Abstract
Objective
This study aimed to examine the association of primary open-angle glaucoma (POAG) with autonomic dysfunction by assessing the differences in systemic and ocular responses to an autonomic provocation test, the Valsalva manoeuvre (VM), between POAG patients and normal subjects.
Methods
Forty POAG and forty control subjects were subjected to the VM. Systemic and ocular parameters were measured at baseline, phase 2, and phase 4 of the VM (VM2 and VM4), where VM2 and VM4 are sympathetic and parasympathetic nervous activation states, respectively. Heart rate variability was used to assess the autonomic nervous activity, among which the high-frequency component (HF) and the low-frequency (LF)/HF ratio were used as indices of parasympathetic and sympathetic activation, respectively.
Results
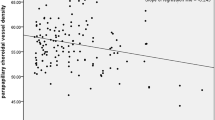
POAG patients demonstrated higher sympathetic activation (LF/HF ratio median: 2.17 vs. 1.53, P=0.000) than controls at baseline and exhibited attenuated sympathetic and parasympathetic responses (a smaller change in LF/HF and HF values) during the VM than controls. During VM, the intraocular pressure (IOP), mean blood pressure (MAP), mean ocular perfusion pressure (MOPP), and the Schlemm’s canal area (SCAR) increased from baseline to VM2 and then decreased from VM2 to VM4 in both the POAG and control groups (all P<0.05). However, when we compared the changes above, the fluctuations in IOP, MAP, and MOPP were more pronounced in POAG than in controls (all P<0.05), while the changes in amplitudes of SCAR were smaller in POAG (P<0.05). Furthermore, from VM2 to VM4, the choroid thickness (ChT) in the POAG group was significantly decreased, while it was unchanged in normal subjects (P=0.258). A regression analysis showed a significant correlation of the baseline LF/HF with IOP change values (ΔIOP) from baseline to VM2 in POAG (R2=0.147, P=0.014).
Conclusion
Patients with POAG showed more pronounced fluctuations in IOP, MAP, MOPP and ChT during the VM than controls. These reactions could be associated with autonomic dysfunction in POAG.
Similar content being viewed by others
References
The effectiveness of intraocular pressure reduction in the treatment of normal-tension glaucoma. Collaborative Normal-Tension Glaucoma Study Group. Am J Ophthalmol, 1998,126(4):498–505
Choi J, Kook MS. Systemic and Ocular Hemodynamic Risk Factors in Glaucoma. Biomed Res Int, 2015,2015:141905
McDougal DH, Gamlin PD. Autonomic control of the eye. Compr Physiol, 2015,5(1):439–473
Pasquale LR. Vascular and autonomic dysregulation in primary open-angle glaucoma. Curr Opin Ophthalmol, 2016,27(2):94–101
Kurysheva NI, Ryabova TY, Shlapak VN. Heart rate variability: the comparison between high tension and normal tension glaucoma. Epma J, 2018,9(1):35–45
Brown CM, Dütsch M, Michelson G, et al. Impaired cardiovascular responses to baroreflex stimulation in open-angle and normal-pressure glaucoma. Clin Sci (Lond), 2002,102(6):623–630
Cao L, Graham SL, Pilowsky PM. Carbohydrate ingestion induces differential autonomic dysregulation in normal-tension glaucoma and primary open angle glaucoma. PLoS One, 2018,13(6):e0198432
Wierzbowska J, Wierzbowski R, Stankiewicz A, et al. Cardiac autonomic dysfunction in patients with normal tension glaucoma: 24-h heart rate and blood pressure variability analysis. Br J Ophthalmol, 2012,96(5):624–628
Jellinek EH. The Valsalva manoeuvre and Antonio Valsalva (1666–1723). J R Soc Med, 2006,99(9):448–451
Sun L, Chen W, Chen Z, et al. Dual effect of the Valsalva maneuver on autonomic nervous system activity, intraocular pressure, Schlemm’s canal, and iridocorneal angle morphology. BMC Ophthalmol, 2020,20(1):5
Cheshire WP, Freeman R, Gibbons CH, et al. Electrodiagnostic assessment of the autonomic nervous system: A consensus statement endorsed by the American Autonomic Society, American Academy of Neurology, and the International Federation of Clinical Neurophysiology. Clin Neurophysiol, 2021,132(2):666–682
Khan JC, Hughes EH, Tom BD, et al. Pulsatile ocular blood flow: the effect of the Valsalva manoeuvre in open angle and normal tension glaucoma: a case report and prospective study. Brit J Ophthalmol, 2002,86(10):1089–1092
Heathers JA. Everything Hertz: methodological issues in short-term frequency-domain HRV. Front Physiol, 2014,5:177
Chen W, Chen Z, Xiang Y, et al. Simultaneous influence of sympathetic autonomic stress on Schlemm’s canal, intraocular pressure and ocular circulation. Sci Rep, 2019,9(1):20060
