Statement Cytotherapy 8:315–317
Feisst V, Meidinger S, Locke MB (2015) From bench to bedside: use of human adipose-derived stem cells. Stem Cells Cloning: Adv Appl 8:149–162
Gregoire FM, Smas CM, Sul HS (1998) Understanding adipocyte differentiation. Physiol Rev 78:783–809
Lu T, Xiong H, Wang K, Wang S, Ma Y, Guan W (2014) Isolation and characterization of adipose-derived mesenchymal stem cells (ADSCs) from cattle. Appl Biochem Biotechnol 174:719–728
Mateescu RG, Garrick DJ, Garmyn AJ, VanOverbeke DL, Mafi GG, Reecy JM (2015) Genetic parameters for sensory traits in longissimus muscle and their associations with tenderness, marbling score, and intramuscular fat in Angus cattle. J Anim Sci 93:21–27
Mounier C, Posner BI (2006) Transcriptional regulation by insulin: from the receptor to the gene. Can J Physiol Pharmacol 84:713–724
Ren Y, Wu H, Zhou X, Wen J, Jin M, Cang M, Guo X, Wang Q, Liu D, Ma Y (2012) Isolation, expansion, and differentiation of goat adipose-derived stem cells. Res Vet Sci 93:404–411
Roberts R, Hodson L, Dennis AL, Neville MJ, Humphreys SM, Harnden KE, Micklem KJ, Frayn KN (2009) Markers of de novo lipogenesis in adipose tissue: associations with small adipocytes and insulin sensitivity in humans. Diabetologia 52:882–890
Sampaio RV, Chiaratti MR, Santos DC, Bressan FF, Sangalli JR, Sa AL, Silva TV, Costa NN, Cordeiro MS, Santos SS, Ambrosio CE, Adona PR, Meirelles FV, Miranda MS, Ohashi OM (2015) Generation of bovine (Bos indicus) and buffalo (Bubalus bubalis) adipose tissue derived stem cells: isolation, characterization, and multipotentiality. Genet Mol Res: GMR 14:53–62
Shin JH, Kim IY, Kim YN, Shin SM, Roh KJ, Lee SH, Sohn M, Cho SY, Lee SH, Ko CY, Kim HS, Choi CS, Bae YS, Seong JK (2015) Obesity resistance and enhanced insulin sensitivity in Ahnak−/− mice fed a high fat diet are related to impaired adipogenesis and increased energy expenditure. PLoS One 10:e0139720
Sun Y, Luo J, Zhu J, Shi H, Li J, Qiu S, Wang P, Loor JJ (2016) Effect of short-chain fatty acids on triacylglycerol accumulation, lipid droplet formation and lipogenic gene expression in goat mammary epithelial cells. Anim Sci J= Nihon Chikusan Gakkaiho 87:242–249
Yamauchi T, Kamon J, Minokoshi Y, Ito Y, Waki H, Uchida S, Yamashita S, Noda M, Kita S, Ueki K, Eto K, Akanuma Y, Froguel P, Foufelle F, Ferre P, Carling D, Kimura S, Nagai R, Kahn BB, Kadowaki T (2002) Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med 8:1288–1295
Yan H, Ajuwon KM (2015) Mechanism of butyrate stimulation of triglyceride storage and adipokine expression during adipogenic differentiation of porcine Stromovascular cells. PLoS One 10:e0145940
Yoshimura H, Muneta T, Nimura A, Yokoyama A, Koga H, Sekiya I (2007) Comparison of rat mesenchymal stem cells derived from bone marrow, synovium, periosteum, adipose tissue, and muscle. Cell Tissue Res 327:449–462
Zeng G, Lai K, Li J, Zou Y, Huang H, Liang J, Tang X, Wei J, Zhang P (2013) A rapid and efficient method for primary culture of human adipose-derived stem cells. Organ 9:287–295
Zeng L, Tang W, Yin J, Feng L, Li Y, Yao X, Zhou B (2016) Alisol A 24-acetate prevents hepatic steatosis and metabolic disorders in HepG2 cells. Cell Physiol Biochem: Int J Exp Cell Physiol Biochem Pharmacol 40:453–464
Zuk PA (2010) The adipose-derived stem cell: looking back and looking ahead. Mol Biol Cell 21:1783–1787
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH (2001) Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7:211–228
Zuk PA, Zhu M, Ashjian P, Ugarte DAD, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH (2002) Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 87:125–128
Funding
This work was supported by National Science and Technology Major Project of China (No. 2014ZX08010005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yue, Y., Zhang, L., Zhang, X. et al. De novo lipogenesis and desaturation of fatty acids during adipogenesis in bovine adipose-derived mesenchymal stem cells. In Vitro Cell.Dev.Biol.-Animal 54, 23–31 (2018). https://doi.org/10.1007/s11626-017-0205-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-017-0205-7