Abstract
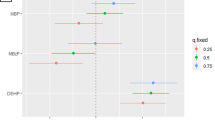
Phthalates (PAEs) are widespread persistent organic pollutants and endocrine disruptors. However, the associations between PAE exposure and the risk of miscarriage in humans are unclear, and an insufficient number of studies have evaluated the possible threshold or dose-dependent effects of first trimester PAE exposure on miscarriage risk. Our research measured the levels of mono-methyl phthalate (MMP), mono-ethyl phthalate, mono-isobutyl phthalate, MiBP mono-butyl phthalate (MBP), mono-octyl phthalate, mono-benzyl phthalate, mono(2-ethylhexyl) phthalate, mono(2-ethyl-5-oxohexyl) phthalate, and mono(2-ethyl-5-hydroxyhexyl) phthalate (MEHHP) in maternal urine collected in early gestation between 150 pregnancies ending in miscarriage and 150 pregnancies with live birth. We also estimated the odds ratios (ORs) and 95% confidence intervals (CIs) for miscarriage and each PAE as a continuous variable or quartile. A restrictive cubic splines was used to assess dose-dependent effects after controlling for maternal characteristics (e.g., age, educational level). we identified monotonically increasing dose-dependent effects of MEHHP and MMP on the risk of miscarriage. The largest effect estimates were approximately threefold higher for the highest MBP (OR = 2.57; 95% CI = 1.32–5.01) or MMP quartile (OR = 3.57; 95% CI = 1.82–7.00) and two-fold higher for the highest MEHHP quartile (OR = 2.12; 95% CI = 1.10–4.11). Our research preliminarily obtained possible thresholds of MBP, MEHHP, and MMP which were 18.07, 2.38, and 0.80 µg/g Cr for the risk of miscarriage, respectively. First-trimester exposure to MBP, MEHHP, and MMP exceeding certain thresholds increases the risk of miscarriage.



Similar content being viewed by others
Data availability
Data associated with the present study can be accessed on request to the author (xxlwangxia@163.com).
Abbreviations
- PAEs:
-
Phthalates
- ZBC:
-
Zunyi Birth Cohort
- MMP:
-
Mono-methyl
- MEP:
-
Mono-methyl phthalate
- MiBP:
-
Mono-ethyl phthalate
- MBP:
-
Mono-butyl phthalate
- MOP:
-
Mono-octyl phthalate
- MBzP:
-
Mono-benzyl phthalate
- MEHP:
-
Mono(2-ethylhexyl) phthalate
- MEOHP:
-
Mono(2-ethyl-5-oxohexyl) phthalate
- MEHHP:
-
Mono(2-ethyl-5-hydroxyhexyl) phthalate
- OR :
-
Odds ratio
- CI :
-
Confidence interval
- EVT:
-
Extravillous trophoblast
- BMI:
-
Body mass index
- GC–MS/MS:
-
High-performance gas chromatography–mass spectrometry
- LOD:
-
The limit of detection
- Cr:
-
Creation
References
Adde KS, Darteh EKM, Kumi-Kyereme A (2021) Experiences of women seeking post-abortion care services in a Regional Hospital in Ghana. PLoS One 16(4):e0248478. https://doi.org/10.1371/journal.pone.0248478
Cai C, Vandermeer B, Khurana R, Nerenberg K, Featherstone R, Sebastianski M, Davenport MH (2019) The impact of occupational shift work and working hours during pregnancy on health outcomes: a systematic review and meta-analysis. Am J Obstet Gynecol 221(6):563–576. https://doi.org/10.1016/j.ajog.2019.06.051
Cantonwine DE, Cordero JF, Rivera-González LO, Anzalota Del Toro LV, Ferguson KK, Mukherjee B … Meeker JD (2014) Urinary phthalate metabolite concentrations among pregnant women in Northern Puerto Rico: distribution, temporal variability, and predictors. Environ Int 62(1):11. https://doi.org/10.1016/j.envint.2013.09.014
Chang WH, Chou WC, Waits A, Liao KW, Kuo PL, Huang PC (2021) Cumulative risk assessment of phthalates exposure for recurrent pregnancy loss in reproductive-aged women population using multiple hazard indices approaches. Environ Int 154:106657. https://doi.org/10.1016/j.envint.2021.106657
Chen H, Feng W, Chen K, Qiu X, Xu H, Mao G, … Yang L (2021) Transcriptomic responses predict the toxic effect of parental co-exposure to dibutyl phthalate and diisobutyl phthalate on the early development of zebrafish offspring. AquatToxicol 235:105838. https://doi.org/10.1016/j.aquatox.2021.105838
