Abstract
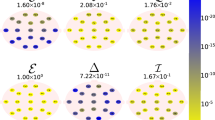
Aging is the inevitable biological process that results in a progressive structural and functional decline associated with alterations in the resting/task-related brain activity, morphology, plasticity, and functionality. In the present study, we analyzed the effects of physiological aging on the human brain through entropy measures of electroencephalographic (EEG) signals. One hundred sixty-one participants were recruited and divided according to their age into young (n = 72) and elderly (n = 89) groups. Approximate entropy (ApEn) values were calculated in each participant for each EEG recording channel and both for the total EEG spectrum and for each of the main EEG frequency rhythms: delta (2–4 Hz), theta (4–8 Hz), alpha 1 (8–11 Hz), alpha 2 (11–13 Hz), beta 1 (13–20 Hz), beta 2 (20–30 Hz), and gamma (30–45 Hz), to identify eventual statistical differences between young and elderly. To demonstrate that the ApEn represents the age-related brain changes, the computed ApEn values were used as features in an age-related classification of subjects (young vs elderly), through linear, quadratic, and cubic support vector machine (SVM). Topographic maps of the statistical results showed statistically significant difference between the ApEn values of the two groups found in the total spectrum and in delta, theta, beta 2, and gamma. The classifiers (linear, quadratic, and cubic SVMs) revealed high levels of accuracy (respectively 93.20 ± 0.37, 93.16 ± 0.30, 90.62 ± 0.62) and area under the curve (respectively 0.95, 0.94, 0.93). ApEn seems to be a powerful, very sensitive–specific measure for the study of cognitive decline and global cortical alteration/degeneration in the elderly EEG activity.



Similar content being viewed by others
Data Availability
The data that support the findings of this study are available on request from the corresponding author.
References
Rose MA. Evolutionary biology of aging. New York: Oxford Univ. Press; 1991.
Rose MR, Flatt T, Graves JL, Greer LF, Martinez DE, Matos M, et al. What is aging? Front Genet. 2012;3:134.
Fjell AM, Walhovd KB. Structural brain changes in aging: courses, causes and cognitive consequences. Rev Neurosci. 2010;21(3):187–221.
Angelova DM, Brown DR. Microglia and the aging brain: are senescent microglia the key to neurodegeneration? J Neurochem. 2019;151(6):676–88 (12).
Hou Y, Dan X, Babbar M, Wei Y, Hasselbalch SG, Croteau DL, et al. Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol. 2019;15(10):565–81 (10).
Grimm A, Eckert A. Brain aging and neurodegeneration: from a mitochondrial point of view. J Neurochem. 2017;143(4):418–31 (11).
Kumral D, Şansal F, Cesnaite E, Mahjoory K, Al E, Gaebler M, et al. BOLD and EEG signal variability at rest differently relate to aging in the human brain. Neuroimage. 2020;15(207):116373 (02).
Vecchio F, Babiloni C, Lizio R, Fallani Fde V, Blinowska K, Verrienti G, et al. Resting state cortical EEG rhythms in Alzheimer’s disease: toward EEG markers for clinical applications: a review. Suppl Clin Neurophysiol. 2013;62:223–36.
Roca-Stappung M, Fernández T, Becerra J, Mendoza-Montoya O, Espino M, Harmony T. Healthy aging: relationship between quantitative electroencephalogram and cognition. Neurosci Lett. 2012;510(2):115–20.
Rossini PM, Del Percio C, Pasqualetti P, Cassetta E, Binetti G, Dal Forno G, et al. Conversion from mild cognitive impairment to Alzheimer’s disease is predicted by sources and coherence of brain electroencephalography rhythms. Neurosci. 2006;143(3):793–803.
Vecchio F, Miraglia F, Bramanti P, Rossini PM. Human brain networks in physiological aging: a graph theoretical analysis of cortical connectivity from EEG data. J Alzheimers Dis. 2014;41(4):1239–49.
Rossini PM, Di Iorio R, Granata G, Miraglia F, Vecchio F. From mild cognitive impairment to Alzheimer’s disease: a new perspective in the “land” of human brain reactivity and connectivity. J Alzheimers Dis. 2016;53(4):1389–93.
Miraglia F, Vecchio F, Pappalettera C, Nucci L, Cotelli M, Judica E, et al. Brain connectivity and graph theory analysis in Alzheimer’s and Parkinson’s disease: the contribution of electrophysiological techniques. Brain Sci. 2022;18(3):12.
