Summary
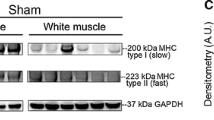
In order to investigate the role of the Notch signaling pathway in skeletal muscle fibrosis after nerve injury, 60 Sprague-Dawley rats were selected and divided randomly into a control and two experimental groups. Group A served as controls without any treatment. Rats in groups B were injected intraperitoneally with 0.2 mL PBS and those in group C were injected intraperitoneally with 0.2 mL PBS+100 μmol/L, 0.2 mL N-[N-(3,5-difluorophenacetyl)-l-alanyl]- S-phenylglycine t-butyl ester (DAPT, a gamma-secretase inhibitor that suppresses Notch signaling) respectively, on postoperative days 1, 3, 7, 10, and 14 in a model of denervation-induced skeletal muscle fibrosis by right sciatic nerve transection. Five rats from each group were euthanized on postoperative days 1, 7, 14, and 28 to collect the right gastrocnemii, and hematoxylin and eosin (HE) staining, immunohistochemistry test, real-time PCR, and Western blotting were performed to assess connective tissue hyperplasia and fibroblast density as well as expression of Notch 1, Jagged 1, and Notch downstream molecules Hes 1 and collagen I (COL I) on day 28. There was no significant difference in HE-stained fibroblast density between group B and C on postoperative day 1. However, fibroblast density was significantly higher in group B than in group C on postoperative days 7, 14, and 28. Notch 1, Jagged 1, Hes 1, and COL I proteins in the gastrocnemius were expressed at very low levels in group A but at high levels in group B. Expression levels of these proteins were significantly lower in group C than in group B (P<0.05), but they were higher in group C than in group A (P<0.05) on postoperative day 28. We are led to conclude that locking the Notch signaling pathway inhibits fibrosis progression of denervated skeletal muscle. Thus, it may be a new approach for treatment of fibrosis of denervated skeletal muscle.
Similar content being viewed by others
References
Wu X, Cong XB, Huang QS, et al. Transposition of branches of radial nerve innervating supinator to posterior interosseous nerve for functional reconstruction of finger and thumb extension in 4 patients with middle and lower trunk root avulsion injuries of brachial plexus. J Huazhong Univ Sci Technolog Med Sci, 2017, 37(6):933–937
Bielesz B, Sirin Y, Si H, et al. Epithelial Notch signaling regulates interstitial fibrosis development in the kidneys of mice and humans. J Clin Investig, 2010, 120(11):4040–4054
Chadwick N, Zeef L, Portillo V, et al. Identification of novel Notch target genes in T cell leukaemia. Mol Cancer, 2010, 45(3):201–209
Chen Y, Zheng S, Qi D, et al. Inhibition of Notch signaling by a γ-secretase inhibitor attenuates hepatic fibrosis in rats. PLoS One, 2012,7(10):e46512
Chigurupati S, Arumugam TV, Son TG, et al. Involvement of notch signaling in wound healing. PLoS One, 2007,2(11):e1167
Dees C, Tomcik M, Zerr P, et al. Notch signalling regulates fibroblast activation and collagen release in systemic sclerosis. Ann Rheum Dis, 2011, 70(7):1304–1310
Dees C, Zerr P, Tomcik M, et al. Inhibition of Notch signaling prevents experimental fibrosis and induces regression of established fibrosis. Arthritis Rheumatol, 2011, 63(5):1396–1404
Gazave E, Lapébie P, Richards GS, et al. Origin and evolution of the Notch signalling pathway: an overview from eukaryotic genomes. BMC Evol Biol, 2009,9:249
Han AC, Deng JX, Huang QS, et al. Verapamil inhibits scar formation after peripheral nerve repair in vivo. Neural Regen Res, 2016, 11(3):508–511
Kavian N, Servettaz A, Mongaret C, et al. Argeting ADAM-17/notch signaling abrogates the development of systemic sclerosis in a murine model. Arthritis Rheum, 2010, 62(11):3477–3487
Khuong HT, Midha R. Advances in nerve repair. Curr Neurol Neurosci Rep, 2013,13(1):322
Liu MZ, Zhang WM, Zhu WQ. Advances in the study of nerve transplantation for the prevention of denervated skeletal muscular atrophy. J Tissue Eng Reconstr Surg, 2006,2:238–240
Nemir M, Metrich M, Plaisance I, et al. The Notch pathway controls fibrotic and regenerative repair in the adult heart. Eur Heart J, 2012, 35(32):2174–2185
Plantier L, Crestani B, Wert SE, et al. Ectopic respiratory epithelial cell differentiation in bronchiolised distal airspaces in idiopathic pulmonary fibrosis. Thorax, 2011, 66(8):651–657
Russell JL, Goetsch SC, Gaiano NR, et al. A Dynamic Notch Injury Response Activates Epicardium and Contributes to Fibrosis Repair Novelty and Significance. Circ Res, 2011, 108(1):51–59
Sörensen-Zender I, Rong S, Susnik N, et al. Renal tubular Notch signaling triggers a prosenescent state after acute kidney injury. Am J Physiol Renal Physiol, 2014, 306(8):907–915
Butko E, Pouget C, Traver D. Complex regulation of HSC emergence by the Notch signaling pathway. Dev Biol, 2016, 409(1):129–138
Logan GJ, Wright MC, Kubicki AC. et al. Notch pathway signaling in the skin antagonizes Merkel cell development. Dev Biol, 2018, 434(2):207–214
Liu Y, Huang G, Mo B, et al. Artesunate ameliorates lung fibrosis via inhibiting the Notch signaling pathway. Exp Ther Med, 2017, 14(1):561–566
Lou YL, Guo F, Liu F, et al. MiR-210 activates notch signaling pathway in angiogenesisinduced by cerebral ischemia. Mol Cell Biochem, 2012, 37(8):45–51
Yabuuchi S, Pai SG, Campbell NR, et al. Notch signaling pathway targeted therapy suppresses tumor progression and metastatic spread in pancreatic cancer. Cancer Lett, 2013, 335(1):41–51
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Feng, F., Shan, L., Deng, Jx. et al. Role of the Notch Signaling Pathway in Fibrosis of Denervated Skeletal Muscle. CURR MED SCI 39, 419–425 (2019). https://doi.org/10.1007/s11596-019-2053-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-019-2053-z