Abstract
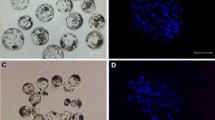
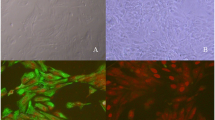
In the present study, nuclear transferred embryos (NTEs) were reconstructed by using pig fetal fibroblasts as donors and in vitro matured oocytes as recipients. The effects of G418 selection on donor cells, duration of IVM of prepubertal gilt oocytes and oxygen tension in IVM of oocytes were investigated. The results were as follows: (i) When G418 selected cells expressing GFP were used as donors, the cleavage rate of NTEs decreased drastically in comparison to NTEs derived from donors without antibiotic selection (45.7% vs. 71.6%, p<0.05). For the blastocyst rate, no significant difference was observed between two groups (10% vs. 10.4%, p>0.05). (ii) The rate of nuclear maturation of oocytes increased significantly when IVM duration time was extended from 36h to 42h (83.6% vs. 96.7%, p<0.05). However, no statistical difference were observed between NTEs derived from oocytes of 36 h IVM group and NTEs from oocytes of 42 h IVM group in the rates of cleavage (59.3% vs. 73.6%, p>0.05) and blastocyst formation (9.3% vs. 13.2%, p>0.05); (iii) no significant difference was observed between NTEs reconstructed from oocytes matured under lower oxygen (7% O2) tension and NTEs derived from oocytes matured under higher oxygen tension (20% O2) in cleavage rate (70.6% vs. 67.1%, p>0.05) and blastocyst rate (11.8% vs. 12.8%, p>0.05). These results suggest that: (i) G418 selection does not have a significant effect on cleavage rate of NTEs expressing GFP. (ii) Nuclear maturation is greatly improved by prolonging IVM duration from 36 to 42 h, while no significant differences were observed for developmental potential of transgenic embryos. Thus IVM 42 h is the better choice in order to obtain maximum number of MII oocytes as recipients. (iii) Lower oxygen tension and higher oxygen tension in IVM have no significant effect on development of cloned embryos.
Similar content being viewed by others
References
Cibelli, J. B., Stice, S. L., Golueke, P. J. et al., Cloned transgenic calves produced from nonquiescent fetal fibroblasts, Science, 1998, 280(5367): 1256–1258.
Schnieke, A. E., Kind, A. J., Ritchie, W. A. et al., Human factor IX transgenic sheep produced by transfer of nuclei from transfected fetal fibroblasts, Science, 1997, 278(5346): 2130–2133.
Campbell, K. H., A background to nuclear transfer and its applications in agriculture and human therapeutic medicine, J. Anat., 2002, 200(Pt3): 267–275.
Lai, L., Kolber-Simonds, D., Park, K. W. et al., Production of alpha-1,3-galactosyltransferase knockout pigs by nuclear transfer cloning, Science, 2002, 295(5557): 1089–1092.
Dai, Y., Vaught, T. D., Boone, J. et al., Targeted disruption of the alpha1,3-galactosyltransferase gene in cloned pigs, Nat. Biotechnol., 2002, 20(3): 251–255.
Phelps, C. J., Koike, C., Vaught, T. D. et al., Production of alpha 1,3-galactosyltransferase-deficient pigs, Science, 2003, 299(5605): 411–414.
Kolber-Simonds, D., Lai, L., Watt, S. R. et al., Production of alpha-1,3-galactosyltransferase null pigs by means of nuclear transfer with fibroblasts bearing loss of heterozygosity mutations, Proc. Natl. Acad. Sci. USA, 2004, 101(19): 7335–7340.
Guo, J., An, Z., Li, Y. et al., Cloned goats (Capra hircus) from adult ear cells, Science in China, Ser. C, 2002, 45(3): 260–267.
Zou, X., Chen, Y., Wang, Y. et al., Production of cloned goats from enucleated oocytes injected with cumulus cell nuclei or fused with cumulus cells, Cloning, 2001, 3(1): 31–37.
