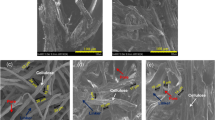
Abstract
The increase in biodiesel production has been leading to an excess amount of crude glycerol and, consequently, serious environmental issues. For this reason, electrospun chitosan-based nanofibers (CB-EN), composed by chitosan and poly(ethylene oxide) (PEO), were synthesized to apply in the biosorption of impurities from industrial glycerol. To evaluate the biosorption efficiency, the chitosan-based nanofiber was compared to other chitosan-based biosorbents (chitosan biopolymeric film and chitosan powder). The equilibrium and thermodynamic studies were successfully performed to comprehend the interaction mechanisms through the biosorption of glycerol pigments onto electrospun chitosan-based nanofibers. The temperature effect was evaluated by experimental equilibrium curves. Freundlich and BET models were used to estimate isotherm parameters. Gibbs free energy change, enthalpy change, entropy change, and isosteric heat of biosorption were quantified. The equilibrium curves showed that the highest equilibrium relative adsorption (340.7 g−1) was reached at 60 °C. The BET model was the most suitable to represent the equilibrium behavior. The thermodynamic parameters indicated that the biosorption was spontaneous, exothermic, random, and energetic heterogeneous. Therefore, this work developed a green and efficient alternative to refine industrial glycerol.

Note: This data is mandatory. Please provide




Similar content being viewed by others
Abbreviations
- 1/n :
-
Heterogeneity factor (dimensionless)
- A :
-
Glycerol pigment absorbance (dimensionless)
- A o, A e :
-
Initial and equilibrium glycerol pigment absorbances (dimensionless)
- f :
-
Dilution factor (dimensionless)
- K D :
-
Thermodynamic equilibrium constant (L−1 g)
- K F :
-
Freundlich constant (g−1/n)
- K S, K L :
-
BET equilibrium constants (dimensionless)
- L' :
-
Integration constant (dimensionless)
- m ad :
-
Adsorbent weight (g)
- m o, m f :
-
Initial and final glycerol pigment weights (g)
- q e :
-
Equilibrium adsorption capacity (g mg−1)
- q m :
-
Monolayer adsorption capacity (g−1)
- q r :
-
Relative adsorption capacity (g−1)
- q re :
-
Equilibrium relative adsorption capacity (g−1)
- R :
-
Gas constant (kJ mol−1 K−1)
- R 2, ARE :
-
Statistical coefficients (%)
- T :
-
Absolute temperature (K)
- V :
-
Solution volume (mL)
- ΔG 0 :
-
Gibbs free energy change (kJ mol−1)
- ΔH 0 :
-
Enthalpy change (kJ mol−1)
- ΔH a :
-
Isosteric heat of biosorption (kJ mol−1)
- ΔS 0 :
-
Entropy change (J mol−1 K−1)
- ρ :
-
Glycerol density (g L−1)
- εc :
-
Constant related to analyte nature and cuvette size (m3 g−1)
References
Ardi MS, Aroua MK, Hashim NA (2015) Progress, prospect and challenges in glycerol purification process: a review. Renew Sust Energ Rev 42:1164–1173. https://doi.org/10.1016/j.rser.2014.10.091
Bayrak Y (2003) Adsorption isotherms in bleaching hazelnut oil. J Am Oil Chem Soc 80:1143–1146. https://doi.org/10.1007/s11746-003-0833-7
Crini G, Badot PM (2008) Application of chitosan, a natural aminopolysaccharide, for dye removal from aqueous solutions by adsorption process using batch studies: a review of recent literature. Prog Polym Sci 33(4):399–447. https://doi.org/10.1016/j.progpolymsci.2007.11.001
Dhabhai R, Ahmadifeijani E, Dalai AK, Reaney M (2016) Purification of crude glycerol using a sequential physico–chemical treatment, membrane filtration, and activated charcoal adsorption. Sep Purif Technol 168:101–106. https://doi.org/10.1016/j.seppur.2016.05.030
Dotto GL, Souza VC, Moura JM, Moura CM, Pinto LAA (2011) Influence of drying techniques on the characteristics of chitosan and the quality of biopolymer films. Dry Technol 29(15):1784–1791. https://doi.org/10.1080/07373937.2011.602812
Dotto GL, Moura JM, Cadaval TRS Jr, Pinto LAA (2013) Application of chitosan films for the removal of food dyes from aqueous solutions by adsorption. Chem Eng J 214:8–16. https://doi.org/10.1016/j.cej.2012.10.027