Bein B. Entropy. Best Pract Res Clin Anaesthesiol. 2006;20(1):101–9.
Feutrill A, Roughan M. A review of Shannon and differential entropy rate estimation. Entropy (Basel). 2021;23(8).
Keshmiri S. Entropy and the brain: an overview. Entropy (Basel). 2020;22(9).
Carhart-Harris RL. The entropic brain - revisited. Neuropharmacol. 2018;11(142):167–78.
Takahashi T, Cho RY, Murata T, Mizuno T, Kikuchi M, Mizukami K, et al. Age-related variation in EEG complexity to photic stimulation: a multiscale entropy analysis. Clin Neurophysiol. 2009;120(3):476–83.
McIntosh AR, Vakorin V, Kovacevic N, Wang H, Diaconescu A, Protzner AB. Spatiotemporal dependency of age-related changes in brain signal variability. Cereb Cortex. 2014;24(7):1806–17.
Mohamed AMA, Uçan ON, Bayat O, Duru AD. Classification of resting-state status based on sample entropy and power spectrum of electroencephalography (EEG). Appl Bionics Biomech. 2020;2020:8853238.
Wang Z. Initiative AsDN Brain entropy mapping in healthy aging and Alzheimer’s disease. Front Aging Neurosci. 2020;12:596122.
Alù F, Orticoni A, Judica E, Cotelli M, Rossini PM, Miraglia F, et al. Entropy modulation of electroencephalographic signals in physiological aging. Mech Ageing Dev. 2021;06(196):111472.
Pappalettera C, Miraglia F, Cotelli M, Rossini PM, Vecchio F. Analysis of complexity in the EEG activity of Parkinson’s disease patients by means of approximate entropy. Geroscience. 2022 Mar 28.
Vecchio F, Miraglia F, Pappalettera C, Orticoni A, Alù F, Judica E, et al. Entropy as measure of brain networks’ complexity in eyes open and closed conditions. Symmetry. 2021;13(11):2178.
Javaid H, Kumarnsit E, Chatpun S. Age-related alterations in EEG network connectivity in healthy aging. Brain Sci. 2022;12(2).
Salmaso D, Longoni AM. Problems in the assessment of hand preference. Cortex. 1985;21(4):533–49.
Miraglia F, Vecchio F, Rossini PM. Brain electroencephalographic segregation as a biomarker of learning. Neural Netw. 2018;106:168–74.
Vecchio F, Miraglia F, Judica E, Cotelli M, Alù F, Rossini PM. Human brain networks: a graph theoretical analysis of cortical connectivity normative database from EEG data in healthy elderly subjects. Gerosci. 2020.
Miraglia F, Vecchio F, Marra C, Quaranta D, Alù F, Peroni B, et al. Small world index in default mode network predicts progression from mild cognitive impairment to dementia. Int J Neural Syst. 2020;30(2):2050004.
Jung TP, Makeig S, Humphries C, Lee TW, McKeown MJ, Iragui V, et al. Removing electroencephalographic artifacts by blind source separation. Psychophysiol. 2000;37(2):163–78.
Iriarte J, Urrestarazu E, Valencia M, Alegre M, Malanda A, Viteri C, et al. Independent component analysis as a tool to eliminate artifacts in EEG: a quantitative study. J Clin Neurophysiol. 2003 2003 Jul-Aug;20(4):249–57.
Vecchio F, Pappalettera C, Miraglia F, Deinite G, Manenti R, Judica E, Caliandro P, Rossini PM. Prognostic Role of Hemispherical Functional Connectivity in Stroke: A Study via Graph Theory Versus Coherence of Electroencephalography Rhythms. Stroke. 2022.
Vecchio F, Miraglia F, Iberite F, Lacidogna G, Guglielmi V, Marra C, et al. Sustainable method for Alzheimer dementia prediction in mild cognitive impairment: electroencephalographic connectivity and graph theory combined with apolipoprotein E. Ann Neurol. 2018;84(2):302–14 (08).
Vecchio F, Nucci L, Pappalettera C, Miraglia F, Iacoviello D, Rossini PM. Time-frequency analysis of brain activity in response to directional and non-directional visual stimuli: an event related spectral perturbations (ERSP) study. J Neural Eng. 2022.
Sun R, Wong WW, Wang J, Tong RK. Changes in electroencephalography complexity using a brain computer interface-motor observation training in chronic stroke patients: a fuzzy approximate entropy analysis. Front Hum Neurosci. 2017;11:444.
