Abstract
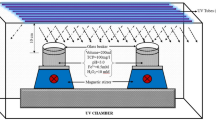
Industrial units based on chemical processes—the textile and paper industries—are major sources of chlorophenols in the environment, and chlorophenolic compounds persist within the environment for a long time with high toxicity levels. The photo-assisted Fenton’s and photocatalysis processes were investigated for the degradation of chlorophenols in the present study. Response surface methodology was employed to get optimised conditions for photocatalysis and photo-Fenton process-governing factors, thus, yielding a profound removal efficiency. Under optimised conditions, with a photocatalyst dose of 0.2 g/L, oxidant concentration of 10.0 mM and pH 5.0, complete removal of 2,4-dichlorophenol (2,4-DCP) was observed in 210 minutes in photocatalytic treatment. In the case of the photo-Fenton process, at an H2O2 dose of 5.0 mM and Fe2+ concentration of 0.5 mM, the organic pollutant was eliminated within 5 minutes of reaction time under acidic conditions (pH 3.0). The RSM model reported the perfect fit of experimental data with the predicted response. Among different isotherm models, the Langmuir isotherm was the best fit. The process followed pseudo-first order rate kinetics among various kinetics models. For the obtained optimised conditions, sonication and solar energy-driven processes were incorporated to study enhanced mineralisation. The solar-assisted Fenton process reported maximum mineralisation (90%) and cost-effective ($0.01/litre for 100 mg/L 2,4-DCP) treatment among different hybrid oxidation processes. The work provides insight into harnessing the naturally available solar energy, reducing the overall treatment cost and opting for a sustainable treatment method.















Similar content being viewed by others
Data Availability
Necessary data has been provided along with this manuscript.
References
Abdelwahab O, Amin NK (2013) Adsorption of phenol from aqueous solutions by Luffa cylindrica fibers: kinetics, isotherm and thermodynamic studies. Egypt J Aquatic Res 39(4):215–223. https://doi.org/10.1016/j.ejar.2013.12.011
Ahmadzadeh S, Dolatabadi M (2018) In situ generation of hydroxyl radical for efficient degradation of 2, 4-dichlorophenol from aqueous solutions. Environ Monit Assess 190:1–11. https://doi.org/10.1007/s10661-018-6697-0
Ameta SC, Ameta R (eds) (2018) Advanced oxidation processes for wastewater treatment: emerging green chemical technology. Academic press
Anandan S, Ponnusamy VK, Ashokkumar M (2020) A review on hybrid techniques for the degradation of organic pollutants in aqueous environment. Ultrason Sonochem 67:105130. https://doi.org/10.1016/j.ultsonch.2020.105130
Bayarri B, Gimenez J, Curco D, Esplugas S (2005) Photocatalytic degradation of 2, 4-dichlorophenol by TiO2/UV: kinetics, actinometries and models. Catal Today 101(3-4):227–236. https://doi.org/10.1016/j.cattod.2005.03.019
Chu W, Choy WK, So TY (2007) The effect of solution pH and peroxide in the TiO2-induced photocatalysis of chlorinated aniline. J Hazard Mater 141(1):86–91. https://doi.org/10.1016/j.jhazmat.2006.06.093
Faludi T, Balogh C, Serfőző Z, Molnár-Perl I (2015) Analysis of phenolic compounds in the dissolved and suspended phases of Lake Balaton water by gas chromatography-tandem mass spectrometry. Environ Sci Pollut Res 22:11966–11974. https://doi.org/10.1007/s11356-015-4734-x
Field JA, Sierra-Alvarez R (2008) Microbial degradation of chlorinated phenols. Rev Environ Sci Biotechnol 7(3):211–241. https://doi.org/10.1007/s11157-007-9124-5
Freundlich H (1906) Concerning adsorption in solutions. J PhysChem-Stochiometrie Verwandtschaftslehre 57A:385–470
Ge M, Hu Z, Wei J, He Q, He Z (2021) Recent advances in persulfate-assisted TiO2-based photocatalysis for wastewater treatment: performances, mechanism and perspectives. J Alloys Compd 888:161625. https://doi.org/10.1016/j.jallcom.2021.161625
Ghoneim MM, El-Desoky HS, Zidan NM (2011) Electro-Fenton oxidation of sunset yellow FCF azo-dye in aqueous solutions. Desalination 274(1-3):22–30. https://doi.org/10.1016/j.desal.2011.01.062
Gong X, Wang H, Yang C, Li Q, Chen X, Hu J (2015) Photocatalytic degradation of high ammonia concentration wastewater by TiO2. Future Cities Environ 1(1):1–12. https://doi.org/10.1186/s40984-015-0012-9
Hoffmann MR, Martin ST, Choi W, Bahnemann DW (1995) Environmental applications of semiconductor photocatalysis. Chem Rev 95(1):69–96. https://doi.org/10.1021/cr00033a004
