Abstract
Introduction and objective
Taking into consideration the challenges of lipid analytics, present study aims to design the best high-throughput workflow for detection and annotation of lipids.
Material and methods
Serum lipid profiling was performed on CSH-C18 and EVO-C18 columns using UHPLC Q-TOF-MS and generated lipid features were annotated based on m/z and fragment ion using different software.
Result and discussion
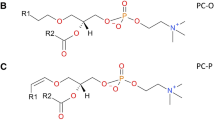
Better detection of features was observed in CSH-C18 than EVO-C18 with enhanced resolution except for Glycerolipids (triacylglycerols) and Sphingolipids (sphingomyelin).
Conclusion
The study revealed an optimized untargeted Lipidomics-workflow with comprehensive lipid profiling (CSH-C18 column) and confirmatory annotation (LipidBlast).


Similar content being viewed by others
References
Adibhatla, R. M., & Hatcher, J. F. (2007). Role of lipids in brain injury and diseases. Future Lipidology, 2(4), 403–422. https://doi.org/10.2217/17460875.2.4.403
Cajka, T., & Fiehn, O. (2016). Increasing lipidomic coverage by selecting optimal mobile-phase modifiers in LC–MS of blood plasma. Metabolomics, 12(2), 34. https://doi.org/10.1007/s11306-015-0929-x
Cajka, T., & Fiehn, O. (2017). LC-MS-based lipidomics and automated identification of lipids using the LipidBlast in-silico MS/MS library. Methods in Molecular Biology (clifton, N.J.). https://doi.org/10.1007/978-1-4939-6996-8_14
Cheema, A. K., Li, Y., Girgis, M., Jayatilake, M., Simas, M., Wise, S. Y., Olabisi, A. O., Seed, T. M., & Singh, V. K. (2019). Metabolomic studies in tissues of mice treated with amifostine and exposed to gamma-radiation. Scientific Reports, 9(1), 15701. https://doi.org/10.1038/s41598-019-52120-w
Damen, C. W. N., Isaac, G., Langridge, J., Hankemeier, T., & Vreeken, R. J. (2014). Enhanced lipid isomer separation in human plasma using reversed-phase UPLC with ion-mobility/high-resolution MS detection. Journal of Lipid Research, 55(8), 1772–1783. https://doi.org/10.1194/jlr.D047795
Heintz, M. M., Kumar, R., Maner-Smith, K. M., Ortlund, E. A., & Baldwin, W. S. (2022). Age- and diet-dependent changes in hepatic lipidomic profiles of phospholipids in male mice: Age acceleration in Cyp2b-null mice. Journal of Lipids, 2022, 7122738. https://doi.org/10.1155/2022/7122738
Hogan, S. R., Phan, J. H., Alvarado-Velez, M., Wang, M. D., Bellamkonda, R. V., Fernández, F. M., & LaPlaca, M. C. (2018). Discovery of lipidome alterations following traumatic brain injury via high-resolution metabolomics. Journal of Proteome Research, 17(6), 2131–2143. https://doi.org/10.1021/acs.jproteome.8b00068
Kind, T., Liu, K.-H., Lee, D. Y., DeFelice, B., Meissen, J. K., & Fiehn, O. (2013). LipidBlast in silico tandem mass spectrometry database for lipid identification. Nature Methods, 10(8), 755–758. https://doi.org/10.1038/nmeth.2551
Kinetex EVO columns, Introducing New Kinetex Evo UHPLC/HPLC Columns. Retrieved April 11, 2022, from https://www.phenomenex.com/Info/Page/2014knxevo
Lu, S., Liu, H., Jin, C., Li, Q., & Guo, L. (2019). An efficient and comprehensive plant glycerolipids analysis approach based on high-performance liquid chromatography-quadrupole time-of-flight mass spectrometer. Plant Direct, 3(11), e00183. https://doi.org/10.1002/pld3.183
Matyash, V., Liebisch, G., Kurzchalia, T. V., Shevchenko, A., & Schwudke, D. (2008). Lipid extraction by methyl-tert-butyl ether for high-throughput lipidomics. Journal of Lipid Research, 49(5), 1137–1146. https://doi.org/10.1194/jlr.D700041-JLR200
Pellegrino, R. M., Di Veroli, A., Valeri, A., Goracci, L., & Cruciani, G. (2014). LC/MS lipid profiling from human serum: A new method for global lipid extraction. Analytical and Bioanalytical Chemistry, 406(30), 7937–7948. https://doi.org/10.1007/s00216-014-8255-0