Abásolo D, Hornero R, Espino P, Poza J, Sánchez CI, de la Rosa R. Analysis of regularity in the EEG background activity of Alzheimer’s disease patients with approximate entropy. Clin Neurophysiol. 2005;116(8):1826–34.
Burioka N, Miyata M, Cornélissen G, Halberg F, Takeshima T, Kaplan DT, et al. Approximate entropy in the electroencephalogram during wake and sleep. Clin EEG Neurosci. 2005;36(1):21–4.
Lee GM, Fattinger S, Mouthon AL, Noirhomme Q, Huber R. Electroencephalogram approximate entropy influenced by both age and sleep. Front Neuroinform. 2013;7:33.
Montesinos L, Castaldo R, Pecchia L. On the use of approximate entropy and sample entropy with centre of pressure time-series. J Neuroeng Rehabil. 2018;15(1):116 (12).
Pincus SM. Approximate entropy as a measure of system complexity. Proc Natl Acad Sci U S A. 1991;88(6):2297–301.
Yan R, Gao RX. Approximate entropy as a diagnostic tool for machine health monitoring. Mech Syst Signal Process. 2007;21:824–39.
Natal J, Ávila I, Tsukahara VB, Pinheiro M, Maciel CD. Entropy: from thermodynamics to information processing. Entropy (Basel). 2021;23(10).
Carhart-Harris RL, Leech R, Hellyer PJ, Shanahan M, Feilding A, Tagliazucchi E, et al. The entropic brain: a theory of conscious states informed by neuroimaging research with psychedelic drugs. Front Hum Neurosci. 2014;8:20.
Craik A, He Y, Contreras-Vidal JL. Deep learning for electroencephalogram (EEG) classification tasks: a review. J Neural Eng. 2019;16(3):031001 (06).
Byvatov E, Fechner U, Sadowski J, Schneider G. Comparison of support vector machine and artificial neural network systems for drug/nondrug classification. J Chem Inf Comput Sci. 2003;43(6):1882–9.
Peltier SJ, Lisinski JM, Noll DC, LaConte SM. Support vector machine classification of complex fMRI data. Annu Int Conf IEEE Eng Med Biol Soc. 2009;2009:5381–4.
Kang J, Han X, Song J, Niu Z, Li X. The identification of children with autism spectrum disorder by SVM approach on EEG and eye-tracking data. Comput Biol Med. 2020;05(120):103722.
Shoker L, Sanei S, Chambers J. Artifact removal from electroencephalograms using a hybrid BSS-SVM algorithm. IEEE Signal Process Letters. 2005;12(10):721–4 (10/01).
Cortes C, Vapnik V. Support-vector networks. Mach Learn. 1995;20(3):273–97.
Platt J. Sequential minimal optimization: a fast algorithm for training support vector machines. Microsoft; 1998.
Arti Patle and Deepak Singh C. SVM kernel functions for classification. 2013 International Conference on Advances in Technology and Engineering (ICATE). 2013:1–9.
Ji N, Ma L, Dong H, Zhang X. EEG signals feature extraction based on DWT and EMD combined with approximate entropy. Brain Sci. 2019;9(8).
Sahiner B, Chan HP, Petrick N, Wagner RF, Hadjiiski L. Feature selection and classifier performance in computer-aided diagnosis: the effect of finite sample size. Med Phys. 2000;27(7):1509–22.
Derek Pisner and David MS. Support vector machine. Machine Learning. 2020.
Vecchio F, Miraglia F, Alù F, Menna M, Judica E, Cotelli M, et al. Classification of Alzheimer’s disease respect to physiological aging with innovative EEG biomarkers in a machine learning implementation. (in press). J Alzheimer Dis. 2020.
Vecchio F, Miraglia F, Alù F, Judica E, Cotelli M, Pellicciari MC, et al. Human brain networks in physiological and pathological aging: reproducibility of electroencephalogram graph theoretical analysis in cortical connectivity. Brain Connect. 2022;12(1):41–51.
Zhang Y. Support vector machine classification algorithm and its application. In: Liu C, Wang L, Yang A, editors. Information Computing and Applications. Berlin, Heidelberg: Springer Berlin Heidelberg 2012;179–86.
Kreuzer M, Stern MA, Hight D, Berger S, Schneider G, Sleigh JW, et al. Spectral and entropic features are altered by age in the electroencephalogram in patients under sevoflurane anesthesia. Anesthesiol. 2020;132(5):1003–16 (05).