Ferguson KK, Cantonwine DE, Rivera-González LO, Loch-Caruso R, Mukherjee B, Anzalota Del Toro LV … Meeker JD (2014) Urinary phthalate metabolite associations with biomarkers of inflammation and oxidative stress across pregnancy in Puerto Rico. Environ Sci Technol 48(12)7018-7025. https://doi.org/10.1021/es502076j
Ferguson KK, McElrath TF, Cantonwine DE, Mukherjee B, Meeker JD (2015a) Phthalate metabolites and bisphenol-A in association with circulating angiogenic biomarkers across pregnancy. Placenta 36(6):699–703. https://doi.org/10.1016/j.placenta.2015.04.002
Ferguson KK, McElrath TF, Chen YH, Mukherjee B, Meeker JD (2015b) Urinary phthalate metabolites and biomarkers of oxidative stress in pregnant women: a repeated measures analysis. Environ Health Perspect 123(3):210–216. https://doi.org/10.1289/ehp.1307996
Fisher M, Arbuckle TE, Mallick R, LeBlanc A, Hauser R, Feeley M … Walker M (2015) Bisphenol A and phthalate metabolite urinary concentrations: daily and across pregnancy variability. J Expo Sci Environ Epidemiol 25(3):231-239. https://doi.org/10.1038/jes.2014.65
Gao F, Hu W, Li Y, Shen H, Hu J (2017a) Mono-2-ethylhexyl phthalate inhibits human extravillous trophoblast invasion via the PPARγ pathway. Toxicol Appl Pharmacol 327:23–29. https://doi.org/10.1016/j.taap.2017.04.014
Gao H, Zhang YW, Huang K, Yan SQ, Mao LJ, Ge X … Tao FB (2017b) Urinary concentrations of phthalate metabolites in early pregnancy associated with clinical pregnancy loss in Chinese women. Sci Rep 7(1):6800. https://doi.org/10.1038/s41598-017-06450-2
Gao H, Zhu YD, Xu YY, Zhang YW, Yao HY, Sheng J … Tao FB (2017c) Season-dependent concentrations of urinary phthalate metabolites among Chinese pregnant women: repeated measures analysis. Environ Int 104:110-117. https://doi.org/10.1016/j.envint.2017c.03.021
He J, Chang K, Liu S, Ji J, Liu L, Feng Y, Wei J (2021) Phthalate levels in urine of pregnant women and their associated missed abortion risk. Reprod Biol 21(1):100476. https://doi.org/10.1016/j.repbio.2020.100476
He X, Zang J, Liao P, Zheng Y, Lu Y, Zhu Z … Wang W (2019) Distribution and dietary predictors of urinary phthalate metabolites among pregnant women in Shanghai, China. Int J Environ Res Public Health, 16(8). https://doi.org/10.3390/ijerph16081366
Hlisníková H, Petrovičová I, Kolena B, Šidlovská M, Sirotkin A (2020) Effects and mechanisms of phthalates' action on reproductive processes and reproductive health: a literature review. Int J Environ Res Public Health 17(18). https://doi.org/10.3390/ijerph17186811
Jauniaux E, Burton GJ (2005) Pathophysiology of histological changes in early pregnancy loss. Placenta 26(2–3):114–123. https://doi.org/10.1016/j.placenta.2004.05.011
Jiang HH, Du YY, Li YF (2021a) Ovarian toxicity and epigenetic mechanisms of phthalates and their metabolites. Curr Med Sci 41(2):236–249. https://doi.org/10.1007/s11596-021-2342-1
Jiang Y, Shi H, Liu Y, Zhao S, Zhao H (2021b) Applications of Melatonin in Female Reproduction in the Context of Oxidative Stress. Oxid Med Cell Longev 2021:6668365. https://doi.org/10.1155/2021/6668365
Jukic AM, Calafat AM, McConnaughey DR, Longnecker MP, Hoppin JA, Weinberg CR … Baird DD (2016) Urinary concentrations of phthalate metabolites and bisphenol A and associations with follicular-phase length, luteal-phase length, fecundability, and early pregnancy loss. Environ Health Perspect 124(3):321-328. https://doi.org/10.1289/ehp.1408164
Katsikantami I, Sifakis S, Tzatzarakis MN, Vakonaki E, Kalantzi OI, Tsatsakis AM, Rizos AK (2016) A global assessment of phthalates burden and related links to health effects. Environ Int 97:212–236. https://doi.org/10.1016/j.envint.2016.09.013