Quehenberger, O., Armando, A. M., Brown, A. H., Milne, S. B., Myers, D. S., Merrill, A. H., Bandyopadhyay, S., Jones, K. N., Kelly, S., Shaner, R. L., Sullards, C. M., Wang, E., Murphy, R. C., Barkley, R. M., Leiker, T. J., Raetz, C. R. H., Guan, Z., Laird, G. M., …Dennis, E. A. (2010). Lipidomics reveals a remarkable diversity of lipids in human plasma. Journal of Lipid Research, 51(11), 3299–3305. https://doi.org/10.1194/jlr.M009449
Sandra, K., Pereira, A. D. S., Vanhoenacker, G., David, F., & Sandra, P. (2010). Comprehensive blood plasma lipidomics by liquid chromatography/quadrupole time-of-flight mass spectrometry. Journal of Chromatography. A, 1217(25), 4087–4099. https://doi.org/10.1016/j.chroma.2010.02.039
Sarafian, M. H., Gaudin, M., Lewis, M. R., Martin, F.-P., Holmes, E., Nicholson, J. K., & Dumas, M.-E. (2014). Objective set of criteria for optimization of sample preparation procedures for ultra-high throughput untargeted blood plasma lipid profiling by ultra performance liquid chromatography-mass spectrometry. Analytical Chemistry, 86(12), 5766–5774. https://doi.org/10.1021/ac500317c
Sud, M., Fahy, E., Cotter, D., Brown, A., Dennis, E. A., Glass, C. K., Merrill, A. H., Murphy, R. C., Raetz, C. R. H., Russell, D. W., & Subramaniam, S. (2007). LMSD: LIPID MAPS structure database. Nucleic Acids Research, 35, D527-532. https://doi.org/10.1093/nar/gkl838
Xu, T., Hu, C., Xuan, Q., & Xu, G. (2020). Recent advances in analytical strategies for mass spectrometry-based lipidomics. Analytica Chimica Acta, 1137, 156–169. https://doi.org/10.1016/j.aca.2020.09.060
Yang, K., & Han, X. (2016). Lipidomics: Techniques, applications, and outcomes related to biomedical sciences. Trends in Biochemical Sciences, 41(11), 954–969. https://doi.org/10.1016/j.tibs.2016.08.010
Zhao, Y.-Y., Wu, S.-P., Liu, S., Zhang, Y., & Lin, R.-C. (2014). Ultra-performance liquid chromatography-mass spectrometry as a sensitive and powerful technology in lipidomic applications. Chemico-Biological Interactions, 220, 181–192. https://doi.org/10.1016/j.cbi.2014.06.029
Zhao, Z., & Xu, Y. (2010). An extremely simple method for extraction of lysophospholipids and phospholipids from blood samples. Journal of Lipid Research, 51(3), 652–659. https://doi.org/10.1194/jlr.D001503
Funding
This work was supported by the Defence R&D Organization (DRDO), Ministry of Defence, India (INM 324); The Council of Scientific and Industrial Research (CSIR), India; and The University Grant Commission (UGC), India.
Author information
Authors and Affiliations
Contributions
SD and PR conceived and designed the study. SD, KM and AS conducted the experiments and data acquisition. SD, KM and RB analyzed the data. SD, KM, RB and DM wrote the manuscript with input and critical feedback from all authors. PR evaluated the manuscript critically and all the authors approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There have been no conflicts of interest declared by the authors.
Research involving human and animal rights
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (XLSX 64 KB)
Supplementary Table 1: CSH-C18 (a) and EVO-C18 (b) columns specific annotated lipid categories and their sub-classes acquired on UHPLC-MS.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dhariwal, S., Maan, K., Baghel, R. et al. Systematic untargeted UHPLC–Q-TOF–MS based lipidomics workflow for improved detection and annotation of lipid sub-classes in serum. Metabolomics 19, 24 (2023). https://doi.org/10.1007/s11306-023-01983-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-023-01983-2