Abstract
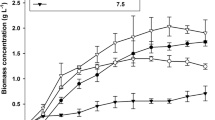
The effect of sequential batch cultures of the marine microalgae Nannochloropsis oculata on lipid and biomass production was studied in 200-L raceway ponds for 167 days (nine harvesting cycles) during winter and spring seasons under greenhouse conditions. The highest biomass concentration and productivity were 1.2 g/L and 49.8 mg/L/day on days 73 (5th cycle) and 167 (9th cycle), respectively. The overall interval of lipid production was between 131 and 530 mg/L. Despite the daily and seasonal variations of light irradiance (0–1099 μmol photon/m2 s), greenhouse temperature (2.1–50.7 °C), and culture temperature (12.5–31.4 °C), ANOVA analysis showed no statistical difference (p value > 0.01) on the fatty acid methyl ester (FAMES) composition over the nine harvesting cycles evaluated. The most abundant FAMES were palmitic (C16:0), stearic (C18:0) and palmitoleic (C16:1∆9) acids with 37.1, 28.6, and 8.4 %, respectively. The sequential batch cultures of N. oculata in raceway ponds showed an increasing biomass production in each new cycle while keeping the quality of the fatty acid mixture under daily and seasonal variations of light irradiance and temperature.





Similar content being viewed by others
Abbreviations
- DCW:
-
Dry cell weight
- FAMES:
-
Fatty acid methyl esters
- SBC:
-
Sequential batch culture
References
Bao Y, Liu M, Wu X, Cong W, Ning Z (2012) In situ carbon supplementation in large-scale cultivations of Spirulina platensis in open raceway pond. Biotechnol Bioprocess Eng 17(1):93–99
Borowitzka M, Moheimani N (2013) Open pond culture systems. In: Borowitzka MA, Moheimani NR (eds) Algae for Biofuels and Energy, vol 5. Developments in Applied Phycology. Springer Netherlands, pp 133–152
Boruff BJ, Moheimani NR, Borowitzka MA (2015) Identifying locations for large-scale microalgae cultivation in Western Australia: a GIS approach. Appl Energy 149:379–391
Cabello J, Toledo-Cervantes A, Sánchez L, Revah S, Morales M (2015) Effect of the temperature, pH and irradiance on the photosynthetic activity by Scenedesmus obtusiusculus under nitrogen replete and deplete conditions. Bioresour Technol 181:128–135
Capellán-Pérez I, Mediavilla M, de Castro C, Carpintero Ó, Miguel LJ (2014) Fossil fuel depletion and socio-economic scenarios: an integrated approach. Energy 77:641–666
Chen M, Tang H, Ma H, Holland TC, Ng KY, Salley SO (2011) Effect of nutrients on growth and lipid accumulation in the green algae Dunaliella tertiolecta. Bioresour Technol 102(2):1649–1655
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25(3):294–306
Chiu SY, Kao CY, Tsai MT, Ong SC, Chen CH, Lin CS (2009) Lipid accumulation and CO2 utilization of Nannochloropsis oculata in response to CO2 aeration. Bioresour Technol 100(2):833–838
Converti A, Casazza AA, Ortiz EY, Perego P, Del Borghi M (2009) Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis oculata and Chlorella vulgaris for biodiesel production. Chem Eng Process 48(6):1146–1151
Das P, Aziz SS, Obbard JP (2011) Two phase microalgae growth in the open system for enhanced lipid productivity. Renew Energy 36(9):2524–2528
Fábregas J, Patiño M, Arredondo-Vega BO, Tobar JL, Otero A (1995) Renewal rate and nutrient concentration as tools to modify productivity and biochemical composition of cyclostat cultures of the marine microalga Dunaliella tertiolecta. Appl Microbiol Biotechnol 44(3):287–292
García-Ferris C, de los Ríos A, Ascaso C, Moreno J (1996) Correlated biochemical and ultrastructural changes in nitrogen-starved Euglena glacilis. J Phycol 32(6):953–963
Gwak Y, Hwang Y, Wang B, Kim M, Jeong J, Lee C, Hu Q, Han D, Jin E (2014) Comparative analyses of lipidomes and transcriptomes reveal a concerted action of multiple defensive systems against photooxidative stress in Haematococcus pluvialis. J Exp Bot 65(15):4317–4334
Hase R, Oikawa H, Sasao C, Morita M, Watanabe Y (2000) Photosynthetic production of microalgal biomass in a raceway system under greenhouse conditions in Sendai city. J Biosci Bioeng 89(2):157–163
He Q, Yang H, Wu L, Hu C (2015) Effect of light intensity on physiological changes, carbon allocation and neutral lipid accumulation in oleaginous microalgae. Bioresour Technol 191:219–228
Ho SH, Lu WB, Chang JS (2012) Photobioreactor strategies for improving the CO2 fixation efficiency of indigenous Scenedesmus obliquus CNW-N: statistical optimization of CO2 feeding, illumination, and operation mode. Bioresour Technol 105:106–113
Ho SH, Chen CNN, Lai YY, Lu WB, Chang JS (2014) Exploring the high lipid production potential of a thermotolerant microalga using statistical optimization and semi-continuous cultivation. Bioresour Technol 163:128–135
Janssen M, Bresser L, Baijens T, Tramper J, Mur LR, Snel JFH, Wijffels RH (2000) Scale-up aspects of photobioreactors: effects of mixing-induced light/dark cycles. J Appl Phycol 12:225–237
Keeney DR, Nelson DW (1982) Nitrogen inorganic forms. In: Black CA (ed) Methods of soil analysis, part 2: agronomy. American Society of Agronomy, Madison, pp 643–698