Dotto GL, Sharma SK, Pinto LAA (2015) Biosorption of organic dyes: research opportunities and challenges. Scrivener Publishing, Massachusetts
Dotto GL, Campana-Filho SP, Pinto LAA (2017a) Chitosan based materials and its applications. Bentham Science Publishers, Sharjah
Dotto GL, Santos JMN, Tanabe EH, Bertuol DA, Foletto EL, Lima EC, Pavan FA (2017b) Chitosan/polyamide nanofibers prepared by Forcespinning® technology: a new adsorbent to remove anionic dyes from aqueous solutions. J Clean Prod 144:120–129. https://doi.org/10.1016/j.jclepro.2017.01.004
Ebadi A, Mohammadzadeh JSS, Khudiev A (2009) What is the correct form of BET isotherm for modeling liquid phase adsorption? Adsorption (Boston) 15(1):65–73. https://doi.org/10.1007/s10450-009-9151-3
Farias BS, Vidal ÉM, Ribeiro NT, Silveira N Jr, Vaz BS, Kuntzler SG, Morais MG, Cadaval TRS Jr, Pinto LAA (2018) Electrospun chitosan/poly(ethylene oxide) nanofibers applied for the removal of glycerol impurities from biodiesel production by biosorption. J Mol Liq 268:365–370. https://doi.org/10.1016/j.molliq.2018.07.081
Frantz TS, Silveira N Jr, Quadro MS, Andreazza R, Barcelos AA, Cadaval TRS Jr, Pinto LAA (2017) Cu(II) adsorption from copper mine water by chitosan films and the matrix effects. Environ Sci Pollut Res 24:5908–5917. https://doi.org/10.1007/s11356-016-8344-z
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:385–471
Giles CH, MacEwan TH, Nakhwa SN, Smith D (1960) Studies in adsorption. Part XI.* A system of classification of solution adsorption isotherms, and its use in diagnosis of adsorption mechanisms and in measurements of specific surface areas of solids. J Chem Soc:3973–3993. https://doi.org/10.1039/JR9600003973
Goldstein JI, Newbury DE, Echlin P, Joy DC, Romig AD Jr, Lyman CE, Fiori C, Lifshin E (2003) Scanning electron microscopy and X–ray microanalysis. Springer US, New York
Gore PM, Khurana L, Siddique S, Panicker A, Kandasubramanian B (2018) Ion–imprinted electrospun nanofibers of chitosan/1–butyl–3–methylimidazolium tetrafluoroborate for the dynamic expulsion of thorium (IV) ions from mimicked effluents. Environ Sci Pollut Res 25(4):3320–3334. https://doi.org/10.1007/s11356-017-0618-6
Hoekman SK, Broch A, Robbins C, Ceniceros E, Natarajan M (2012) Review of biodiesel composition, properties, and specifications. Renew Sust Energ Rev 16(1):143–169. https://doi.org/10.1016/j.rser.2011.07.143
Hossini H, Soltani RD, Safari M, Maleki A, Rezaee R, Ghanbari R (2017) The application of a natural chitosan/bone char composite in adsorbing textile dyes from water. Chem Eng Commun 204(9):1082–1093. https://doi.org/10.1080/00986445.2017.1340274
Hu S, Luo X, Wan C, Li Y (2012) Characterization of crude glycerol from biodiesel plants. J Agric Food Chem 60:5915–5921. https://doi.org/10.1021/jf3008629
Kwork KCM, Koong LF, Ansari TA, McKay G (2018) Adsorption/desorption or arsenite and arsenate on chitosan and nanochitosan. Environ Sci Pollut Res 25(15):14734–14742. https://doi.org/10.1007/s11356-018-1501-9
Kyzas GZ, Fu J, Matis KA (2013) The change from past to future for adsorbent materials in treatment of dyeing wastewaters. Materials (Basel) 6(11):5131–5158. https://doi.org/10.3390/ma6115131
León O, Muñoz-Bonilla A, Soto D, Perez D, Rangel M, Colina M, Fernandez-García M (2018) Removal of anionic and cationic dyes with bioadsorbent oxidized chitosans. Carbohydr Polym 194:375–383. https://doi.org/10.1016/j.carbpol.2018.04.072
Li L, Zhang J, Yanxiang L, Yang C (2017) Removal of CR (VI) with spiral wound chitosan nanofiber membrane module via dead–end filtration. J Membr Sci Technol 544:333–341. https://doi.org/10.1016/j.memsci.2017.09.045
Liu Y (2009) Is the free energy change of adsorption correctly calculated? J Chem Eng Data 54(7):1981–1985. https://doi.org/10.1021/je800661q
