Abstract
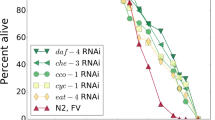
We present an initial molecular characterization of a morphological transition between two early aging states. In previous work, an age score reflecting physiological age was developed using a machine classifier trained on images of worm populations at fixed chronological ages throughout their lifespan. The distribution of age scores identified three stable post-developmental states and transitions. The first transition occurs at day 5 post-hatching, where a significant percentage of the population exists in both state I and state II. The temperature dependence of the timing of this transition (Q 10 ~ 1.17) is too low to be explained by a stepwise process with an enzymatic or chemical rate-limiting step, potentially implicating a more complex mechanism. Individual animals at day 5 were sorted into state I and state II groups using the machine classifier and analyzed by microarray expression profiling. Despite being isogenic, grown for the same amount of time, and indistinguishable by eye, these two morphological states were confirmed to be molecularly distinct by hierarchical clustering and principal component analysis of the microarray results. These molecular differences suggest that pharynx morphology reflects the aging state of the whole organism. Our expression profiling yielded a gene set that showed significant overlap with those from three previous age-related studies and identified several genes not previously implicated in aging. A highly represented group of genes unique to this study is involved in targeted ubiquitin-mediated proteolysis, including Skp1-related (SKR), F-box-containing, and BTB motif adaptors.




Similar content being viewed by others
References
Benjamin H (1947) Biologic versus chronologic age. J Gerontol 2(3):217–227
Ben-Zvi A, Miller EA, Morimoto RI (2009) Collapse of proteostasis represents an early molecular event in Caenorhabditis elegans aging. Proc Natl Acad Sci U S A 106(35):14914–14919
Budovskaya YV et al (2008) An elt-3/elt-5/elt-6 GATA transcription circuit guides aging in C. elegans. Cell 134(2):291–303
Cheadle C et al (2003) Analysis of microarray data using Z score transformation. J Mol Diagn: JMD 5(2):73–81
Chow DK et al (2006) Sarcopenia in the Caenorhabditis elegans pharynx correlates with muscle contraction rate over lifespan. Exp Gerontol 41(3):252–260
Dillin A et al (2002) Rates of behavior and aging specified by mitochondrial function during development. Science 298(5602):2398–2401
Durieux J, Wolff S, Dillin A (2011) The cell-non-autonomous nature of electron transport chain-mediated longevity. Cell 144(1):79–91
Eide EJ et al (2005) Control of mammalian circadian rhythm by CKIε-regulated proteasome-mediated PER2 degradation. Mol Cell Biol 25(7):2795–2807
Feng J, Bussière F, Hekimi S (2001) Mitochondrial electron transport is a key determinant of life span in Caenorhabditis elegans. Dev Cell 1(5):633–644
Friedman DB, Johnson TE (1988a) A mutation in the age-1 gene in Caenorhabditis elegans lengthens life and reduces hermaphrodite fertility. Genetics 118(1):75–86
Friedman DB, Johnson TE (1988b) Three mutants that extend both mean and maximum life span of the nematode, Caenorhabditis elegans, define the age-1 gene. J Gerontol 43(4):B102–109
Gems D, McElwee JJ (2005) Broad spectrum detoxification: the major longevity assurance process regulated by insulin/IGF-1 signaling? Mech Ageing Dev 126(3):381–387
Ghazi A, Henis-Korenblit S, Kenyon C (2007) Regulation of Caenorhabditis elegans lifespan by a proteasomal E3 ligase complex. Proc Natl Acad Sci 104(14):5947–5952
Golden TR, Melov S (2004) Microarray analysis of gene expression with age in individual nematodes. Aging Cell 3(3):111–124
Golden TR et al (2008) Age-related behaviors have distinct transcriptional profiles in Caenorhabditis elegans. Aging Cell 7(6):850–865
Harman D (1956) Aging: a theory based on free radical and radiation chemistry. J Gerontol 11(3):298–300
Hong CI, Conrad ED, Tyson JJ (2007) A proposal for robust temperature compensation of circadian rhythms. Proc Natl Acad Sci 104(4):1195–1200
Huang DW, Sherman BT, Lempicki RA (2009a) Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res 37(1):1–13
Huang DW, Sherman BT, Lempicki RA (2009b) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4(1):44–57
Hunt T, Sassone-Corsi P (2007) Riding tandem: circadian clocks and the cell cycle. Cell 129(3):461–464
Johnson TE (1987) Aging can be genetically dissected into component processes using long-lived lines of Caenorhabditis elegans. Proc Natl Acad Sci U S A 84(11):3777–3781
Johnston J et al (2008) Quantitative image analysis reveals distinct structural transitions during aging in Caenorhabditis elegans tissues. PLoS One 3(7):e2821
