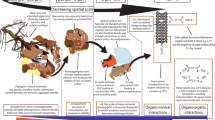
Abstract
Dissolved organic matter (DOM) from biosolids can alter the sorption of orthophosphate (inorganic phosphorus (IP)) to soils and, therefore, affect the bioavailability of IP. It is not clear how clay mineralogy and solution composition interfere with DOM effects on IP sorption by soils. Hence, we studied the effect of DOM on IP sorption to five semi-arid soils dominated by either illite/smectite (I/S) or kaolinite clays. IP sorption isotherms were constructed in either NaCl or CaCl2 background solution, with and without the addition of DOM. The IP sorption capacity maxima (SMAX, Langmuir model) of the I/S soils were 33–102% higher in the presence of CaCl2, as compared to NaCl. Although DOM had no effect on the IP-SMAX in the presence of CaCl2, it increased the IP-SMAX by 35–59% in the presence of the NaCl solution. Surprisingly, DOM sorption to the I/S soils was 30–90% greater in the presence of a Na+-dominated solution, as compared to a Ca2+-dominated solution. In contrast to the I/S soils, the SMAX of the kaolinitic soil was not affected by the background electrolyte (Na+, Ca2+) or the addition of DOM. Furthermore, the addition of IP reduced the sorption of DOM to the kaolinitic soil (by up to 50%) in both background electrolyte solutions. These results highlight the contrasting roles of divalent and monovalent cations in conjunction with DOM in IP sorption to semi-arid I/S soils. We propose a new approach based on two conceptual mechanisms to explain the DOM’s enhancement of IP sorption to I/S soils. (1) Under dispersion conditions in the Na+-dominated solutions, Ca2+-mediated DOM-IP complexes bind to the clay’s negative planar surfaces. (2) Under flocculation conditions in the Ca+-dominated solutions, the distance between adjacent platelets decreases, reducing both the electronegative charge spillover and Ca2+ bridge-mediated DOM sorption. In contrast, the addition of DOM to kaolinite, a multi-platelet clay with a low isomorphic negative charge, reduces IP sorption due to competitive sorption on the clay’s broken edges.




Similar content being viewed by others
Data availability
Upon request.
Abbreviations
- ADSC:
-
Anaerobically digested sewage sludge compost
- DOM/DOC:
-
Dissolved organic matter or carbon
- EC:
-
Electrical conductivity
- EDL:
-
Electrical double layer
- IP:
-
Inorganic phosphorus/orthophosphate
- I/S:
-
Illite/smectite
- PZC:
-
Point of zero charge
- RO:
-
Reverse-osmosed
References
Abdala DB, Ghosh AK, da Silva IR, de Novais RF, Alvarez Venegas VH (2012) Phosphorus saturation of a tropical soil and related P leaching caused by poultry litter addition. Agric Ecosyst Environ 162:15–23. https://doi.org/10.1016/j.agee.2012.08.004
Arai Y, Livi KJT, Sparks DL (2005) Phosphate reactivity in long-term poultry litter-amended southern delaware sandy soils. Soil Sci Soc Amer J 69(3):616–629. https://doi.org/10.2136/sssaj2004.0218
Arnarson TS, Keil RG (2000) Mechanisms of pore water organic matter adsorption to montmorillonite. Mar Chem 71(3–4):309–320. https://doi.org/10.1016/S0304-4203(00)00059-1
Baham J, Sposito G (1994) Adsorption of dissolved organic carbon extracted from sewage sludge on montmorillonite and kaolinite in the presence of metal ions. J Environ Qual 23(1):147–153. https://doi.org/10.2134/jeq1994.00472425002300010023x
Bar-Yosef B, Rosenberg R, Kafkafi U, Sposito G (1988) Phosphorus adsorption by kaolinite and montmorillonite: I. effect of time, ionic strength, and pH. Soil Sci Soc Amer J 52(6):1580–1585. https://doi.org/10.2136/sssaj1988.03615995005200060011x
Beji R, Hamdi W, Kesraoui A, Seffen M (2017) Effects of salts on phosphorus adsorption in alkalize Tunisian soil. Euro-Mediterr J Environ Integr 2(1):1–9. https://doi.org/10.1007/s41207-016-0012-7
Bergaya F, Jaber M, Lambert JF (2011) Clays and clay minerals. Rubber‐Clay Nanocomposites: Science, Technology, and Applications 1–44
Bolan NS, Naidu R, Syers JK, Tillman RW (1999) Surface charge and solute interactions in soils. Adv Agron 67:87–140