Igbinosa EO, Odjadjare EE, Chigor VN, Igbinosa IH, Emoghene AO, Ekhaise FO, Igiehon NO, Idemudia OG (2013) Toxicological profile of chlorophenols and their derivatives in the environment: the public health perspective. Sci World J. https://doi.org/10.1155/2013/460215
Kansal SK, Chopra M (2012) Photocatalytic degradation of 2, 6-dichlorophenol in aqueous phase using titania as a photocatalyst. https://doi.org/10.4236/eng.2012.48055
Kansal SK, Kaur G, Singh S (2009) Studies on the photocatalytic degradation of 2, 3-dichlorophenol using different oxidants in aqueous solutions. React Kinet Catal Lett 98:177–186. https://doi.org/10.1007/s11144-009-0058-5
Kavitha V, Palanivelu K (2016) Degradation of phenol and trichlorophenol by heterogeneous photo-Fenton process using granular ferric hydroxide®: comparison with homogeneous system. Int J Environ Sci Technol 13:927–936. https://doi.org/10.1007/s13762-015-0922-y
Lagergren S (1898) About the theory of so-called adsorption of solute substances. Kungliga Svenska Vetenskaps akademiens 24:1–39
Langmuir I (1918) The adsorption of gases on plane surfaces of glass,mica and platinum. J Am Chem Soc 40:1361–1367
Melián EP, Díaz OG, Rodríguez JD, Araña J, Peña JP (2013) Adsorption and photocatalytic degradation of 2, 4-dichlorophenol in TiO2 suspensions. Effect of hydrogen peroxide, sodium peroxodisulphate and ozone. Appl Catal A Gen 455:227–233. https://doi.org/10.1016/j.apcata.2013.02.007
Michałowicz J, Duda W (2007) Phenols-sources and toxicity. Pol J Environ Stud 16(3):347–362
Nagy VD, Zhumakayev A, Vörös M, Bordé Á, Szarvas A, Szűcs A, Kocsubé S, Jakab P, Monostori T, Škrbić BD, Mohai E (2023) Development of a multicomponent microbiological soil inoculant and its performance in sweet potato cultivation. Microorganisms 11(4):914. https://doi.org/10.3390/microorganisms11040914
Ocampo-Perez R, Leyva-Ramos R, Mendoza-Barron J, Guerrero-Coronado RM (2011) Adsorption rate of phenol from aqueous solution onto organobentonite: surface diffusion and kinetic models. J Colloid Interface Sci 364(1):195–204. https://doi.org/10.1016/j.jcis.2011.08.032
Ortiz I, Rivero MJ, Margallo M (2019) Advanced oxidative and catalytic processes. In: Sustainable Water and Wastewater Processing. Elsevier, pp 161–201. https://doi.org/10.1016/B978-0-12-816170-8.00006-5
Othmer CG, Overberger GT (1983) Ultrasonics. In: Seaborg (ed) Kirk-Othmer Encyclopedia of Chemical Technology, vol 23, p 462. https://doi.org/10.1021/j150469a016
Pedrazzani R, Bertanza G, Brnardić I, Cetecioglu Z, Dries J, Dvarionienė J, García-Fernández AJ, Langenhoff A, Libralato G, Lofrano G, Škrbić B (2019) Opinion paper about organic trace pollutants in wastewater: toxicity assessment in a European perspective. Sci Total Environ 651:3202–3221. https://doi.org/10.1016/j.scitotenv.2018.10.027
Pera-Titus M, Garcia-Molina V, Baños MA, Giménez J, Esplugas S (2004) Deradation of chlorophenols by means of advanced oxidation processes: a general review. Appl Catal B Environ 47(4):219–256. https://doi.org/10.1016/j.apcatb.2003.09.010
Pipil H, Yadav S, Chawla H, Taneja S, Verma M, Singla N, Haritash AK (2022) Comparison of TiO2 catalysis and Fenton’s treatment for rapid degradation of Remazol Red dye in textile industry effluent Rendiconti Lincei. Scienze Fisiche e Naturali 33(1):105–114. https://doi.org/10.1007/s12210-021-01040-x
Rout PR, Zhang TC, Bhunia P, Surampalli RY (2021) Treatment technologies for emerging contaminants in wastewater treatment plants: a review. Sci Total Environ 753:141990. https://doi.org/10.1016/j.scitotenv.2020.141990
Sajben-Nagy E, Manczinger L, Škrbić B et al (2014) Characterization of an extracellular laccase of Leptosphaerulina chartarum. World J Microbiol Biotechnol 30:2449–2458. https://doi.org/10.1007/s11274-014-1670-8
Samsudin MFR, Bacho N, Sufian S, Ng YH (2019) Photocatalytic degradation of phenol wastewater over Z-scheme g-C3N4/CNT/BiVO4 heterostructure photocatalyst under solar light irradiation. J Mol Liq 277:977–988. https://doi.org/10.1016/j.molliq.2018.10.160
Shoneye A, Sen Chang J, Chong MN, Tang J (2022) Recent progress in photocatalytic degradation of chlorinated phenols and reduction of heavy metal ions in water by TiO2-based catalysts. Int Mater Rev 67(1):47–64. https://doi.org/10.1080/09506608.2021.1891368
Škrbić B (2007) Organochlorine and organophosphate pesticide residues in wheat varieties from Serbia. Food Addit Contam 24(7):695–703. https://doi.org/10.1080/02652030601182888
Temkin MJ, Pyzhev V (1940) Recent modifications to Langmuir isotherms, Acta Physiochem. USSR 12:217–222
