Abstract
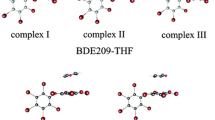
As the derivatives and structural analogs of polybrominated diphenyl ethers (PBDEs), hydroxylated polybrominated diphenyl ethers (OH-PBDEs) and methoxylated polybrominated diphenyl ethers (MeO-PBDEs) have attracted increasing concern. However, knowledge of the photochemical behaviors of OH-PBDEs and MeO-PBDEs in water is limited. Here, we used density functional theory and time-dependent density functional theory to examine the structure-related photochemical properties of OH-PBDEs and MeO-PBDEs in water and the effects of metal ions as environmental factors. Eight 6-OH-PBDEs with 1–8 bromine substituents and eight 6-MeO-PBDEs with 1–8 bromine substituents were selected for this study. The optimized geometries of the selected congeners and their complexes with metals in the lowest excited triplet state (T1) showed that one C–Br bond moderately or significantly elongated. The elongated C–Br bond in the T1 state was shown in the ortho-position for the 6-OH-PBDE congeners and the ortho-position or the meta-position for the 6-MeO-PBDE congeners. For the selected congeners, there were significant positive linear correlations between the number of bromine atoms (NBr) and the calculated average atomic charge of bromine and maximum electronic absorbance wavelength (λmax), and a negative linear correlation between the NBr and average bond dissociation energy of C–O bonds (BDEC–O). The photoreactivities of the 6-OH-PBDEs and 6-MeO-PBDEs increased with an increase in the bromination degree with or without metal ions. The calculated average atomic charge of bromine and BDEC–O of the complexes with Mg2+/Zn2+ was higher and lower than those of the corresponding monomers, respectively, indicating that the presence of Mg2+/Zn2+ increased the photoreactivity (debromination and dissociation of C–O bond) of the selected 6-OH-PBDEs and 6-MeO-PBDEs. The effects of the coordination of Mg2+/Zn2+ may be overestimated due to their missing explicit solvation shell. These results provide vital insight into the photochemical properties of OH-PBDEs and MeO-PBDEs in water.




Similar content being viewed by others
References
Arnoldsson K, Andersson PL, Haglund P (2012) Photochemical formation of polybrominated dibenzo-p-dioxins from environmentally abundant hydroxylated polybrominated diphenyl ethers. Environ Sci Technol 46:7567–7574
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652
Bezares-Cruz J, Jafvert CT, Hua I (2004) Solar photodecomposition of decabromodiphenyl ether: products and quantum yield. Environ Sci Technol 38:4149–4156
Bendig P, Vetter W (2010) Photolytical transformation rates of individual polybrominated diphenyl ethers in technical octabromo diphenyl ether (DE-79). Environ Sci Technol 44:1650–1655
Bendig P, Vetter W (2013) UV-induced formation of bromophenols from polybrominated diphenyl ethers. Environ Sci Technol 47:3665–3670
Burke K, Werschnik J, Gross EKU (2005) Time-dependent density functional theory: past, present, and future. J Chem Phys 123:062206
Chen Z, Liu H, Ho K, Huang H, Liu Q, Man Y, Lam MH, Du J, Wong M, Wang H (2016) Hydroxylated polybrominated diphenyl ethers (OH-PBDEs) in paired maternal and neonatal samples from South China: placental transfer and potential risks. Environ Res 148:72–78
Chen A, Park J, Linderholm L, Rhee A, Petreas M, DeFranco EA, Dietrich KN, Ho S (2013) Hydroxylated polybrominated diphenyl ethers in paired maternal and cord sera. Environ Sci Technol 47:3902–3908
Chen J, Wang D, Wang S, Qiao X, Huang L (2007) Quantitative structure-property relationships for direct photolysis of polybrominated diphenyl ethers. Ecotoxicol Environ Saf 66:348–352
Choudhry GG, Sundström G, Ruzo LO, Hutzinger O (1977) Photochemistry of chlorinated diphenyl ethers. J Agric Food Chem 25:1371–1376