Gherghel D, Hosking SL, Armstrong R, et al. Autonomic dysfunction in unselected and untreated primary open angle glaucoma patients: a pilot study. Ophthalmic Physiol Opt, 2007,27(4):336–341
Goldstein DS, Cheshire WP, Jr. Beat-to-beat blood pressure and heart rate responses to the Valsalva maneuver. Clin Auton Res, 2017,27(6):361–367
Park HL, Jung SH, Park SH, et al. Detecting autonomic dysfunction in patients with glaucoma using dynamic pupillometry. Medicine (Baltimore), 2019,98(11):e14658
Koike Y, Kawabe T, Nishihara K, et al. Effects of azelnidipine and amlodipine on exercise-induced sympathoexcitation assessed by pupillometry in hypertensive patients. Hypertens Res, 2016,39(12):863–867
Yan X, Li M, Song Y, et al. Influence of exercise on intraocular pressure, schlemm’s canal, and the trabecular meshwork. Invest Ophthalmol Vis Sci, 2016,57(11):4733–4739
Chen W, Chen L, Chen Z, et al. Influence of the water-drinking test on intraocular pressure, schlemm’s canal, and autonomic nervous system activity. Invest Ophthalmol Vis Sci, 2018,59(8):3232–3238
Oggel K, Sommer G, Neuhann T, et al., Variations of intraocular pressure during valsalva’s maneuver in relation to body position and length of the bulbus in myopia (author’s transl) (Article in German). Graefes Arch Clin Exp Ophthalmol, 1982,218(1):51–54
Brody S, Erb C, Veit R, et al. Intraocular pressure changes: the influence of psychological stress and the Valsalva maneuver. Biol Psychol, 1999,51(1):43–57
Schuman JS, Massicotte EC, Connolly S, et al. Increased intraocular pressure and visual field defects in high resistance wind instrument players. Ophthalmology, 2000,107(1):127–133
Palamar M, Dag MY, Yagci A. The effects of Valsalva manoeuvre on Ocular Response Analyzer measurements. Clin Exp Optom, 2015,98(5):447–450
Mansouri K, Shaarawy T. Update on Schlemm’s canal based procedures. Middle East Afr J Ophthalmol, 2015,22(1):38–44
Johnstone MA, Grant WG. Pressure-dependent changes in structures of the aqueous outflow system of human and monkey eyes. Am J Ophthalmol, 1973,75(3):365–383
Hann CR, Vercnocke AJ, Bentley MD, et al. Anatomic changes in Schlemm’s canal and collector channels in normal and primary open-angle glaucoma eyes using low and high perfusion pressures. Invest Ophthalmol Vis Sci, 2014,55(9):5834–5841
Ji P, Chen L, Gong J, et al. Co-expression of vasoactive intestinal peptide and protein gene product 9.5 surrounding the lumen of human Schlemm’s canal. Exp Eye Res, 2018,170:1–7
Allingham RR, de Kater AW, Ethier CR. Schlemm’s canal and primary open angle glaucoma: correlation between Schlemm’s canal dimensions and outflow facility. Exp Eye Res, 1996,62(1):101–109
Allingham RR, de Kater AW, Ethier CR, et al. The relationship between pore density and outflow facility in human eyes. Invest Ophthalmol Vis Sci, 1992,33(5):1661–1669
Johnson M, Chan D, Read AT, et al. The pore density in the inner wall endothelium of Schlemm’s canal of glaucomatous eyes. Invest Ophthalmol Vis Sci, 2002,43(9):2950–2955
Riva CE, Cranstoun SD, Grunwald JE, et al. Choroidal blood flow in the foveal region of the human ocular fundus. Invest Ophthalmol Vis Sci, 1994,35(13):4273–4281
Asrani S, Zeimer R, Wilensky J, et al. Large diurnal fluctuations in intraocular pressure are an independent risk factor in patients with glaucoma. J Glaucoma, 2000,9(2):134–142
Choi J, Jeong J, Cho HS, et al. Effect of nocturnal blood pressure reduction on circadian fluctuation of mean ocular perfusion pressure: a risk factor for normal tension glaucoma. Invest Ophthalmol Vis Sci, 2006,47(3):831–836
Choi J, Kim KH, Jeong J, et al. Circadian fluctuation of mean ocular perfusion pressure is a consistent risk factor for normal-tension glaucoma. Invest Ophthalmol Vis Sci, 2007,48(1):104–111
Bonomi L, Marchini G, Marraffa M, et al. Vascular risk factors for primary open angle glaucoma: the Egna-Neumarkt Study. Ophthalmology, 2000,107(7):1287–1293
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest Statement
The authors declare that they have no competing interest.
This work was supported by the National Natural Science Foundation of China (No. 82070965).
Rights and permissions
About this article
Cite this article
Liu, Bb., Zhao, Bw., Wu, Fp. et al. Abnormal Systemic and Ocular Responses to the Valsalva Manoeuvre in Primary Open-Angle Glaucoma: A Case for Autonomic Failure?. CURR MED SCI 43, 376–383 (2023). https://doi.org/10.1007/s11596-023-2701-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-023-2701-1