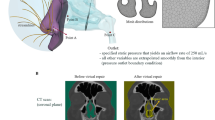
Abstract
Purpose
A deviated nasal septum is the most common etiology for nasal airway obstruction (NAO), and septoplasty is the most common surgical procedure performed by ear–nose–throat surgeons in adults. However, quantitative criteria are rarely adopted to select patients for surgery, which may explain why up to 50% of patients report persistent or recurrent symptoms of nasal obstruction postoperatively. This study reports a systematic virtual surgery method to identify patients who may benefit from septoplasty.
Methods
One patient with symptoms of NAO due to a septal deviation was selected to illustrate the virtual surgery concept. Virtual septoplasty was implemented in three steps: (1) determining if septal geometry is abnormal preoperatively, (2) virtually correcting the deviation while preserving the anatomical shape of the septum, and (3) estimating the post-surgical improvement in airflow using computational fluid dynamics. Anatomical and functional changes predicted by the virtual surgery method were compared to a standard septoplasty performed independently from the computational analysis.
Results
A benchmark healthy nasal septum geometry was obtained by averaging the septum dimensions of 47 healthy individuals. A comparison of the nasal septum geometry in the NAO patient with the benchmark geometry identified the precise locations where septal deviation and thickness exceeded the healthy range. Good agreement was found between the virtual surgery predictions and the actual surgical outcomes for both airspace minimal cross-sectional area (0.05 cm2 pre-surgery, 0.54 cm2 virtual surgery, 0.50 cm2 actual surgery) and nasal resistance (0.91 Pa.s/ml pre-surgery, 0.08 Pa.s/ml virtual surgery, 0.08 Pa.s/ml actual surgery).
Conclusions
Previous virtual surgery methods for NAO were based on manual edits and subjective criteria. The virtual septoplasty method proposed in this study is objective and has the potential to be fully automated. Future implementation of this method in virtual surgery planning software has the potential to improve septoplasty outcomes.








Similar content being viewed by others
References
Hildebrandt T, Bruning JJ, Lamecker H, Zachow S, Heppt WJ, Schmidt N, Goubergrits L (2019) Digital analysis of nasal airflow facilitating decision support in rhinosurgery. Facial Plast Surg 35(1):3–8. https://doi.org/10.1055/s-0039-1677720
Quammen CW, Taylor RM 2nd, Krajcevski P, Mitran S, Enquobahrie A, Superfine R, Davis B, Davis S, Zdanski C (2016) The virtual pediatric airways workbench. Stud Health Technol Inform 220:295–300
Vanhille DL, Garcia GJM, Asan O, Borojeni AAT, Frank-Ito DO, Kimbell JS, Pawar SS, Rhee JS (2018) Virtual surgery for the nasal airway: a preliminary report on decision support and technology acceptance. JAMA Facial Plast Surg 20(1):63–69. https://doi.org/10.1001/jamafacial.2017.1554
Sanmiguel-Rojas E, Burgos MA, Esteban-Ortega F (2018) Nasal surgery handled by CFD tools. Int J Numer Methods Biomed Eng. https://doi.org/10.1002/cnm.3126
Roblin DG, Eccles R (2002) What, if any, is the value of septal surgery? Clin Otolaryngol Allied Sci 27(2):77–80
Sundh C, Sunnergren O (2015) Long-term symptom relief after septoplasty. Eur Arch Oto-Rhino-Laryngol 272(10):2871–2875. https://doi.org/10.1007/s00405-014-3406-7
Bhattacharyya N (2010) Ambulatory sinus and nasal surgery in the United States: demographics and perioperative outcomes. Laryngoscope 120(3):635–638. https://doi.org/10.1002/lary.20777
Jones NS, Strobl A, Holland I (1997) A study of the CT findings in 100 patients with rhinosinusitis and 100 controls. Clin Otolaryngol Allied Sci 22(1):47–51
Min YG, Jung HW, Kim CS (1995) Prevalence study of nasal septal deformities in Korea: results of a nation-wide survey. Rhinology 33(2):61–65