Chu C, Stamatelatos D, McNeill K (2017) Aquatic indirect photochemical transformations of natural peptidic thiols: impact of thiol properties, solution pH, solution salinity and metal ions. Environ Sci Proc Impacts 19:1518–1527
Dahlberg A, Chen VL, Larsson K, Bergman Å, Asplund L (2016) Hydroxylated and methoxylated polybrominated diphenyl ethers in long-tailed ducks (Clangula hyemalis) and their main food, Baltic blue mussels (Mytilus trossulus × Mytilus edulis). Chemosphere 144:1475–1483
de Wit CA (2002) An overview of brominated flame retardants in the environment. Chemosphere 46:583–624
Eriksson J, Green N, Marsh G, Bergman Å (2004) Photochemical decomposition of 15 polybrominated diphenyl ether congeners in methanol/water. Environ Sci Technol 38:3119–3125
Fan Y, Zhao Z, Yu F, Lan J (2017) A 21-year record of methoxylated and hydroxylated polybrominated diphenyl ethers in sediments from the East China Sea. Environ Chem Lett 15:679–687
Fang L, Huang J, Yu G, Wang L (2008) Photochemical degradation of six polybrominated diphenyl ether congeners under ultraviolet irradiation in hexane. Chemosphere 71:258–267
Fang L, Huang J, Yu G, Li X (2009) Quantitative structure-property relationship studies for direct photolysis rate constants and quantum yields of polybrominated diphenyl ethers in hexane and methanol. Ecotoxicol Environ Saf 72:1587–1593
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE Jr, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09 Package. Gaussian, Inc., Wallingford CT
Ge J, Wang X, Li C, Wang S, Wang L, Qu R, Wang Z (2019) Photodegradation of polychlorinated diphenyl sulfides mediated by reactive oxygen species on silica gel. Chem Eng J 359:1056–1064
Herschmann JR, Ali A, Harris M, McClinton M, Zamadar M (2019) Effect of toxic metal ions on photosensitized singlet oxygen generation for photodegradation of polyaromatic hydrocarbon derivatives and inactivation of Escherichia coli. Photochem Photobiol 95:823–832
Jiang J, Zhao H, Sun S, Wang Y, Liu S, Xie Q, Li X (2018) Occurrence and profiles of halogenated phenols, polybrominated diphenyl ethers and hydroxylated polybrominated diphenyl ethers in the effluents of waste water treatment plants around Huang-Bo Sea, North China. Sci Total Environ 622-623:1–7
Kelly BC, Ikonomou MG, Blair JD, Gobas FAPC (2008) Hydroxylated and methoxylated polybrominated diphenyl ethers in a Canadian arctic marine food web. Environ Sci Technol 42:7069–7077
Li X, Huang J, Yu G, Deng S (2010) Photodestruction of BDE-99 in micellar solutions of nonionic surfactants of Brij 35 and Brij 58. Chemosphere 78:752–759
Li X, Gao Y, Guo L, Jiang G (2013) Structure-dependent activities of hydroxylated polybrominated diphenyl ethers on human estrogen receptor. Toxicology 309:15–22
Li X, Dong S, Zhang W, Fan X, Li Y, Wang R, Su X (2018) Global occurrence of polybrominated diphenyl ethers and their hydroxylated and methoxylated structural analogues in an important animal feed (fishmeal). Environ Pollut 234:620–629
Liu D, Wu S, Zhang Q, Guo M, Cheng J, Zhang S, Yao C, Chen J (2017) Occurrence, spatial distribution, and ecological risks of typical hydroxylated polybrominated diphenyl ethers in surface sediments from a large freshwater lake of China. Environ Sci Pollut Res 24:5773–5780
Liu X, Jiao Y, Lin C, Sun K, Zhao Y (2014) PBDEs, hydroxylated PBDEs and methoxylated PBDEs in bivalves from Beijing markets. Chemosphere 110:97–103
Liu Y, Liu J, Yu M, Zhou Q, Jiang G (2018) Hydroxylated and methoxylated polybrominated diphenyl ethers in marine food web of Chinese Bohai Sea and their human dietary exposure. Environ Pollut 233:604–611
Malmvarn A, Marsh G, Kautsky L, Athanasiadou M, Bergman Å, Asplund L (2005) Hydroxylated and methoxylated brominated diphenyl ethers in the red algae Ceramium tenuicorne and blue mussels from the Baltic Sea. Environ Sci Technol 39:2990–2997
