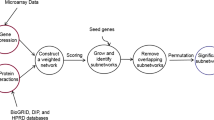
Summary
Traumatic brain injury (TBI) is a common injury caused by external forces that lead to damaged brain function or pathological changes in the brain tissue. To explore the molecular mechanism and the hub genes of TBI, we downloaded gene expression profiles of the TBI model of rat and the sham control for the subsequent gene set enrichment analysis, pathway analysis and protein-protein interactions analysis. The results of Gene Ontology and Kyoto Encyclopedia of Genes and Genomes analysis indicated that multiple biological pathways, including immune response, inflammatory response and cellular response to interleukin-1, as well as signaling pathways, such as tumor necrosis factor signaling pathway, chemokine signaling pathway, cytokine-cytokine receptor interaction, Toll-like receptor signaling pathway and nuclear factor kappa B signaling pathway were implicated in the TBI. In conclusion, this study provides insights into the molecular mechanism of TBI by screening the differentially expressed genes and hub genes that can be used as biomarkers and therapeutic targets.
Similar content being viewed by others

References
Menon DK, Schwab K, Wright DW, et al. Position statement: definition of traumatic brain injury. Arch Phys Med Rehabil, 2010,91(11):1637–1640
Maas AIR, Menon DK, Adelson PD, et al. Traumatic brain injury: integrated approaches to improve prevention, clinical care, and research. Lancet Neurol, 2017,16(12):987–1048
Hinzman JM, Thomas TC, Burmeister JJ, et al. Diffuse brain injury elevates tonic glutamate levels and potassium-evoked glutamate release in discrete brain regions at two days post-injury: an enzyme-based microelectrode array study. J Neurotrauma, 2010,27(5):889–899
Nikolian VC, Dekker SE, Bambakidis T, et al. Improvement of Blood-Brain Barrier Integrity in Traumatic Brain Injury and Hemorrhagic Shock Following Treatment With Valproic Acid and Fresh Frozen Plasma. Crit Care Med, 2018,46(1):e59–e66
Loane DJ, Faden AI. Neuroprotection for traumatic brain injury: translational challenges and emerging therapeutic strategies. Trends Pharmacol Sci, 2010,31(12):596–604
Hui J, Liu R, Zhang H, et al. Screening and identification of critical biomarkers in erectile dysfunction: evidence from bioinformatic analysis. PeerJ, 2020,8:e8653
Shojo H, Borlongan CV, Mabuchi T. Genetic and Histological Alterations Reveal Key Role of Prostaglandin Synthase and Cyclooxygenase 1 and 2 in Traumatic Brain Injury-Induced Neuroinflammation in the Cerebral Cortex of Rats Exposed to Moderate Fluid Percussion Injury. Cell Transplant, 2017,26(7):1301–1313
Barrett T, Troup DB, Wilhite SE, et al. NCBI GEO: archive for functional genomics data sets—10 years on. Nucleic Acids Res, 2011,39:D1005–D1010
Shojo H, Kaneko Y, Mabuchi T, et al. Genetic and histologic evidence implicates role of inflammation in traumatic brain injury-induced apoptosis in the rat cerebral cortex following moderate fluid percussion injury. Neuroscience, 2010,171(4):1273–1282
Huber W, Carey VJ, Gentleman R, et al. Orchestrating high-throughput genomic analysis with Bioconductor. Nat Methods, 2015,12(2):115–121
Blake JA, Chan J, Kishore R, et al. Gene Ontology Consortium: going forward. Nucleic Acids Research, 2015,43:D1049–D1056
Kanehisa M, Goto S, Furumichi M, et al. KEGG for representation and analysis of molecular networks involving diseases and drugs. Nucleic Acids Res, 2010, 38: D355–D360
Kotera M, Hirakawa M, Tokimatsu T, et al. The KEGG databases and tools facilitating omics analysis: latest developments involving human diseases and pharmaceuticals. Methods Mol Biol, 2012,802:19–39
Huang DW, Sherman BT, Tan Q, et al. DAVID Bioinformatics Resources: expanded annotation database and novel algorithms to better extract biology from large gene lists. Nucleic Acids Res, 2007,35: W169–W175
Huang DW, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc, 2008,4(1):44–57
Szklarczyk D, Franceschini A, Wyder S, et al. STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res, 2015,43:D447–D452
Shannon P. Cytoscape: A Software Environment for Integrated Models of Biomolecular Interaction Networks. Genome Res, 2003,13(11):2498–2504
Godoy DA, Lubillo S, Rabinstein AA. Pathophysiology and Management of Intracranial Hypertension and Tissular Brain Hypoxia After Severe Traumatic Brain Injury: An Integrative Approach. Neurosurg Clin N Am, 2018,29(2):195–212
Monson Kenneth L, Converse Matthew I, Manley Geoffrey T. Cerebral blood vessel damage in traumatic brain injury. Clin Biomech (Bristol Avon), 2019,64:98–113
Anghinah R, Amorim RLO, Paiva WS, et al. Traumatic brain injury pharmacological treatment: recommendations. Arquivos de Neuro-Psiquiatria, 2018, 76(2):100–103
Abou El Fadl MH, O’Phelan KH. Management of Traumatic Brain Injury: An Update. Neurosurg Clin N Am, 2018,29(2):213–221
Goodwin S, Mcpherson JD, Mccombie WR. Coming of age: ten years of next-generation sequencing technologies. Nat Rev Genet, 2016,17(6):333–351
