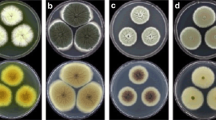
Abstract
The stem rot caused by Sclerotium hydrophilum and false smut caused by Ustilaginoidea virens are two of the major production constraints in rice cultivation in India and other countries. Stem rot and false smut can be effectively controlled with synthetic fungicides. However, the indiscriminate use of chemical fungicides may cause development of resistance among the pathogens. In addition to this, synthetic fungicides also exhibit harmful impacts on the environment. Exploiting microbe-based alternatives for managing plant diseases diminishes public concerns about the ill effects of pesticide usage in crops. In this regard, the present study was designed to investigate the potential of native microbial biocontrol agents (BCAs) from rice rhizosphere for the sustainable management of stem rot and false smut diseases in rice. Potential BCAs and pathogens were identified and characterized through morphological, biochemical, and sanger sequencing techniques. Bio-efficacy tests of potential BCAs against stem rot and false smut diseases on rice under glasshouse conditions indicated higher seed vigour index of the treated seeds, significant improvement in the growth of the seedling, increased dry weight, reduction in percentage disease index viz., 70.03% (stem rot) and 69.24% (false smut) over the control plants. Phytohormones indole acetic acid (IAA), abscisic acid (ABA), gibberellic acid (GA), salicylic acid (SA), and zeatin (tZ) were detected and quantified in the four potential BCAs using liquid chromatography- tandem mass spectrometry (LC–MS/MS). Scanning electron microscopy (SEM) studies revealed the endophytic nature of the strains in rice. The study indicated a positive correlation between the diversity and concentration of phytohormones released by the bioagents and enhanced plant growth promotion and disease suppression in rice.













Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article. Any additional information can be made available upon request by the corresponding author.
References
Adrien B, Martin F (2018) Phenazines in plant-beneficial Pseudomonas spp.: biosynthesis, regulation, function and genomics. Environ Microbiol 20(11):3905–3917. https://doi.org/10.1111/1462-2920.14395
Akhtar SS, Mekureyaw MF, Pandey C, Roitsch T (2020) Role of cytokinins for interactions of plants with microbial pathogens and pest insects. Front Plant Sci. https://doi.org/10.3389/fpls.2019.01777
Alexander DB, Zuberer DA (1991) Use of chrome azurol S reagents to evaluate siderophore production by rhizosphere bacteria. Biol Fertil Soils 12(1):39–45. https://doi.org/10.1007/BF00369386
Bashyal BM, Parmar P, Zaidi NW, Sunil KS, Prakash G, Rashmi A (2021) Improved methodology for the isolation of false smut pathogen Ustilaginoidea virens of rice. Indian Phytopathol 74:249–252. https://doi.org/10.1007/s42360-020-00282-3
Bergey DH, Holt JG (1994) Bergey’s manual of determinative bacteriology, 9th edn. Williams & Wilkins, Baltimore
Bric JM, Bostock RM, Silverstone SE (1991) Rapid in situ assay for indole acetic acid production by bacteria immobilized on a nitrocellulose membrane. Appl Environ Microbiol 57:535–538. https://doi.org/10.1128/aem.57.2.535-538.1991
Brueske CH (1980) Phenylalanine ammonia lyase activity in tomato roots infected and resistant to the root-knot nematode, Meloidogyne incognita. Physiol Plant Pathol 16:409–414. https://doi.org/10.1016/S0048-4059(80)80012-9
Cao D, Barbier F, Yoneyama K, Beveridge CA (2020) A rapid method for quantifying RNA and phytohormones from a small amount of plant tissue. Front Plant Sci 11:1664–2462. https://doi.org/10.3389/fpls.2020.605069
Cappuccino JC, Sherman N (1992) Microbiology: “a laboratory manual.” Benjamin/Cummings, New York
Caulier S, Nannan C, Gillis A, Licciardi F, Bragard C, Mahillon J (2019) Overview of the antimicrobial compounds produced by members of the Bacillus subtilis group. Front Microbiol. https://doi.org/10.3389/fmicb.2019.003
Cavaglieri L, Passone A, Miriam E (2004) Screening procedures for selecting rhizobacteria with biocontrol effects upon Fusarium verticillioides growth and fumonisin B1 production. Microbiol Res 155(9):747–754. https://doi.org/10.1016/j.resmic.2004.06.001