Thind PS, Kumari D, John S (2018) TiO2/H2O2 mediated UV photocatalysis of chlorpyrifos: optimization of process parameters using response surface methodology. J Environ Chem Eng 6(3):3602–3609. https://doi.org/10.1016/j.jece.2017.05.031
USEPA (2004) Edition of the drinking water standards and health advisories. EPA, US, Washington (DC)
Van Aken P, Lambert N, Van den Broeck R, Degrève J, Dewil R (2019) Advances in ozonation and biodegradation processes to enhance chlorophenol abatement in multisubstrate wastewaters: a review. Environ Sci Water Res Technol 5(3):444–481. https://doi.org/10.1039/C8EW00562A
Verma M, Haritash AK (2019) Degradation of amoxicillin by Fenton and Fenton-integrated hybrid oxidation processes. J Environ Chem Eng 7(1):102886. https://doi.org/10.1016/j.jece.2019.102886
Verma M, Haritash AK (2020) Photocatalytic degradation of amoxicillin in pharmaceutical wastewater: a potential tool to manage residual antibiotics. Environ Technol Innov 20:101072. https://doi.org/10.1016/j.eti.2020.101072
Wang P (2017) Aggregation of TiO2 nanoparticles in aqueous media: effects of pH, ferric ion and humic acid. Int J Environ Sci Nat Resour 1(5):157–162. https://doi.org/10.19080/IJESNR.2017.01.555575
Weber R, Gaus C, Tysklind M, Johnston P, Forter M, Hollert H et al (2008) Dioxin-and POP-contaminated sites—contemporary and future relevance and challenges: overview on background, aims and scope of the series. Environ Sci Pollut Res 15:363–393. https://doi.org/10.1007/s11356-008-0024-1
Wei D, Zhao C, Khan A, Sun L, Ji Y, Ai Y, Wang X (2019) Sorption mechanism and dynamic behavior of graphene oxide as an effective adsorbent for the removal of chlorophenol based environmental-hormones: a DFT and MD simulation study. Chem Eng J 375:121964. https://doi.org/10.1016/j.cej.2019.121964
Yadav S, Pipil H, Chawla H, Taneja S, Kumar S, Haritash AK (2022) Textile industry wastewater treatment using eco-friendly techniques. In: Kanwar VS, Sharma SK, Prakasam C (eds) Proceedings of International Conference on Innovative Technologies for Clean and Sustainable Development (ICITCSD – 2021). Springer, Cham. https://doi.org/10.1007/978-3-030-93936-6_6
Yadav S, Kumar S, Haritash AK (2023a) A comprehensive review of chlorophenols: fate, toxicology and its treatment. J Environ Manag 342:118254. https://doi.org/10.1016/j.jenvman.2023.118254
Yadav S, Kumar S, Haritash AK (2023b) Solar light and ultrasound-assisted rapid Fenton’s oxidation of 2,4,6-trichlorophenol: comparison, optimisation, and mineralisation. Rend Fis Acc Lincei. https://doi.org/10.1007/s12210-023-01192-y
Ye Z, Sirés I, Zhang H, Huang YH (2019) Mineralization of pentachlorophenol by ferrioxalate-assisted solar photo-Fenton process at mild pH. Chemosphere 217:475–482. https://doi.org/10.1016/j.chemosphere.2018.10.221
Zhumakayev AR, Vörös M, Szekeres A et al (2021) Comprehensive characterization of stress tolerant bacteria with plant growth-promoting potential isolated from glyphosate-treated environment. World J Microbiol Biotechnol 37:94. https://doi.org/10.1007/s11274-021-03065-8
Zulfiqar M, Samsudin MFR, Sufian S (2019) Modelling and optimization of photocatalytic degradation of phenol via TiO2 nanoparticles: an insight into response surface methodology and artificial neural network. J Photochem Photobiol A Chem 384:112039. https://doi.org/10.1016/j.jphotochem.2019.112039
Acknowledgements
The authors are grateful to the Department of Environmental Engineering at DTU for providing the infrastructure and opportunity for carrying out the present study.
Author information
Authors and Affiliations
Contributions
Harsh Pipil: conceptualization, execution, data compilation and draft writing.
Shivani Yadav: conceptualization, execution, data compilation and draft writing.
Sunil Kumar: conceptualization, supervision, reviewing and editing
Anil Kumar Haritash: conceptualization, supervision, reviewing and editing.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Consent to participate
Authors provide the consent to participate.
Consent for publication
Authors provide the consent to publish the manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pipil, H., Yadav, S., Kumar, S. et al. Synergistic potency of ultrasound and solar energy towards oxidation of 2,4-dichlorophenol: a chemometrics approach. Environ Sci Pollut Res 31, 8186–8209 (2024). https://doi.org/10.1007/s11356-023-31598-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-31598-y