Ochiai M, Nomiyama K, Isobe T, Yamada TK, Tajima Y, Matsuda A, Shiozaki A, Matsuishi T, Amano M, Iwata H, Tanabe S (2017) Polybrominated diphenyl ethers (PBDEs) and their hydroxylated and methoxylated analogues in the blood of harbor, Dall’s and finless porpoises from the Japanese coastal waters. Mar Environ Res 128:124–132
Pan L, Sun J, Wu X, Wei Z, Zhu L (2016) Transformation of hydroxylated and methoxylated 2,2′,4,4′,5-brominated diphenyl ether (BDE-99) in plants. J Environ Sci-China 49:197–202
Parry E, Zota AR, Park J, Woodruff TJ (2018) Polybrominated diphenyl ethers (PBDEs) and hydroxylated PBDE metabolites (OH-PBDEs): a six-year temporal trend in Northern California pregnant women. Chemosphere 195:777–783
Qiu X, Mercado-Feliciano M, Bigsby MR, Hites RA (2007) Measurement of polybrominated diphenyl ethers and metabolites in mouse plasma after exposure to a commercial pentabromodiphenyl ether mixture. Environ Health Perspect 115:1052–1058
Qu R, Liu H, Feng M, Yang X, Wang Z (2012) Investigation on intramolecular hydrogen bond and some thermodynamic properties of polyhydroxylated anthraquinones. J Chem Eng Data 57:2442–2455
Qu R, Li C, Pan X, Zeng X, Liu J, Huang Q, Feng J, Wang Z (2017) Solid surface-mediated photochemical transformation of decabromodiphenyl ether (BDE-209) in aqueous solution. Water Res 125:114–122
Qu R, Li C, Liu J, Xiao R, Pan X, Zeng X, Wang Z, Wu J (2018) Hydroxyl radical based photocatalytic degradation of halogenated organic contaminants and paraffin on silica gel. Environ Sci Technol 52:7220–7229
Ren X, Guo L, Gao Y, Zhang B, Wan B (2013) Hydroxylated polybrominated diphenyl ethers exhibit different activities on thyroid hormone receptors depending on their degree of bromination. Toxicol Appl Pharmacol 268:256–263
Routti H, Letcher RJ, Chu S, Bavel BV, Gabrielsen GW (2009) Polybrominated diphenyl ethers and their hydroxylated analogues in ringed seals (Phoca hispida) from Svalbard and the Baltic Sea. Environ Sci Technol 43:3494–3499
Shah S, Hao C (2016) Density functional theory study of direct and indirect photodegradation mechanisms of sulfameter. Environ Sci Pollut Res 23:19921–19930
Shi J, Qu R, Feng M, Wang X, Wang L, Yang S, Wang Z (2015) Oxidative degradation of decabromodiphenyl ether (BDE 209) by potassium permanganate: reaction pathways, kinetics, and mechanisms assisted by density functional theory calculations. Environ Sci Technol 49:4209–4217
Sturini M, Speltini A, Maraschi F, Profumo A, Pretali L, Fasani E, Albini A (2010) Photochemical degradation of marbofloxacin and enrofloxacin in natural waters. Environ Sci Technol 44:4564–4569
Sun J, Liu J, Liu Y, Jiang G (2013) Levels and distribution of methoxylated and hydroxylated polybrominated diphenyl ethers in plant and soil samples surrounding a seafood processing factory and a seafood market. Environ Pollut 176:100–105
Tomasi J, Mennucci B, Cammi R (2005) Quantum mechanical continuum solvation models. Chem Rev 105:2999–3093
Ueno D, Darling C, Alaee M, Pacepavicius G, Teixelira C, Campbell L, Letcher RJ, Bergman Å, Marsh G, Muir D (2008) Hydroxylated polybrominated diphenyl ethers (OH-PBDEs) in the abiotic environment: surface water and precipitation from Ontario, Canada. Environ Sci Technol 42:1657–1664
Verreault J, Gabrielsen GW, Chu S, Muir DCG (2005) Flame retardants and methoxylated and hydroxylated polybrominated diphenyl ethers in two Norwegian arctic top predators: glaucous gulls and polar bears. Environ Sci Technol 39:6021–6028
Vetter W, Haase-Aschoff P, Rosenfelder N, Komarova T, Mueller JF (2009) Determination of halogenated natural products in passive samplers deployed along the Great Barrier Reef, Queensland/Australia. Environ Sci Technol 43(16):6131–6137