Nowak K, Costa-Faidella J, Dacewicz A, Escera C, Szelag E. Altered event-related potentials and theta oscillations index auditory working memory deficits in healthy aging. Neurobiol Aging. 2021;12(108):1–15.
Spironelli C, Angrilli A. EEG delta band as a marker of brain damage in aphasic patients after recovery of language. Neuropsychologia. 2009;47(4):988–94.
Ishii R, Canuet L, Aoki Y, Hata M, Iwase M, Ikeda S, et al. Healthy and pathological brain aging: from the perspective of oscillations, functional connectivity, and signal complexity. Neuropsychobiol. 2017;75(4):151–61.
Rossini PM, Di Iorio R, Bentivoglio M, Bertini G, Ferreri F, Gerloff C, et al. Methods for analysis of brain connectivity: an IFCN-sponsored review. Clin Neurophysiol. 2019;130(10):1833–58.
Barry RJ, Clarke AR, Johnstone SJ, Magee CA, Rushby JA. EEG differences between eyes-closed and eyes-open resting conditions. Clin Neurophysiol. 2007;118(12):2765–73.
Desai R, Tailor A, Bhatt T. Effects of yoga on brain waves and structural activation: a review. Complement Ther Clin Pract. 2015;21(2):112–8.
Wang R, Wang J, Yu H, Wei X, Yang C, Deng B. Decreased coherence and functional connectivity of electroencephalograph in Alzheimer’s disease. Chaos. 2014;24(3):033136.
Güntekin B, Başar E. Gender differences influence brain’s beta oscillatory responses in recognition of facial expressions. Neurosci Lett. 2007;424(2):94–9.
Vysata O, Kukal J, Prochazka A, Pazdera L, Valis M. Age-related changes in the energy and spectral composition of EEG. Neurophysiol. 2012;44(1):63–7.
Jabès A, Klencklen G, Ruggeri P, Antonietti JP, Banta Lavenex P, Lavenex P. Age-related differences in resting-state EEG and allocentric spatial working memory performance. Front Aging Neurosci. 2021;13:704362.
Vecchio F, Miraglia F, Quaranta D, Granata G, Romanello R, Marra C, et al. Cortical connectivity and memory performance in cognitive decline: a study via graph theory from EEG data. Neurosci. 2016;316:143–50.
Miraglia F, Vecchio F, Rossini PM. Searching for signs of aging and dementia in EEG through network analysis. Behav Brain Res. 2017;01(317):292–300.
Vecchio F, Miraglia F, Piludu F, Granata G, Romanello R, Caulo M, et al. “Small World” architecture in brain connectivity and hippocampal volume in Alzheimer’s disease: a study via graph theory from EEG data. Brain Imaging Behav. 2017;11(2):473–85.
Palva JM, Monto S, Kulashekhar S, Palva S. Neuronal synchrony reveals working memory networks and predicts individual memory capacity. Proc Natl Acad Sci U S A. 2010;107(16):7580–5.
Palva S, Monto S, Palva JM. Graph properties of synchronized cortical networks during visual working memory maintenance. Neuroimage. 2010;49(4):3257–68.
Subasi A. EEG signal classification using wavelet feature extraction and a mixture of expert model. Expert Syst Appl. 2007;32(4):1084–93.
Yuvaraj R, Murugappan M. Hemispheric asymmetry non-linear analysis of EEG during emotional responses from idiopathic Parkinson’s disease patients. Cogn Neurodyn. 2016;10(3):225–34.
Sharmila A, Aman Raj S, Shashank P, Mahalakshmi P. Epileptic seizure detection using DWT-based approximate entropy, Shannon entropy and support vector machine: a case study. J Med Eng Technol. 2018;42(1):1–8.
Stefan S, Schorr B, Lopez-Rolon A, Kolassa IT, Shock JP, Rosenfelder M, et al. Consciousness indexing and outcome prediction with resting-state EEG in severe disorders of consciousness. Brain Topogr. 2018;31(5):848–62 (09).
Acknowledgements
This work was partially supported by the Italian Ministry of Health for Institutional Research (Ricerca corrente) and by Toto Holding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Pappalettera, C., Cacciotti, A., Nucci, L. et al. Approximate entropy analysis across electroencephalographic rhythmic frequency bands during physiological aging of human brain. GeroScience 45, 1131–1145 (2023). https://doi.org/10.1007/s11357-022-00710-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-022-00710-4