Keramat A, Malary M, Moosazadeh M, Bagherian N, Rajabi-Shakib MR (2021) Factors influencing stress, anxiety, and depression among Iranian pregnant women: the role of sexual distress and genital self-image. BMC Pregnancy Childbirth 21(1):87. https://doi.org/10.1186/s12884-021-03575-1
Larsen EC, Christiansen OB, Kolte AM, Macklon N (2013) New insights into mechanisms behind miscarriage. BMC Med 11:154. https://doi.org/10.1186/1741-7015-11-154
Li IC, Chen WP, Chen YP, Lee LY, Tsai YT, Chen CC (2018a) Acute and developmental toxicity assessment of erincine A-enriched Hericium erinaceus mycelia in Sprague-Dawley rats. Drug Chem Toxicol 41(4):459–464. https://doi.org/10.1080/01480545.2017.1381110
Li X, Sun H, Yao Y, Zhao Z, Qin X, Duan Y, Wang L (2018b) Distribution of phthalate metabolites between paired maternal-fetal samples. Environ Sci Technol 52(11):6626–6635. https://doi.org/10.1021/acs.est.8b00838
Lovekamp-Swan T, Davis BJ (2003) Mechanisms of phthalate ester toxicity in the female reproductive system. Environ Health Perspect 111(2):139–145. https://doi.org/10.1289/ehp.5658
Messerlian C, Wylie BJ, Mínguez-Alarcón L, Williams PL, Ford JB, Souter IC … Hauser R (2016) Urinary concentrations of phthalate metabolites and pregnancy loss among women conceiving with medically assisted reproduction. Epidemiology 27(6):879 888. https://doi.org/10.1097/ede.0000000000000525
Mu D, Gao F, Fan Z, Shen H, Peng H, Hu J (2015) Levels of phthalate metabolites in urine of pregnant women and risk of clinical pregnancy loss. Environ Sci Technol 49(17):10651–10657. https://doi.org/10.1021/acs.est.5b02617
Peng F, Ji W, Zhu F, Peng D, Yang M, Liu R … Yin L (2016) A study on phthalate metabolites, bisphenol A and nonylphenol in the urine of Chinese women with unexplained recurrent spontaneous abortion. Environ Res 150:622-628. https://doi.org/10.1016/j.envres.2016.04.003
Polinski KJ, Dabelea D, Hamman RF, Adgate JL, Calafat AM, Ye X, Starling AP (2018) Distribution and predictors of urinary concentrations of phthalate metabolites and phenols among pregnant women in the Healthy Start Study. Environ Res 162:308–317. https://doi.org/10.1016/j.envres.2018.01.025
Qi ST, Liang LF, Xian YX, Liu JQ, Wang W (2014) Arrested human embryos are more likely to have abnormal chromosomes than developing embryos from women of advanced maternal age. J Ovarian Res 7:65. https://doi.org/10.1186/1757-2215-7-65
Qureshi MS, Yusoff AR, Wirzal MD, Sirajuddin BJ, Afridi HI, Üstündag Z (2016) Methods for the determination of endocrine-disrupting phthalate esters. Crit Rev Anal Chem 46 (2):146-159. https://doi.org/10.1080/10408347.2015.1004157
Radke EG, Glenn BS, Braun JM, Cooper GS (2019) Phthalate exposure and female reproductive and developmental outcomes: a systematic review of the human epidemiological evidence. Environ Int 130:104580. https://doi.org/10.1016/j.envint.2019.02.003
San Lazaro Campillo I, Meaney S, Corcoran P, Spillane N, O'Donoghue K (2019) Risk factors for miscarriage among women attending an early pregnancy assessment unit (EPAU): a prospective cohort study. Ir J Med Sci 188(3):903-912. https://doi.org/10.1007/s11845-018-1955-2
Schmidt JS, Schaedlich K, Fiandanese N, Pocar P, Fischer B (2012) Effects of di(2-ethylhexyl) phthalate (DEHP) on female fertility and adipogenesis in C3H/N mice. Environ Health Perspect 120(8):1123–1129. https://doi.org/10.1289/ehp.1104016
Sheikh IA (2016) Stereoselectivity and the potential endocrine disrupting activity of di-(2-ethylhexyl)phthalate (DEHP) against human progesterone receptor: a computational perspective. J Appl Toxicol 36(5):741–747. https://doi.org/10.1002/jat.3302
Toft G, Jönsson BA, Lindh CH, Jensen TK, Hjollund NH, Vested A, Bonde JP (2012) Association between pregnancy loss and urinary phthalate levels around the time of conception. Environ Health Perspect 120(3):458–463. https://doi.org/10.1289/ehp.1103552