Chen H, Koopal LK, Xiong J, Avena M, Tan W (2017) Mechanisms of soil humic acid adsorption onto montmorillonite and kaolinite. J Colloid Interf Sci 504:457–467. https://doi.org/10.1016/j.jcis.2017.05.078
Cordell D, Drangert JO, White S (2009) The story of phosphorus: global food security and food for thought. Global Environ Change 19(2):292–305. https://doi.org/10.1016/j.gloenvcha.2008.10.009
Dan J (1983) Soil chronosequences in israel. Catena 10(4):287–319
Feng X, Simpson AJ, Simpson MJ (2005) Chemical and mineralogical controls on humic acid sorption to clay mineral surfaces. Org Geochem 36(11):1553–1566. https://doi.org/10.1016/j.orggeochem.2005.06.008
Fine P, Mingelgrin U (1996) Release of phosphorus from waste-activated sludge. Soil Sci Soc Amer J 60(2):505–511. https://doi.org/10.2136/sssaj1996.03615995006000020024x
Freiberg Y, Fine P, Borisover M, Levkovitch I, Baram S (2022) Biosolids increase phosphate adsorption of semi-arid Mediterranean soils. J Environ Manag 305:114361. https://doi.org/10.1016/j.jenvman.2021.114361
Gérard F (2016) Clay minerals, iron/aluminum oxides, and their contribution to phosphate sorption in soils—a myth revisited. Geoderma 262:213–226. https://doi.org/10.1016/j.geoderma.2015.08.036
Gerke J (1992) Orthophosphate and organic phosphate in the soil solution of four sandy soils in relation to pH-evidence for humic- Fe- (Al-) phosphate complexes. Commun Soil Sci Plant Anal 23(5–6):601–612. https://doi.org/10.1080/00103629209368612
Gerke J (2010) Humic (organic matter)-Al(Fe)-phosphate complexes: an underestimated phosphate form in soils and source of plant-available phosphate. Soil Sci 175(9):417–425. https://doi.org/10.1097/SS.0b013e3181f1b4dd
Gerke Jorg, Hermann R (1992) Adsorption of orthophosphate to humic-Fe-complexes and to amorphous Fe-oxide summary - Zusammenfassung. Adsorption J Int Adsorption Soc 155(3):233–236
Graetz DA, Nair VD (2000) Phosphorus sorption isotherm determination. In Methods of phosphorus analysis for soils, sediments, residuals, and waters (Vol. 369, pp. 35–38). North Carolina State University Raleigh
Guppy CN, Menzies NW, Moody PW, Blamey FPC (2005) Competitive sorption reactions between phosphorus and organic matter in soil: a review. Aust J Soil Res 43(2):189–202. https://doi.org/10.1071/SR04049
Holford ICR (1997) Soil phosphorus: its measurement, and its uptake by plants. Aust J Soil Res 35(2):227–239. https://doi.org/10.1071/S96047
Huang PM, Li Y, Sumner ME (eds) (2011) Handbook of soil sciences: resource management and environmental impacts. CRC Press
Ijagbemi CO, Baek M, Kim D (2009) Montmorillonite surface properties and sorption characteristics for heavy metal removal from aqueous solutions. J Hazard Mater 166:538–546. https://doi.org/10.1016/j.jhazmat.2008.11.085
Jarvie HP, Withers JA, Neal C (2002) Review of robust measurement of phosphorus in river water: sampling, storage, fractionation and sensitivity. Hydrol Earth Syst Sci 6(1):113–132. https://doi.org/10.5194/hess-6-113-2002
Kafkafi U, Bar-Yosef B, Rosenberg R, Sposito G (1988) Phosphorus adsorption by kaolinite and montmorillonite: II. organic anion competition. Soil Sci Soc Amer J 52(6):1585–1589. https://doi.org/10.2136/sssaj1988.03615995005200060012x
Kang J, Hesterberg D, Osmond DL (2009) Soil organic matter effects on phosphorus sorption: a path analysis. Soil Sci Soc Amer J 73(2):360–366. https://doi.org/10.2136/sssaj2008.0113
Koyumdjisky H, Dan J, Suriano S, Nisim S (1988) Selected Soil Profile Descriptions of Soils of the Land of Israel. Pub. By the ARO, Volcanic Center. Inst. of Soil and Water, Dept. of Pedology, p 250
Laboski CAM, Lamb JA (2004) Impact of manure application on soil phosphorus sorption characteristics and subsequent water quality implications. Soil Sci 169(6):440–448. https://doi.org/10.1097/01.ss.0000131229.58849.0f
Lehmann J, Lan Z, Hyland C, Sato S, Solomon D, Ketterings QM (2005) Long-term dynamics of phosphorus forms and retention in manure-amended soils. Environ Sci Technol 39(17):6672–6680. https://doi.org/10.1021/es047997g