Wang S, Hao C, Gao Z, Chen J, Qiu J (2012) Effects of excited-state structures and properties on photochemical degradation of polybrominated diphenyl ethers: a TDDFT study. Chemosphere 88:33–38
Wang S, Hao C, Gao Z, Chen J, Qiu J (2014) Theoretical investigations on direct photolysis mechanisms of polychlorinated diphenyl ethers. Chemosphere 111:7–12
Wei X, Chen J, Xie Q, Zhang S, Ge L, Qiao X (2013) Distinct photolytic mechanisms and products for different dissociation species of ciprofloxacin. Environ Sci Technol 47:4284–4290
Werner JJ, Arnold WA, Mcneill K (2006) Water hardness as a photochemical parameter: tetracycline photolysis as a function of calcium concentration, magnesium concentration, and pH. Environ Sci Technol 40:7236–7241
Xie Q, Chen J, Shao J, Chen C, Zhao H, Hao C (2009) Important role of reaction field in photodegradation of deca-bromodiphenyl ether: theoretical and experimental investigations of solvent effects. Chemosphere 76:1486–1490
Xie Q, Chen J, Zhao H, Qiao X, Cai X, Li X (2013) Different photolysis kinetics and photooxidation reactivities of neutral and anionic hydroxylated polybrominated diphenyl ethers. Chemosphere 90:188–194
Xie Q, Chen J, Zhao H, Wang X, Xie H (2015) Distinct photoproducts of hydroxylated polybromodiphenyl ethers from different photodegradation pathways: a case study of 2′-HO-BDE-68. Environ Sci Proc Impacts 17:351–357
Xue W, Chen J, Xie Q, Zhao H (2015) Direct photolysis of MeO-PBDEs in water and methanol: focusing on cyclization product MeO-PBDFs. Chemosphere 139:518–524
Xu B, Wu M, Wang M, Pan C, Qiu W, Tang L, Xu G (2018) Polybrominated diphenyl ethers (PBDEs) and hydroxylated PBDEs in human serum from Shanghai, China: a study on their presence and correlations. Environ Sci Pollut Res 25:3518–3526
Yu B, Zhang R, Liu P, Zhang Y, Zhang Y, Bai Y (2015) Determination of nine hydroxylated polybrominated diphenyl ethers in water by precolumn derivatization-gas chromatography-mass spectrometry. J Chromatogr A 1419:19–25
Zhang K, Wan Y, Giesy JP, Lam MHW, Wiseman S, Jones PD, Hu J (2010) Tissue concentrations of polybrominated compounds in Chinese sturgeon (Acipenser sinensis): origin, hepatic sequestration, and maternal transfer. Environ Sci Technol 44:5781–5786
Zhang Y, Xie Q, Chen J, Li Y, Fu Z (2015) Insights into the photochemical transformation pathways of triclosan and 2′-HO-BDE-28. J Hazard Mater 300:354–358
Zhao G-J, Han K-L (2010) pH-controlled twisted intramolecular charge transfer (TICT) excited state via changing the charge transfer direction. Phys Chem Chem Phys 12:8914–8918
Zhao G-J, Han K-L (2012) Hydrogen bonding in the electronic excited state. Acc Chem Res 45:404–413
Zhao HX, Jiang JQ, Wang YL, Xie Q, Qu BC (2017) Phototransformation of 2,4,6-tribromophenol in aqueous solution: kinetics and photolysis products. J Environ Sci Healh A 52:45–54
Zhao Q, Zhao H, Quan X, He X, Chen S (2015) Photochemical formation of hydroxylated polybrominated diphenyl ethers (OH-PBDEs) from polybrominated diphenyl ethers (PBDEs) in aqueous solution under simulated solar light irradiation. Environ Sci Technol 49:9092–9099
Funding
This research was supported by the National Natural Science Foundation of China (41601519) and the Natural Science Foundation of Jiangsu Province (BK20150891).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Suresh Pillai
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 2899 kb)
Rights and permissions
About this article
Cite this article
Wang, S., Wang, S., Shah, S. et al. A density functional theory/time-dependent density functional theory study of the structure-related photochemical properties of hydroxylated polybrominated diphenyl ethers and methoxylated polybrominated diphenyl ethers and metal ion effects. Environ Sci Pollut Res 27, 9297–9306 (2020). https://doi.org/10.1007/s11356-019-07538-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-07538-0