Garcia GJ, Rhee JS, Senior BA, Kimbell JS (2010) Septal deviation and nasal resistance: an investigation using virtual surgery and computational fluid dynamics. Am J Rhinol Allergy 24(1):e46-53. https://doi.org/10.2500/ajra.2010.24.3428
Holmstrom M (2010) The use of objective measures in selecting patients for septal surgery. Rhinology 48(4):387–393. https://doi.org/10.4193/Rhino10.072
Andre RF, Vuyk HD, Ahmed A, Graamans K, Nolst Trenite GJ (2009) Correlation between subjective and objective evaluation of the nasal airway. A systematic review of the highest level of evidence. Clin Otolaryngol 34(6):518–525. https://doi.org/10.1111/j.1749-4486.2009.02042.x
Borojeni AAT, Garcia GJM, Moghaddam MG, Frank-Ito DO, Kimbell JS, Laud PW, Koenig LJ, Rhee JS (2020) Normative ranges of nasal airflow variables in healthy adults. Int J Comput Assist Radiol Surg 15(1):87–98. https://doi.org/10.1007/s11548-019-02023-y
Stewart MG, Witsell DL, Smith TL, Weaver EM, Yueh B, Hannley MT (2004) Development and validation of the nasal obstruction symptom evaluation (NOSE) scale. Otolaryngol Head Neck Surg 130(2):157–163. https://doi.org/10.1016/j.otohns.2003.09.016
Kimbell JS, Frank DO, Laud P, Garcia GJ, Rhee JS (2013) Changes in nasal airflow and heat transfer correlate with symptom improvement after surgery for nasal obstruction. J Biomech 46(15):2634–2643. https://doi.org/10.1016/j.jbiomech.2013.08.007
Sullivan CD, Garcia GJ, Frank-Ito DO, Kimbell JS, Rhee JS (2014) Perception of better nasal patency correlates with increased mucosal cooling after surgery for nasal obstruction. Otolaryngol Head Neck Surg 150(1):139–147. https://doi.org/10.1177/0194599813509776
Rhee JS, Sullivan CD, Frank DO, Kimbell JS, Garcia GJ (2014) A systematic review of patient-reported nasal obstruction scores: defining normative and symptomatic ranges in surgical patients. JAMA Facial Plast Surg 16(3):219–225. https://doi.org/10.1001/jamafacial.2013.2473(quiz 232)
Frank-Ito DO, Kimbell JS, Laud P, Garcia GJM, Rhee JS (2014) Predicting postsurgery nasal physiology with computational modeling: current challenges and limitations. Otolaryngol Head Neck Surg 151(5):751–759. https://doi.org/10.1177/0194599814547497
Kelly JT, Prasad AK, Wexler AS (2000) Detailed flow patterns in the nasal cavity. J Appl Physiol 89(1):323–337. https://doi.org/10.1152/jappl.2000.89.1.323
Li C, Jiang J, Dong H, Zhao K (2017) Computational modeling and validation of human nasal airflow under various breathing conditions. J Biomech 64:59–68. https://doi.org/10.1016/j.jbiomech.2017.08.031
Frank-Ito DO, Wofford M, Schroeter JD, Kimbell JS (2016) Influence of mesh density on airflow and particle deposition in sinonasal airway modeling. J Aerosol Med Pulm Drug Deliv 29(1):46–56. https://doi.org/10.1089/jamp.2014.1188
Garcia GJ, Schroeter JD, Segal RA, Stanek J, Foureman GL, Kimbell JS (2009) Dosimetry of nasal uptake of water-soluble and reactive gases: a first study of interhuman variability. Inhal Toxicol 21(7):607–618. https://doi.org/10.1080/08958370802320186
Garcia GJM, Hariri BM, Patel RG, Rhee JS (2016) The relationship between nasal resistance to airflow and the airspace minimal cross-sectional area. J Biomech 49(9):1670–1678. https://doi.org/10.1016/j.jbiomech.2016.03.051
Manoukian PD, Wyatt JR, Leopold DA, Bass EB (1997) Recent trends in utilization of procedures in otolaryngology-head and neck surgery. Laryngoscope 107(4):472–477
Han JK, Stringer SP, Rosenfeld RM, Archer SM, Baker DP, Brown SM, Edelstein DR, Gray ST, Lian TS, Ross EJ, Seiden AM, Setzen M, Tollefson TT, Ward PD, Welch KC, Wise SK, Nnacheta LC (2015) Clinical consensus statement: septoplasty with or without inferior turbinate reduction. Otolaryngol Head Neck Surg 153(5):708–720. https://doi.org/10.1177/0194599815606435