Li K, Bi Q, Liu X, Wang H, Sun C, Zhu Y, Lin X (2022) Unveiling the role of dissolved organic matter on phosphorus sorption and availability in a 5-year manure amended paddy soil. Sci Total Environ 838:155892. https://doi.org/10.1016/j.scitotenv.2022.155892
Lindsay WL (1979) Chemical equilibria in soils. John Wiley and Sons Ltd
Lobartini JC, Tan KH, Pape C (1998) Dissolution of aluminum and iron phosphate by humic acids. Commun Soil Sci Plant Anal 29(5–6):535–544. https://doi.org/10.1080/00103629809369965
Lu P, O’Connor GA (2001) Biosolids effects on phosphorus retention and release in some sandy Florida soils. J Environ Qual 30(3):1059–1063. https://doi.org/10.2134/jeq2001.3031059x
Majzik A, Tombácz E (2007) Interaction between humic acid and montmorillonite in the presence of calcium ions I. Interfacial and aqueous phase equilibria: adsorption and complexation. Org Geochem 38(8):1319–1329. https://doi.org/10.1016/j.orggeochem.2007.04.003
Manning BA, Goldberg S (1996) Modeling arsenate competitive adsorption on kaolinite, montmorillonite and illite. Clays Clay Miner 44(5):609–623. https://doi.org/10.1346/CCMN.1996.0440504
Marshall SK, Laboski CAM (2006) Sorption of inorganic and total phosphorus from dairy and swine slurries to soil. J Environ Qual 35(5):1836–1843. https://doi.org/10.2134/jeq2005.0281
Miller M, O’Connor G (2009) The longer-term phytoavailability of biosolids—phosphorus. Agron J 101(4):889–896. https://doi.org/10.2134/agronj2008.0197x
Moghimi AH, Hamdan J, Shamshuddin J, Samsuri AW, Abtahi A (2013) Physicochemical properties and surface charge characteristics of arid soils in southeastern Iran. Appl Environ Soil Sci 252861. https://doi.org/10.1155/2013/252861
Nasonova A, Levy GJ, Borisover M (2020) Bulk and water-extractable organic matter from compost: evaluation of the selective dissolution in water using infrared absorbance ratios. Environ Sci Pollut Res 27(34):42644–42655. https://doi.org/10.1007/s11356-020-10153-z
O’connor GA, Sarkar D, Brinton SR, Elliott HA, Martin FG (2004) Phytoavailability of biosolids phosphorus. J Environ Qual 3(2):703–712. https://doi.org/10.2134/jeq2004.7030
Pardo MT, Guadalix ME, Garcia-Gonzalez MT (1992) Effect of pH and background electrolyte on P sorption by variable charge soils. Geoderma 54(1–4):275–284. https://doi.org/10.1016/0016-7061(92)90109-K
Pierzynski GM (1991) The chemistry and mineralogy of phosphorus in excessively fertilized soils. Crit Rev Environ Control 21(3–4):265–295. https://doi.org/10.1080/10643389109388418
Quan-xian HUA, Jian-yun LI, Jian-min Z, Huo-yan W, Chang-wen DU (2008) Enhancement of phosphorus solubility by humic substances in ferrosols. Pedosphere 18(4):533–538
Riggle J, Von Wandruszka R (2005) Binding of inorganic phosphate to dissolved metal humates. Talanta 66(2 SPEC. ISS.):372–375. https://doi.org/10.1016/j.talanta.2004.11.003
Robinson JS, Sharpley AN (1996) Reaction in soil of phosphorus released from poultry litter. Soil Sci Soc Amer J 60(5):1583–1588. https://doi.org/10.2136/sssaj1996.03615995006000050043x
Ryden JC, Syers JK (1975) Rationalization of ionic strength and cation effects on phosphate sorption by soils. J Soil Sci 26(4):395–406. https://doi.org/10.1111/j.1365-2389.1975.tb01963.x
Secor RB, Radke CJ (1985) Spillover of the diffuse double layer on montmorillonite particles. J Colloid Interface Sci 103(1):237–244. https://doi.org/10.1016/0021-9797(85)90096-7
Shaheen SM, Tsadilas CD, Eskridge KM (2009) Effect of common ions on phosphorus sorption and lability in Greek alfisols with different pH. Soil Sci 174(1):21–26. https://doi.org/10.1097/SS.0b013e3181935a68
Shainberg I, Levy GJ (2005) Flocculation and dispersion. In Encyclopedia of Soils in the Environment (vol 4, issue 1999, pp 27–34). https://doi.org/10.1016/B0-12-348530-4/00363-5
Sharpley AN, McDowell RW, Kleinman PJ (2004) Amounts, forms, and solubility of phosphorus in soils receiving manure. Soil Sci Soc Amer J 68(6):2048–2057. https://doi.org/10.2136/sssaj2004.2048