Milonjić SK (2007) A consideration of the correct calculation of thermodynamic parameters of adsorption. J Serb Chem Soc 72(12):1363–1367. https://doi.org/10.2298/JSC0712363M
Moura JM, Farias BS, Rodrigues DAS, Moura CM, Dotto GL, Pinto LAA (2015) Preparation of chitosan with different characteristics and its application for biofilms production. J Polym Environ 23(4):470–477. https://doi.org/10.1007/s10924-015-0730-y
Moura JM, Gründmann DDR, Cadaval TRS Jr, Dotto GL, Pinto LAA (2016) Comparison of chitosan with different physical forms to remove reactive black 5 from aqueous solutions. J Environ Chem Eng 4(2):2259–2267. https://doi.org/10.1016/j.jece.2016.04.003
Piccin JS, Cadaval TRS Jr, Pinto LAA, Dotto GL (2017) Adsorption isotherms in liquid phase: experimental, modeling, and interpretations. Springer International Publishing, Switzerland
Pohndorf RS, Cadaval TRS Jr, Pinto LAA (2016) Kinetics and thermodynamics adsorption of carotenoids and chlorophylls in rice bran oil bleaching. J Food Eng 185:9–16. https://doi.org/10.1016/j.jfoodeng.2016.03.028
Qin W, Li J, Tu J, Yang H, Chen Q, Liu H (2017) Fabrication of porous chitosan membranes composed of nanofibers by low temperature thermally induced phase separation, and their adsorption behavior for Cu2+. Carbohydr Polym 178:338–346. https://doi.org/10.1016/j.carbpol.2017.09.051
Silva SM, Sampaio KA, Ceriani R, Verhé R, Stevens C, De Greyt W, Meirelles AJA (2013) Adsorption of carotenes and phosphorus from palm oil onto acid activated bleaching earth: equilibrium, kinetics and thermodynamics. J Food Eng 118(4):341–349. https://doi.org/10.1016/j.jfoodeng.2013.04.026
Silverstein RM, Webster FX, Kiemle DJ (2005) Spectrometric identification of organic compounds. Wiley, New Jersey
Swinehart DF (1962) The Beer–Lambert law. J Chem Educ 39(7):333. https://doi.org/10.1021/ed039p333
Tan HW, Aziz ARA, Aroua MK (2013) Glycerol production and its application as a raw material: a review. Renew Sust Energ Rev 27:118–127. https://doi.org/10.1016/j.rser.2013.06.035
Wan Z, Li M, Zhang Q, Fan Z, Verpoort F (2018) Concurrent reduction–adsorption of chromium using m–phenylediamine–modified magnetic chitosan: kinetics, isotherm, and mechanism. Environ Sci Pollut Res 25(18):17830–17841. https://doi.org/10.1007/s11356-018-1941-2
Weska RF, Moura JM, Batista LM, Rizzi J, Pinto LAA (2007) Optimization of deacetylation in the production of chitosan from shrimp wastes: use of response surface methodology. J Food Eng 80:749–753. https://doi.org/10.1016/j.jfoodeng.2006.02.006
Wisniewska M (2012) The temperature effect on the adsorption mechanism of polyacrylamide on the silica surface and its stability. Appl Surf Sci 258(7):3094–3101. https://doi.org/10.1016/j.apsusc.2011.11.044
Yang F, Hanna MA, Sun R (2012) Value–added uses for crude glycerol—a byproduct of biodiesel production. Biotechnol Biofuels 5(1):13. https://doi.org/10.1186/1754-6834-5-13
Zhang C, Chen Z, Guo W, Zhu C, Zou Y (2018) Simple fabrication of chitosan/graphene nanoplates composite spheres for efficient adsorption of acid dyes from aqueous solution. Int J Biol Macromol 112:1048–1054. https://doi.org/10.1016/j.ijbiomac.2018.02.074
Živković S, Veljković M (2018) Environmental impacts the of production and use of biodiesel. Environ Sci Pollut Res 25(1):191–199. https://doi.org/10.1007/s11356-017-0649-z
Funding
The authors would like to thank CAPES (Brazilian Agency for Improvement of Graduate Personnel) and CNPQ (National Council of Science and Technological Development) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Farias, B.S., Gründmann, D.D.R., Strieder, M.M. et al. Biosorption of glycerol impurities from biodiesel production onto electrospun chitosan-based nanofibers: equilibrium and thermodynamic evaluations. Environ Sci Pollut Res 26, 28436–28443 (2019). https://doi.org/10.1007/s11356-019-04525-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-04525-3