van Dam S, Craig T, de Magalhñes JP. GeneFriends: a human RNA-seq-based gene and transcript co-expression database. Nucleic Acids Res, 2015,43:D1124–D1132
Miao M, De Clercq E, Li G. Clinical significance of chemokine receptor antagonists. Expert Opin Drug Metab Toxicol, 2020,16(1):11–30
Woodcock T, Morganti-Kossmann MC. The role of markers of inflammation in traumatic brain injury. Front Neurol, 2013,4:18
Förstner P, Rehman R, Anastasiadou S, et al. Neuroinflammation after Traumatic Brain Injury Is Enhanced in Activating Transcription Factor 3 Mutant Mice. J Neurotrauma, 2018,35(19):2317–2329
Semple BD, Bye N, Rancan M, et al. Role of CCL2 (MCP-1) in traumatic brain injury (TBI): evidence from severe TBI patients and CCL2-/- mice. J Cereb Blood Flow Metab, 2010,30(4):769–782
Woodcock TM, Frugier T, Nguyen TT, et al. The scavenging chemokine receptor ACKR2 has a significant impact on acute mortality rate and early lesion development after traumatic brain injury. PLoS One, 2017,12:e0188305
Semple BD, Bye N, Ziebell JM, et al. Deficiency of the chemokine receptor CXCR2 attenuates neutrophil infiltration and cortical damage following closed head injury. Neurobiol Dis, 2010,40(2):394–403
Ziebell JM, Morganti-Kossmann MC. Involvement of pro- and anti-inflammatory cytokines and chemokines in the pathophysiology of traumatic brain injury. Neurotherapeutics, 2010,7(1):22–30
Wang Q, Tang XN, Yenari MA. The inflammatory response in stroke. J Neuroimmunol, 2007,184(1–2):53–68
Tuttolomondo A, Pecoraro R, Pinto A. Studies of selective TNF inhibitors in the treatment of brain injury from stroke and trauma: a review of the evidence to date. Drug Des Devel Ther, 2014,8:2221–38
Feldman N, Rotter-Maskowitz A, Okun E. DAMPs as mediators of sterile inflammation in aging-related pathologies. Ageing Res Rev, 2015,24(Pt A):29–39
Chiu CC, Liao YE, Yang LY, et al. Neuroinflammation in animal models of traumatic brain injury. J Neurosci Methods, 2016,272:38–49
Hua F, Ma J, Ha T, et al. Activation of Toll-like receptor 4 signaling contributes to hippocampal neuronal death following global cerebral ischemia/reperfusion. J Neuroimmunol, 2007,190(1–2):101–111
Yu N, Hu S, Hao Z. Benificial Effect of Stachydrine on the Traumatic Brain Injury Induced Neurodegeneration by Attenuating the Expressions of Akt/mTOR/PI3K and TLR4/NFκ-B Pathway. Transl Neurosci, 2018,9:175–182
Simon DW, McGeachy MJ, Bayir H, et al. The far-reaching scope of neuroinflammation after traumatic brain injury. Nat Rev Neurol, 2017,13(3):171–191
Knoblach SM, Fan L, Faden AI. Early neuronal expression of tumor necrosis factor-alpha after experimental brain injury contributes to neurological impairment. J Neuroimmunol, 1999,95(1–2):115–125
Pachter JS, De Vries HE, Fabry Z. The Blood-Brain Barrier and Its Role in Immune Privilege in the Central Nervous System. J Neuropathol Exp Neurol, 2003,62(6):593–604
Sebire G, Hery C, Peudenier S, et al. Adhesion proteins on human microglial cells and modulation of their expression by IL1α and TNFα. Res Virol, 1993,144(1):47–52
Chodobski A, Zink BJ, Szmydynger-Chodobska J. Blood-Brain Barrier Pathophysiology in Traumatic Brain Injury. Transl Stroke Res, 2011,2(4):492–516
Pleines UE, Stover JF, Kossmann T, et al. Soluble ICAM-1 in CSF coincides with the extent of cerebral damage in patients with severe traumatic brain injury. J Neurotrauma, 1998,15(6):399–409
Habgood MD, Bye N, Dziegielewska KM, et al. Changes in blood-brain barrier permeability to large and small molecules following traumatic brain injury in mice. Eur J Neurosci, 2007,25(1):231–238
Hua F, Wang J, Ishrat T, et al. Genomic profile of Toll-like receptor pathways in traumatically brain-injured mice: effect of exogenous progesterone. J Neuroinflammation, 2011,8:42
Zhu HT, Bian C, Yuan JC, et al. Curcumin attenuates acute inflammatory injury by inhibiting the TLR4/MyD88/NF-κB signaling pathway in experimental traumatic brain injury. J Neuroinflammation, 2014,11:59
Lucas K and Maes M. Role of the Toll Like Receptor (TLR) Radical Cycle in Chronic Inflammation: Possible Treatments Targeting the TLR4 Pathway. Mol Neurobiol, 2013,48(1):190–204
Park C, Cho IH, Kim D, et al. Toll-like receptor 2 contributes to glial cell activation and heme oxygenase-1 expression in traumatic brain injury. Neurosci Lett, 2008,431(2):123–128
Yu ZQ, Zha JH. Genetic ablation of toll-like receptor 2 reduces secondary brain injury caused by cortical contusion in mice. Ann Clin Lab Sci, 2012,42(1):26–33
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
There are no conflicts of interest.
Additional information
This work was supported by the Military Logistics Scientific Research Project (No. CLB20J027).
Rights and permissions
About this article
Cite this article
Cao, Xy., Qian, X., Liu, Gd. et al. Bioinformatics-based Identification of Key Pathways and Hub Genes of Traumatic Brain Injury in a Rat Model. CURR MED SCI 41, 610–617 (2021). https://doi.org/10.1007/s11596-021-2365-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-021-2365-7