Kenyon C et al (1993) A C. elegans mutant that lives twice as long as wild type. Nature 366(6454):461–464
Kim S, Yu H (2011) Mutual regulation between the spindle checkpoint and APC/C. Semin Cell Dev Biol 22(6):551–558
Kirkwood TB, Rose MR (1991) Evolution of senescence: late survival sacrificed for reproduction. Philosophical transactions of the royal society of London. Ser B Biol Sci 332(1262):15–24
Kirstein-Miles J, Morimoto RI (2010) Caenorhabditis elegans as a model system to study intercompartmental proteostasis: interrelation of mitochondrial function, longevity, and neurodegenerative diseases. Dev Dyn: An Official Publ of the American Assoc of Anatomists 239(5):1529–1538
Lee SS et al (2003) A systematic RNAi screen identifies a critical role for mitochondria in C. elegans longevity. Nat Genet 33(1):40–48
Li W et al (2007) RLE-1, an E3 ubiquitin ligase, regulates C. elegans aging by catalyzing DAF-16 polyubiquitination. Dev Cell 12(2):235–246
Lund J et al (2002) Transcriptional profile of aging in C. elegans. Curr Biol 12(18):1566–1573
McElwee JJ et al (2004) Shared transcriptional signature in Caenorhabditis elegans Dauer larvae and long-lived daf-2 mutants implicates detoxification system in longevity assurance. J Biol Chem 279(43):44533–44543
Murphy CT et al (2003) Genes that act downstream of DAF-16 to influence the lifespan of Caenorhabditis elegans. Nature 424(6946):277–283
Nicholas HR, Hodgkin J (2004) Responses to infection and possible recognition strategies in the innate immune system of Caenorhabditis elegans. Mol Immunol 41(5):479–493
Oh SW et al (2006) Identification of direct DAF-16 targets controlling longevity, metabolism and diapause by chromatin immunoprecipitation. Nat Genet 38(2):251–257
Oliveros, JC, 2007. Available at: http://bioinfogp.cnb.csic.es/tools/venny/index.html
Orlov N et al (2008) WND-CHARM: multi-purpose image classification using compound image transforms. Pattern Recognit Lett 29(11):1684–1693
Park S-K, Tedesco PM, Johnson TE (2009) Oxidative stress and longevity in Caenorhabditis elegans as mediated by SKN-1. Aging Cell 8(3):258–269
Pincus Z, Slack FJ (2008) Transcriptional (dys)regulation and aging in Caenorhabditis elegans. Genome Biol 9(9):233
Pincus Z, Slack FJ (2010) Developmental biomarkers of aging in Caenorhabditis elegans. Dev Dyn: An Official Publ of the American Association of Anatomists 239(5):1306–1314
Pittendrigh CS (1960) Circadian rhythms and the circadian organization of living systems. Cold Spring Harb Symp Quant Biol 25:159–184
Rea SL et al (2005) A stress-sensitive reporter predicts longevity in isogenic populations of Caenorhabditis elegans. Nat Genet 37(8):894–898
Ruoff P, Rensing L (2004) Temperature effects on circadian clocks. J Therm Biol 29(7–8):445–456
Sánchez-Blanco A, Kim SK (2011) Variable pathogenicity determines individual lifespan in Caenorhabditis elegans. PLoS Genet 7(4):e1002047
Schuster E et al (2010) DamID in C. elegans reveals longevity-associated targets of DAF-16/FoxO. Mol Syst Biol 6:399
Shamir L, Wolkow CA, Goldberg IG (2009) Quantitative measurement of aging using image texture entropy. Bioinformatics (Oxford, England) 25(23):3060–3063
Simm A, Johnson TE (2010) Biomarkers of ageing: a challenge for the future. Exp Gerontol 45(10):731–732
Simonetta SH et al (2009) Timing of locomotor activity circadian rhythms in Caenorhabditis elegans. PLoS One 4(10):e7571
Walker RF (2011) Developmental theory of aging revisited: focus on causal and mechanistic links between development and senescence. Rejuvenation Res 14(4):429–436
Weissman A (1891) The duration of life. In Weissman on Heredity. Oxford University Press, London
Whittenberger JL (1956) The nature of the response to stress with aging. Bull N Y Acad Med 32(5):329–336
Williams GC (1957) Pleiotropy, natural selection and the evolution of senescence. Evolution 11(4):398–411
Wu D et al (2006) Visualizing hidden heterogeneity in isogenic populations of C. elegans. Exp Gerontol 41(3):261–270
Xu L et al (2003) BTB proteins are substrate-specific adaptors in an SCF-like modular ubiquitin ligase containing CUL-3. Nature 425(6955):316–321
Yu S, Driscoll M (2011) EGF signaling comes of age: promotion of healthy aging in C. elegans. Exp Gerontol 46(2-3):129–34
Acknowledgements
This work was supported entirely by the Intramural Research Program of the National Institutes of Health, National Institute on Aging.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Esm 1
(PDF 7 kb)
About this article
Cite this article
Eckley, D.M., Rahimi, S., Mantilla, S. et al. Molecular characterization of the transition to mid-life in Caenorhabditis elegans . AGE 35, 689–703 (2013). https://doi.org/10.1007/s11357-012-9401-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-012-9401-2