Summary
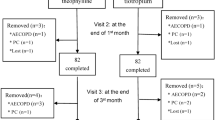
This study compared the efficacy and safety of tiotropium bromide inhalation powder (spiriva) and doxofylline oral tablet (doxofylline) in the treatment of chronic obstructive pulmonary disease (COPD). A multi-center, randomized, double-blind, double-dummy, parallel-controlled study involved 127 eligible stable moderate to severe COPD patients treated with inhaled tiotropium dry powder (18 μg/day) or oral doxofylline tablets (0.2 g/time, 2 times a day) for 12 and 24 weeks. Before and after treatment for 12 weeks and 24 weeks, respectively, pulmonary function, 6-min walking distance and dyspnea index were recorded. The results showed that in both tiotropium group and doxofylline groups, after 12-week treatment, FEV1, FEV1/FVC% and 6-min walk distance were significantly higher than those before the medication, while dyspnea index decreased as compared with that before treatment. After 24-week treatment, a slight improvement in the measures was observed as compared with that of 12-weeks treatment, but the difference was not statistically significant. With both 12-week and 24-week treatment, the effect of tiotropium was slightly better than that of doxofylline tablets, with the difference being statistically insignificant. The major adverse events in the tiotropium group and doxofylline group were observed in 9 cases (9.9%) and 12 cases (12.9%), respectively, and no statistically significant difference was found between them. We are led to conclude that both tiotropium at 18 μg a day and doxofylline tablets at 0.2 g/day (two times a day) are effective and safe for the treatment of COPD.
Similar content being viewed by others
References
Raherison C, Girodet PO. Epidemiology of COPD. Eur Respir Rev, 2009,18(114):213–221
Pauwels RA, Buist AS, Calverley PM, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary. Am J Respir Crit Care Med, 2001,163(5):1256–1276
Vagaggini B, Nieri D, Malagrinò L, et al. Acute administration of bronchodilators on exercise tolerance in treated COPD patients. Pulm Pharmacol Ther, 2011,24(1):49–54
Kaplan A. Effect of tiotropium on quality of life in COPD: a systematic review. Prim Care Respir J, 2010,19(4): 315–325
Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease: NHLBI/WHO workshop report. Available at: www.goldcopd.com. Accessed February 8, 2006.
Chen AM, Bollmeier SG, Finnegan PM. Long-acting bronchodilator therapy for the treatment of chronic obstructive pulmonary disease. Ann Pharmacother, 2008, 42(12):1832–1842
Hanania NA, Sharafkhaneh A. Update on the pharmacologic therapy for chronic obstructive pulmonary disease. Clin Chest Med, 2007,28(3):589–607
Casaburi R, Mahler DA, Jones PW, et al. A long-term evaluation of once-daily inhaled tiotropium in chronic obstructive pulmonary disease. Eur Respir J, 2002; 19(2):217–224
Vincken W, van Noord JA, Greefhorst AP, et al. Improved health outcomes in patients with COPD during 1 year’s treatment with tiotropium. Eur Respir J, 2002,19(2): 209–216
O’Donnell DE, Fluge T, Gerken F, et al. Effects of tiotropium on lung hyperinflation, dyspnoea and exercise tolerance in COPD. Eur Respir J, 2004,23(6):832–840
Wang QY, Zhang H, Wang XQ, et al. Randomized double- blind double-dummy controlled clinical trial of tiotropium bromide in the treament of chronic obstructive pulmonary disease. Chin J Clin Pharmacol, 2005,21(5): 328–331
Zhang JP, Kang J, Cai BQ, et al. Comparsion of tiotropium inhalation capcules and ipratropium metered dose inhaler in a randomized, double-blind, double-dummy, efficacy and safety study in patients with chronic obstructive pulmonary disease. Chin J Tuberc Respir Dis, 2006, 29(6):363–367
Culpitt SV, de Matos C, Russell RE, et al. Effect of theophylline on induced sputum inflammatory indices and neutrophil chemotaxis in chronic obstructive pulmonary disease. Am J Respir Crit Care Med, 2002,165(10): 1371–1376
Barnes PJ. Theophylline: new perspectives for an old drug. Am J Respir Crit Care Med, 2003,167(6):813–818
Zhou YM, Wang XP, Zeng XY, et al. Theophylline in the treatment of chronic obstructive pulmonary disease: a randomized, double-blind, placebo-controlled study. Chin J Tuberc Respir Dis, 2006,29(9):577–582
Goldstein MF, Chervinsky P. Efficacy and safety of doxofylline compared to theophylline in chronic reversible asthma: a double-blind randomized placebo-controlled muhieentre clinical trial. Med Sci Monit, 2002,8(4): CR297–CR304
Mamary AJ, Criner GJ. Tiotropium bromide for chronic obstructive pulmonary disease. Expert Rev Respir Med, 2009,3(3):211–220
Tashkin DP. Long-acting anticholinergic use in chronic obstructive pulmonary disease: efficacy and safety. Curr Opin Pulm Med, 2010,16(2):97–105
Troosters T, Celli B, Lystig T, et al. Tiotropium as a first maintenance drug in COPD: secondary analysis of the UPLIFT trial. Eur Respir J, 2010,36(1):65–73
Michele TM, Pinheiro S, Iyasu S. The safety of tiotropium—the FDA’s conclusions. N Engl J Med, 2010, 363(12):1097–1099
Littner MR, Ilowite JS, Tashkin DP, et al. Long-acting bronchodilation with once-daily dosing of tiotropium (Spiriva) in stable chronic obstructive pulmonary disease. Am J Respir Crit Care Med, 2000,161(4 Pt 1):1136–1142
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by grants from the Clinical Chronic Respiratory Diseases Research Foundation of Chinese Medical Association (No. 07010030011), Key Research Program of the Ministry of Health of China (No. 2007353), the Special Funds for Scientific Research on Public Causes of Ministry of Health of China (No. 201002008).
Rights and permissions
About this article
Cite this article
Wang, T., Luo, G., Hu, Y. et al. Comparative study on the efficacy of tiotropium bromide inhalation and oral doxofylline treatment of moderate to severe stable chronic obstructive pulmonary disease. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 31, 614–618 (2011). https://doi.org/10.1007/s11596-011-0570-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-011-0570-5