Sibanda HM, Young SD (1986) Competitive adsorption of humus acids and phosphate on goethite, gibbsite and two tropical soils. J Soil Sci 37(2):197–204. https://doi.org/10.1111/j.1365-2389.1986.tb00020.x
Siddique MT, Robinson JS (2003) Phosphorus sorption and availability in soils amended with animal manures and sewage sludge. J Environ Qual 32(3):1114–1121. https://doi.org/10.2134/jeq2003.1114
Sposito G (1984) The surface chemistry of soils. Oxford University Press
Staunton S, Leprince F (1996) Effect of pH and some organic anions on the solubility of soil phosphaate: implications for P bioavailability. Europ J Soil Sci 47(2):231–239. https://doi.org/10.1111/j.1365-2389.1996.tb01394.x
Sims JT, Pierzynski GM (2005) Chemistry of phosphorus in soil. In: Tabatabai AM, Sparks DL (eds) Chemical processes in soil, SSSA book series 8. SSSA, Madison, pp 151–119. https://doi.org/10.2136/sssabookser8.c2
Sui Y, Thompson ML (2000) Phosphorus sorption, desorption, and buffering capacity in a biosolids-amended mollisol. Soil Sci Soc Amer J 64(1):164–169. https://doi.org/10.2136/sssaj2000.641164x
Sui Y, Thompson ML, Shang C (1999) Fractionation of phosphorus in a mollisol amended with biosolids. Soil Sci Soc Amer Journal 63(5):1174–1180. https://doi.org/10.2136/sssaj1999.6351174x
Tombácz E, Szekeres M (2004) Colloidal behavior of aqueous montmorillonite suspensions: the specific role of pH in the presence of indifferent electrolytes. Appl Clay Sci 27(1–2):75–94. https://doi.org/10.1016/J.CLAY.2004.01.001
Tombácz E, Szekeres M (2006) Surface charge heterogeneity of kaolinite in aqueous suspension in comparison with montmorillonite. Appl Clay Sci 34(1–4):105–124. https://doi.org/10.1016/J.CLAY.2006.05.009
Tombácz E, Libor Z, Illés E, Majzik A, Klumpp E (2004) The role of reactive surface sites and complexation by humic acids in the interaction of clay mineral and iron oxide particles. Org Geochem 35(3):257–267. https://doi.org/10.1016/j.orggeochem.2003.11.002
Wang X, Yost RS, Linquist BA (2001) Soil aggregate size affects phosphorus desorption from highly weathered soils and plant growth. Soil Sci Soc Amer J 65(1):139–146. https://doi.org/10.2136/sssaj2001.651139x
Wei SY, Tan WF, Liu F, Zhao W, Weng LP (2014) Surface properties and phosphate adsorption of binary systems containing goethite and kaolinite. Geoderma 213:478–484. https://doi.org/10.1016/j.geoderma.2013.09.001
Zmora-Nahum S, Hadar Y, Chen Y (2007) Physico-chemical properties of commercial composts varying in their source materials and country of origin. Soil Biol Biochem 39(6):1263–1276. https://doi.org/10.1016/j.soilbio.2006.12.017
Acknowledgements
Partial funding for this work came from the Israel National Coal Ash Board (INCAB), grant no. 344/17. The authors wish to express their deep gratitude to the late Mr. Omri Lulav, head of the Israel National Coal Ash Board (NCAB). The authors would also like to express their gratitude and appreciation to Ms. Shiran Cohen for her invaluable help with the analyses and Dr. Hillary Voet for her guidance and help with the statistical analysis.
Author information
Authors and Affiliations
Contributions
Yaniv Freiberg: investigation, writing—original draft, visualization, and data curation. Pinchas Fine: review and editing, visualization, data curation, funding acquisition, and supervision. Irit Levkovitch: investigation. Michael Borisover: supervision and writing—original Draft. Shahar Baram: funding acquisition, supervision, and review and editing.
Corresponding author
Ethics declarations
Ethical standard
This research did not involve any human participants and/or animals and informed consent is not applicable
Consent to participate
Not applicable.
Consent for publication
All authors give consent for publication in this journal.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Freiberg, ., Fine, P., Borisover, M. et al. Effect of biosolid-derived dissolved organic matter on orthophosphate sorption to soils depends on clay mineralogy and solution composition. Environ Sci Pollut Res 30, 113649–113659 (2023). https://doi.org/10.1007/s11356-023-30313-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-30313-1