Abstract
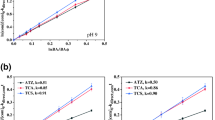
The presence of toxic chlorinated compounds in drinking water, generated during the disinfection step in water treatment plants, is of great concern for public health. In the present study, the performance of the UVC/H2O2 process, preceded by zero-valent-copper reduction, was evaluated for degrading 2,4,6-trichlorophenol (TCP). With this aim, the oxidation performed alone or in combination with the pre-reductive step was evaluated regarding TCP concentration over time, removal rate, mineralization, and toxicity to Vibrio fischeri, as well as oxidant dosage and the effect of water matrix. The UV/H2O2 process achieved fast (kobs = 1.4 min-1) and complete TCP degradation, as well as important mineralization (40.4%), with best results obtained for initial H2O2 concentration of 0.056 mmol L–1. Coupling of reductive and oxidative processes intensified contaminant mineralization, due to the synergistic effect of copper ions leached in the reductive process, particularly Cu(I), providing an additional route of H2O2 activation for generating HO• radicals (photo-Fenton-like process). High toxicity removals and increased mineralization could be successfully accomplished by the combined processes even in tap water, which is a clear advantage for practical application.







Similar content being viewed by others

References
Agency for Toxic Substances and Disease Registry (1999) Toxicological Profile for Chlorophenols. https://www.atsdr.cdc.gov/toxprofiles/tp107.pdf. Accessed 28 August 2019
Braun AM, Maurette MT, Oliveros E (1991) Photochemical Technology. John Wiley & Sons, Chichester
Brown TL, Lemay HE, Bursten BE, Murphy CJ (2009) Chemistry: the central science. Pearson Prentice Hall, Boston
Chanderia K, Kumarb S, Sharmaa J, Ametaa R, Punjabi PB (2017) Degradation of Sunset Yellow FCF using copper loaded bentonite and H2O2 as photo-Fenton like reagent. Arab J Chem 10:S205–S211. https://doi.org/10.1016/j.arabjc.2012.07.023
Choi JH, Kim YH (2009) Reduction of 2,4,6-trichlorophenol with zero-valent zinc and catalyzed zinc. J Hazard Mater 166:984–991. https://doi.org/10.1016/j.jhazmat.2008.12.004
Deng J, Y-q C, Yu-an L, Crittenden JC, S-q Z, N-y G, Li J (2017) Mesoporous manganese cobaltite nanocages as effective and reusable heterogeneous peroxymonosulfate activators for carbamazepine degradation. Chem Eng J 330:505–517. https://doi.org/10.1016/j.cej.2017.07.149
Dorathi PJ, Kandasamy P (2012) Dechlorination of chlorophenols by zero valent iron impregnated silica. J Environ Sci 24:765–773. https://doi.org/10.1016/S1001-0742(11)60817-6
Fang C, Xiao D, Liu W, Lou X, Zhou J, Wang Z (2016) Enhanced AOX accumulation and aquatic toxicity during 2,4,6-trichlorophenol degradation in a Co (II)/peroxymonosulfate/Cl– system. Chemosphere 144:415–2420. https://doi.org/10.1016/j.chemosphere.2015.11.030
Fernandéz-Alba AR, Hernando D, Agüera A, Cáceres J, Malato S (2002) Toxicity assays: a way for evaluating AOPs efficiency. Water Res 36:4255–4262. https://doi.org/10.1016/S0043-1354(02)00165-3
Gopal K, Tripathy SS, Bersillon JL, Dubey SP (2007) Chlorination byproducts, their toxicodynamics and removal from drinking water. J Hazard Mater 140:1–6. https://doi.org/10.1016/j.jhazmat.2006.10.063
Graça CAL, Mendes MA, Teixeira ACSC, Velosa AC (2020) Anoxic degradation of chlorpyrifos by zerovalent monometallic and bimetallic particles in solution. Chemosphere 244:125461. https://doi.org/10.1016/j.chemosphere.2019.125461
Guo Q, Jing L, Lan Y, He M, Xu Y, Xu H, Li H (2021) Construction 3D rod-like Bi3.64Mo0.36O6.55/CuBi2O4 photocatalyst for enhanced photocatalytic activity via a photo-Fenton-like Cu2+/Cu+ redox cycle. Sep Purif Technol 254:117546. https://doi.org/10.1016/j.seppur.2020.117546
Henschler D (1994) Toxicity of chlorinated organic compounds: effects of the introduction of chlorine in organic molecules. Angew Chem 33(19):1920–1935. https://doi.org/10.1002/anie.199419201
Hollanda LR, Graça CAL, Andrade LM, Mendes MA, Chiavone-Filho O, Teixeira ACSC (2018) Non-traditional atrazine degradation induced by zero-valent-copper: process optimization by the Doehlert experimental design, intermediates detection and toxicity assessment. J Chem Technol Biotechnol 94:1156–1164. https://doi.org/10.1002/jctb.5862
Hsieh M-C, Panchangam SC, Lai WW-P, Lin AY-C (2018) Degradation of methadone by the sunlight/FC process: Kkinetics, radical species participation and influence of the water matrix. Chemosphere 209:104–112. https://doi.org/10.1016/j.chemosphere.2018.06.076
Huang Z, Chen Z, Chen Y, Hu Y (2018) Synergistic effects in iron-copper bimetal doped mesoporous g-Al2O3 for Fenton-like oxidation of 4-chlorophenol: Sstructure, composition, electrochemical behaviors and catalytic performance. Chemosphere 203:442–449. https://doi.org/10.1016/j.chemosphere.2018.04.001