van Egmond M, Rovers MM, Tillema AHJ, van Neerbeek N (2018) Septoplasty for nasal obstruction due to a deviated nasal septum in adults: a systematic review. Rhinology 56(3):195–208. https://doi.org/10.4193/Rhin18.016
van Egmond M, Rovers MM, Hannink G, Hendriks CTM, van Heerbeek N (2019) Septoplasty with or without concurrent turbinate surgery versus non-surgical management for nasal obstruction in adults with a deviated septum: a pragmatic, randomised controlled trial. Lancet 394(10195):314–321. https://doi.org/10.1016/S0140-6736(19)30354-X
van Egmond M, van Heerbeek N, Ter Haar ELM, Rovers MM (2017) Clinimetric properties of the Glasgow health status inventory, Glasgow benefit inventory, peak nasal inspiratory flow, and 4-phase rhinomanometry in adults with nasal obstruction. Rhinology 55(2):126–134. https://doi.org/10.4193/Rhin16.296
Carney AS, Bateman ND, Jones NS (2000) Reliable and reproducible anterior active rhinomanometry for the assessment of unilateral nasal resistance. Clin Otolaryngol Allied Sci 25(6):499–503
Clarke RW, Cook JA, Jones AS (1995) The effect of nasal mucosal vasoconstriction on nasal airflow sensation. Clin Otolaryngol Allied Sci 20(1):72–73
Burgos MA, Sanmiguel-Rojas E, Singh N, Esteban-Ortega F (2018) DigBody((R)): a new 3D modeling tool for nasal virtual surgery. Comput Biol Med 98:118–125. https://doi.org/10.1016/j.compbiomed.2018.05.016
Hemtiwakorn K, Mahasitthiwat V, Tungjitkusolmun S, Hamamoto K, Pintavirooj C (2015) Patient-specific aided surgery approach of deviated nasal septum using computational fluid dynamics. IEEJ Trans Electr Electron Eng 10(3):274–286. https://doi.org/10.1002/tee.22084
Frank-Ito DO, Kimbell JS, Borojeni AAT, Garcia GJM, Rhee JS (2019) A hierarchical stepwise approach to evaluate nasal patency after virtual surgery for nasal airway obstruction. Clin Biomech (Bristol, Avon) 61:172–180. https://doi.org/10.1016/j.clinbiomech.2018.12.014
Gu JT, Kaplan S, Greenfield S, Calloway H, Wong BJF (2019) Validation of a septoplasty deformity grading system for the evaluation of nasal obstruction. Laryngoscope 129(3):586–593. https://doi.org/10.1002/lary.27365
Flanagan P, Eccles R (1997) Spontaneous changes of unilateral nasal airflow in man. A re-examination of the ‘nasal cycle’. Acta Oto-Laryngol 117(4):590–595. https://doi.org/10.3109/00016489709113443
Gaberino C, Rhee JS, Garcia GJ (2017) Estimates of nasal airflow at the nasal cycle mid-point improve the correlation between objective and subjective measures of nasal patency. Respir Physiol Neurobiol 238:23–32. https://doi.org/10.1016/j.resp.2017.01.004
Patel RG, Garcia GJ, Frank-Ito DO, Kimbell JS, Rhee JS (2015) Simulating the nasal cycle with computational fluid dynamics. Otolaryngol Head Neck Surg 152(2):353–360. https://doi.org/10.1177/0194599814559385
Funding
This study was funded by Grant R01EB009557 from the National Institutes of Health/National Institute of Biomedical Imaging and Bioengineering to the Medical College of Wisconsin (MCW) and by subcontract from MCW to the University of North Carolina at Chapel Hill and Duke University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures were performed in accordance with the ethical standards of the institutional review boards at The Medical College of Wisconsin and Marquette University and of the 1964 Helsinki Declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Moghaddam, M.G., Garcia, G.J.M., Frank-Ito, D.O. et al. Virtual septoplasty: a method to predict surgical outcomes for patients with nasal airway obstruction. Int J CARS 15, 725–735 (2020). https://doi.org/10.1007/s11548-020-02124-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-020-02124-z