Kliphuis AM, Janssen M, van den End EJ, Martens DE, Wijffels RH (2011) Light respiration in Chlorella sorokiniana. J Appl Phycol 23:935–947
Liu J, Huang J, Sun Z, Zhong Y, Jiang Y, Chen F (2011) Differential lipid and fatty acid profiles of photoautotrophic and heterotrophic Chlorella zofingiensis: assessment of algal oils for biodiesel production. Bioresour Technol 102(1):106–110
Mata TM, Martins AA, Caetano NS (2010) Microalgae for biodiesel production and other applications: a review. Renew Sust Energ Rev 14(1):217–232
Merchuk JC, Ronen M, Giris S, Arad SM (1998) Light/dark cycles in the growth of the red microalga Porphyridium sp. Biotechnol Bioeng 59:705–713
Moheimani N, Borowitzka M (2006) The long-term culture of the coccolithophore Pleurochrysis carterae (Haptophyta) in outdoor raceway ponds. J Appl Phycol 18(6):703–712
Morais MG, Radmann EM, Andrade MR, Teixeira GG, Brusch LRF, Costa JAV (2009) Pilot scale semicontinuous production of Spirulina biomass in southern Brazil. Aquaculture 294:60–64
Moreno J, Vargas MÁ, Rodrıguez H, Rivas J, Guerrero MG (2003) Outdoor cultivation of a nitrogen-fixing marine cyanobacterium, Anabaena sp. ATCC 33047. Biomol Eng 20:191–197
Pawlowski A, Mendoza JL, Guzmán JL, Berenguel M, Acién FG, Dormido S (2015) Selective pH and dissolved oxygen control strategy for a raceway reactor within an event-based approach. Control Eng Pract 44:209–218
Radmann EM, Reinehr CO, Costa JAV (2007) Optimization of the repeated batch cultivation of microalga Spirulina platensis in open raceway ponds. Aquaculture 265:118–126
Raes EJ, Isdepsky A, Muylaert K, Borowitzka MA, Moheimani NR (2014) Comparison of growth of Tetraselmis in a tubular photobioreactor (Biocoil) and a raceway pond. J Appl Phycol 26(1):247–255
Richardson JW, Johnson MD, Outlaw JL (2012) Economic comparison of open pond raceways to photo bio-reactors for profitable production of algae for transportation fuels in the Southwest. Algal Res 1(1):93–100
Ritchie RJ (2006) Consistent sets of spectrophotometric chlorophyll equations for acetone, methanol and ethanol solvents. Photosynth Res 89(1):27–41
Rodolfi L, Chini Zittelli G, Bassi N, Padovani G, Biondi N, Bonini G, Tredici MR (2009) Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102(1):100–112
Selinummi J, Seppala J, Yli-Harja O, Puhakka JA (2005) Software for quantification of labeled bacteria from digital microscope images by automated image analysis. BioTechniques 39(6):859–863
Silva Benavides AM, Torzillo G, Kopecký J, Masojídek J (2013) Productivity and biochemical composition of Phaeodactylum tricornutum (Bacillariophyceae) cultures grown outdoors in tubular photobioreactors and open ponds. Biomass Bioenergy 54:115–122
Singh J, Gu S (2010) Commercialization potential of microalgae for biofuels production. Renew Sust Energ Rev 14(9):2596–2610
Solovchenko AE, Khozin-Goldberg I, Cohen Z, Merzlyak MN (2009) Carotenoid-to-chlorophyll ratio as a proxy for assay of total fatty acids and arachidonic acid content in the green microalga Parietochloris incisa. J Appl Phycol 21(3):361–366
Spolaore P, Joannis-Cassan C, Duran E, Isambert A (2006) Optimization of Nannochloropsis oculata growth using the response surface method. J Chem Technol Biotechnol 81(6):1049–1056
Strickland JDH, Parsons TR (1972) A practical handbook of seawater analysis vol 1977, vol no. 167; no. 1977. Fisheries Research Board of Canada, Ottawa
Suh SS, Kim SJ, Hwang J, Park M, Lee TK, Eui-Joon Kil EJ, Lee L (2015) Fatty acid methyl ester profiles and nutritive values of 20 marine microalgae in Korea. Asian Pac J Trop Med 8(3):191–196
Toledo-Cervantes A, Morales M, Novelo E, Revah S (2013) Carbon dioxide fixation and lipid storage by Scenedesmus obtusiusculus. Bioresour Technol 130:652–658
Zhou W et al (2012) Growing wastewater-born microalga Auxenochlorella protothecoides UMN280 on concentrated municipal wastewater for simultaneous nutrient removal and energy feedstock production. Appl Energy 98:433–440
Zhu CJ, Lee YK (1997) Determination of biomass dry weight of marine microalgae. J Appl Phycol 9(2):189–194
Acknowledgments
The authors are grateful for the financial support provided by the National Science and Technology Council (CONACYT) of Mexico, the Secretary for Research and Postgraduated Studies—Instituto Politécnico Nacional (SIP-IPN, grants 20130388 and 20144620), and the collaboration of Research and Development Center of CARSO. The authors would also like to thank Claudia Guerrero Barajas and Hervé Marie for their participation in reviewing the paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Gerald Thouand
Rights and permissions
About this article
Cite this article
Millán-Oropeza, A., Fernández-Linares, L. Biomass and lipid production from Nannochloropsis oculata growth in raceway ponds operated in sequential batch mode under greenhouse conditions. Environ Sci Pollut Res 24, 25618–25626 (2017). https://doi.org/10.1007/s11356-016-7013-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-7013-6