Ji H, Chang F, Hu X, Qin W, Shen J (2013) Photocatalytic degradation of 2,4,6- trichlorophenol over g-C3N4 under visible light irradiation. Chem Eng J 218:183–190. https://doi.org/10.1016/j.cej.2012.12.033
Juretic D, Puric J, Kusic H, Marin V, Bozic AL (2014) Structural influence on photooxidative degradation of halogenated phenols. Water Air Soil Pollut 225:1–18. https://doi.org/10.1007/s11270-014-2143-2
Kim Y-H, Carraway ER (2003) Dechlorination of chlorinated phenols by zero valent zinc. Environ Technol 24:1455–1463. https://doi.org/10.1080/09593330309385690
Munter R (2001) Advanced oxidation processes-current status and prospect. Proc Estonian Acad Sci Chem 50:59–80 https://www.kirj.ee/public/va_ke/k50-2-1.pdf Accessed 09 October 2020
Nogueira RFP, Oliveira MC, Partelini WC (2005) Simple and fast spectrophotometric determination of H2O2 in photo-Fenton reactions using metavanadate. Talanta 66:86–91. https://doi.org/10.1016/j.talanta.2004.10.001
Oppenländer T (2003) Photochemical purification of water and air. Wiley- VCH, Darmstadt
Peña-Guzmán C, Ulloa-Sancheza S, Morab K, Helena-Bustosc R, Lopez-Barrerad E, Alvareza J, Rodriguez-Pinzon M (2019) Emerging pollutants in the urban water cycle in Latin America: aA review of the current literature. J Environ Manag 237:408–423. https://doi.org/10.1016/j.jenvman.2019.02.100
Pereira TS, Rocha JAV, Duccatti A, Silveira GA, Pastoriza TF, Bringuenti L, Vargas VMF (2007) Evaluation of mutagenic activity in supply water at three sites in the state of Rio Grande do Sul, Brazil. Mutat Res 629:71–80. https://doi.org/10.1016/j.mrgentox.2006.12.008
Saritha P, Raj DSS, Aparna C, Laxmi PNV, Himabindu V, Anjaneyulu Y (2009) Degradative oxidation of 2,4,6-trichlorophenol using advanced oxidation processes–aA comparative study. Water Air Soil Pollut 200:169–179. https://link.springer.com/article/10.1007/s11270-008-9901-y
Shen YS, Ku Y, Lee K-C (1995) The effect of light absorbance on the decomposition of chlorophenols by ultraviolet radiation and UV/H2O2 processes. Water Res 29:907–914. https://doi.org/10.1016/0043-1354(94)00198-G
Sheng-Yang J, Yu-Jun W, Lian-Zhen L, Dong-Mei Z, Shen-Qiang W (2010) Comparison of degradation kinetics of 2,4,6-trichlorophenol using nanometer-and micrometer-sized Fe0. China Environ Sci 30:82–87
Souza LP, Graça CAL, Taqueda MES, Teixeira ACSC, Chiavone-Filho O (2019) Insights into the reactivity of zero-valent-copper-containing materials as reducing agents of 2,4,6-trichlorophenol in a recirculating packed-column system: Ddegradation mechanism and toxicity evaluation. Process Saf Environ Prot 127:348–358. https://doi.org/10.1016/j.psep.2019.05.032
World Health Organization (2011) In: WHO (Ed.), Guidelines for Drinking-Water Quality 4:104-108. https://apps.who.int/iris/bitstream/handle/10665/44584/9789241548151_eng.pdf;jsessionid=9A1140299727C71608B4F93AA166D8CD?sequence=1. Accessed 09 Oct 2020
Yazdanbakhsh A, Eslami A, Moussavi G (2018) Photo-assisted degradation of 2,4,6-trichlorophenol by an advanced reduction process based on sulfite anion radical: degradation, dechlorination and mineralization. Chemosphere 191:156–165. https://doi.org/10.1016/j.chemosphere.2017.10.023
Acknowledgments
The authors are grateful to the National Council for Scientific and Technological Development (CNPq, Brazil), Coordination for the Improvement of Higher Education Personnel (CAPES) - Brazil and São Paulo Research Foundation (FAPESP).
Funding
This study was financed in part by the Coordination for the Improvement of Higher Education Personnel (CAPES) - Brazil (Finance Codes 88881.068433/2014-01 and 3300201) and the São Paulo Research Foundation (FAPESP, grant no. 2013/50218-2).
Author information
Authors and Affiliations
Contributions
Conceptualization: C.A.L.G and A.C.S.C.T.; methodology: C.A.L.G and A.C.S.C.T.; investigation: L.P.S.; writing—original draft preparation: L.P.S. and C.A.L.G; writing—review and editing: C.A.L.G, A.C.S.C.T., and O.C-F.; supervision: C.A.L.G, A.C.S.C.T., and O. C-F; project administration: A.C.S.C.T. and O. C-F.; funding acquisition: A.C.S.C.T. and O.C-F.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
All the authors mentioned in the manuscript have agreed for authorship, read, and approved the manuscript, and given consent for submission and subsequent publication of the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Editorial Responsibility: Vítor Pais Vilar
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Souza, L.P., Graça, C.A.L., Teixeira, A.C.S.C. et al. Degradation of 2,4,6-trichlorophenol in aqueous systems through the association of zero-valent-copper-mediated reduction and UVC/H2O2: effect of water matrix and toxicity assessment. Environ Sci Pollut Res 28, 24057–24066 (2021). https://doi.org/10.1007/s11356-020-11885-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-11885-8