Treinen KA, Dodson WC, Heindel JJ (1990) Inhibition of FSH-stimulated cAMP accumulation and progesterone production by mono(2-ethylhexyl) phthalate in rat granulosa cell cultures. Toxicol Appl Pharmacol 106(2):334–340. https://doi.org/10.1016/0041-008x(90)90252-p
Ushie BA, Izugbara CO, Mutua MM, Kabiru CW (2018) Timing of abortion among adolescent and young women presenting for post-abortion care in Kenya: a cross-sectional analysis of nationally-representative data. BMC Womens Health 18(1):41. https://doi.org/10.1186/s12905-018-0521-4
Wang JJ, Tian Y, Li MH, Feng YQ, Kong L, Zhang FL, Shen W (2021) Single-cell transcriptome dissection of the toxic impact of Di (2-ethylhexyl) phthalate on primordial follicle assembly. Theranostics 11(10):4992–5009. https://doi.org/10.7150/thno.55006
Wang L, Zou Y, Wu P, Meng J, Zhang R (2020) Phthalate exposure in pregnant women and the influence of exposure to environmental tobacco smoke. J Matern Fetal Neonatal Med 33(18):3111–3115. https://doi.org/10.1080/14767058.2019.1569613
Yi H, Gu H, Zhou T, Chen Y, Wang G, Jin Y … Zhang L (2016) A pilot study on association between phthalate exposure and missed miscarriage. Eur Rev Med PharmacolSci 20(9):1894-1902
Zhang H, Gao F, Ben Y, Su Y (2020) Association between phthalate exposure and risk of spontaneous pregnancy loss: a systematic review and meta-analysis. Environ Pollut 267:115446. https://doi.org/10.1016/j.envpol.2020.115446
Acknowledgements
The authors thank all the participants and investigators and the National Key R&D Program of China, Science & Technology Program of Guizhou Province and The Guizhou Province Foundation for postgraduate Scientific Research Found Project for funding.
Funding
This work was supported by the National Key R&D Program of China (2018YFC1004300, 2018YFC1004302), the Science & Technology Program of Guizhou Province, China (QKHHBZ [2020]3002) and The Guizhou Province Foundation for Postgraduate Scientific Research Found Project YJSCXJH [2020] 167 and the Guizhou Genetic Foundation—ZK [2022] 614.
Author information
Authors and Affiliations
Contributions
Xia Wang: formal analysis, investigation, validation, data curation, writing—original draft, funding acquisition. Caidie He: investigation, data curation, visualization. Nian Wu: data curation, conceptualization. Yingkuan Tian: methodology, validation. Shimin Xiong: data curation, validation. Yijun Liu: data curation, validation. Yan Xie: data curation, validation. Kunming Tian: data curation, validation. Linglu Wang: investigation, validation. Juan Liao: investigation, validation. Derong Fang: investigation, validation. Songlin An: investigation, conceptualization. Wei Chen: investigation, conceptualization. Xiang Liu: investigation, conceptualization. Hongyu Yuan: investigation, data curation. Jingyi Huang: investigation, conceptualization. Xiaoshan Chen: investigation, conceptualization. Li Zhang: investigation, data curation. Quan Li: project administration, investigation, validation, resources. Xubo Shen: project administration, investigation, validation, resources. Yuanzhong Zhou: funding acquisition, investigation, validation, resources, writing–review and editing, funding acquisition.
Corresponding author
Ethics declarations
Ethical approval
This study was ethically reviewed by the Affiliated Hospital of Zunyi Medical University (Batch No.: KLL-2019–006).
Consent to participate
All participants agreed to participate in this study and signed the informed consents.
Consent for publication
The authors declare that this manuscript does not contain any individual person’s data and material in any form.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., He, C., Wu, N. et al. Maternal urine phthalate metabolite exposure and miscarriage risk: a nested case–control study of the Zunyi Birth Cohort. Environ Sci Pollut Res 30, 23124–23134 (2023). https://doi.org/10.1007/s11356-022-23717-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23717-y