Chen, D. Y., Li, J. S., Han, Z. M. et al., Somatic cell bovine cloning: Effect of donor cell and recipients, Chinese Science Bulletin, 2003, 48(6): 549–554.
Gong, G., Dai, Y., Fan, B. et al., Birth of calves expressing the enhanced green fluorescent protein after transfer of fresh or vitrified/thawed blastocysts produced by somatic cell nuclear transfer, Mol. Reprod. Dev., 2004, 69(3): 278–288.
Zhou, Q., Renard, J. P., Le Friec, G. et al., Generation of fertile cloned rats by regulating oocyte activation, Science, 2003, 302(5648): 1179.
Wilmut, I., Beaujean, N., de Sousa, P. A. et al., Somatic cell nuclear transfer, Nature, 2002, 419(6907): 583–586.
Lai, L., Prather, R. S., Creating genetically modified pigs by using nuclear transfer, Reprod. Biol. Endocrinol., 2003, 1(1): 82–87.
Machaty, Z., Bondioli, K. R., Ramsoondar, J. J. et al., The use of nuclear transfer to produce transgenic pigs, Cloning Stem Cells, 2002, 4(1): 21–27.
Miyoshi, K., Rzucidlo, S. J., Pratt, S. L. et al., Utility of rapidly matured oocytes as recipients for production of cloned embryos from somatic cells in the pig, Biol. Reprod., 2002, 67(2): 540–545.
Coy, P., Romar, R., In vitro production of pig embryos: a point of view, Reprod. Fertil. Dev., 2002, 14(5–6): 275–286.
Gong, G., Dai, Y., Fan, B. et al., Production of transgenic blastocyst by nuclear transfer from different types of somatic cells in cattle, Science in China, Ser. C, 2004, 47(2): 183–189.
Hyun, S., Lee, G., Kim, D. et al., Production of nuclear transfer-derived piglets using porcine fetal fibroblasts transfected with the enhanced green fluorescent protein, Biol. Reprod., 2003, 69(3): 1060–1068.
Chen, D., Sun, Q., Liu, J. et al., The giant panda (Ailuropoda melanoleuca) somatic nucleus can dedifferentiate in rabbit ooplasm and support early development of the reconstructed egg, Science in China, Ser. C, 1999,42(4): 346–353.
Park, K. W., Cheong, H. T., Lai, L. et al., Production of nuclear transfer-derived swine that express the enhanced green fluorescent protein, Anim. Biotechnol., 2001, 12(2): 173–181.
Yang, X., Tian, X. C., Dai, Y. et al., Transgenic farm animals: Applications in agriculture and biomedicine, Biotechnol. Annu. Rev., 2000, 5: 269–292.
Uhm, S. J., Kim, N. H., Kim, T. et al., Expression of enhanced green fluorescent protein (EGFP) and neomycin resistant (Neo(R)) genes in porcine embryos following nuclear transfer with porcine fetal fibroblasts transfected by retrovirus vector, Mol. Reprod. Dev., 2000, 57(4): 331–337.
Karja, N. W., Wongsrikeao, P., Murakami, M. et al., Effects of oxygen tension on the development and quality of porcine in vitro fertilized embryos, Theriogenology, 2004, 62(9): 1585–1595.
Iwamoto, M., Onishi, A., Fuchimoto, D. et al., Low oxygen tension during in vitro maturation of porcine follicular oocytes improves parthenogenetic activation and subsequent development to the blastocyst stage, Theriogenology, 2005, 63(5): 1277–1289.
Park, J. I., Hong, J. Y., Yong, H. Y. et al., High oxygen tension during in vitro oocyte maturation improves in vitro development of porcine oocytes after fertilization, Anim. Reprod. Sci., 2005, 87(1–2): 133–141.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Y., Pan, D., Sun, X. et al. Production of porcine cloned transgenic embryos expressing green fluorescent protein by somatic cell nuclear transfer. SCI CHINA SER C 49, 1–8 (2006). https://doi.org/10.1007/s11427-005-0071-5
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11427-005-0071-5