Chen Y, Yao J, Li YF, Wang WX, Yang X, Zhang AF (2014) Simple and rapid detection of rice false smut pathogen Ustilaginoidea virens in rice seeds. Phytoparasitica 42:371–375. https://doi.org/10.1007/s12600-013-0372-3
Dafaallah A (2019) Duncan's multiple range test (DMRT). https://doi.org/10.13140/RG.2.2.16262.93764
Daniel MJ, Aiyanathan E, Sevugapperumal N (2017) Virulence and genetic diversity of Sclerotium rolfsii Sacc. infecting groundnut using nuclear (RAPD & ISSR) markers. J Environ Biol 38:147–159. https://doi.org/10.22438/jeb/38/1/ms-274
Dong OX, Ronald PC (2019) Genetic engineering for disease resistance in plants: recent progress and future perspectives. Plant Physiol 180(1):26–38. https://doi.org/10.1104/pp.18.01224
Fabio M, Matt P, Adrian RNT, Prasad B, Joon L, Ossama E, Nandana M, Anton K, Rodrigo L (2022) Search and sequence analysis tools services from EMBL-EBI in 202. Nucleic Acids Res 50(W1):W276-279
Farzand A, Moosa A, Zubair M, Khan AR, Massawe VC, Tahir HAS, Sheikh TMM, Ayaz M, Gao X (2019) Suppression of Sclerotinia sclerotiorum by the induction of systemic resistance and regulation of antioxidant pathways in tomato using fengycin produced by Bacillus amyloliquefaciens FZB42. Biomolecules 9:613. https://doi.org/10.3390/biom9100613
Flores Treviño S, Villarreal Treviño L, Rodríguez Noriega E (2019) Catalase test revisited: implication of intrinsic catalase activity in clinical samples of coagulase negative staphylococci. Eur J Clin Microbiol 7(1):e01486-e1518. https://doi.org/10.3390/antibiotics12030554
Fu SF, Wei JY, Chen HW, Liu YY, Lu HY, Chou JY (2015) Indole-3-acetic acid: a widespread physiological code in interactions of fungi with other organisms. Plant Signal Behav 10(8):e1048052. https://doi.org/10.1080/15592324.2015.1048052
Furtak K, Grządziel J, Gałązka A, Niedźwiecki J (2020) Prevalence of unclassified bacteria in the soil bacterial community from floodplain meadows (fluvisols) under simulated flood conditions revealed by a metataxonomic approachss. CATENA 188:104448
Gangwar GP, Sinha AP (2010) Comparative antagonistic potential of Trichoderma spp. against Xanthomonas oryzae pv. oryzae. Ann Plant Sci 8(2):458–463
Gauillard F, Richardforget F, Nicolas J (1993) New spectrophotometric assay for polyphenol oxidase activity. Anal Biochem 215:59–65. https://doi.org/10.1006/abio.1993.1554
Golden CD, Allison EH, Cheung WW, Dey MM, Halpern BS, McCauley DJ, Smith M, Vaitla B, Zeller D, Myers SS (2016) Nutrition: Fall in fish catch threatens human health. Nature 534(7607):317–320. https://doi.org/10.1038/534317a
Gopalakrishnan S, Srinivas V, Alekhya G (2015) Effect of plant growth-promoting Streptomyces sp. on growth promotion and grain yield in chickpea (Cicer arietinum L). 3 Biotech 5:799–806. https://doi.org/10.1007/s13205-015-0283-8
Halifu S, Deng X, Song X, Song R, Liang X (2020) Inhibitory mechanism of Trichoderma virens ZT05 on Rhizoctonia solani. Plants 9(7):912. https://doi.org/10.3390/plants9070912
Hammerschmidt R, Nuckles EM, Kuc J (1982) Association of enhanced peroxidase activity with induced systemic resistance of cucumber to Colletotrichum lagenarium. Physiol Plant Pathol 20(1):73–82. https://doi.org/10.1016/0048-4059(82)90025-X
Hasan N, Farzand A, Heng Z, Khan IU, Moosa A, Zubair M, NaY YS, Canming T (2020) Antagonistic potential of novel endophytic Bacillus Strains and mediation of plant defense against verticillium wilt in upland cotton. Plants 9(11):1438. https://doi.org/10.3390/plants9111438
He DC, He MH, Amalin DM, Liu W, Alvindia DG, Zhan J (2021) Biological control of plant diseases: an evolutionary and eco-economic consideration. Pathogens (basel, Switzerland) 10(10):1311. https://doi.org/10.3390/pathogens10101311
IIRR, Production Oriented Survey, 2023. https://www.icar-iirr.org/index.php/en/component/content/article/39-iirr-databases/212-production-oriented-survey-pos-data-portal.
IRRI. (2002). Standard Evaluation System for Rice, International Rice Research Institute, Los Banos, Manila, Philippines, p. 24. http://www.knowledgebank.irri.org/images/docs/rice-standard-evaluation-system.pdf
Izquierdo Bueno I, Gonzalez Rodriguez VE, Simon A, Dalmais B, Pradier JM, Le Pêcheur P, Mercier A, Walker AS, Garrido C, Collado IG, Viaud M (2018) Biosynthesis of abscisic acid in fungi: identification of a sesquiterpene cyclase as the key enzyme in Botrytis cinerea. Environ Microbiol 20(7):2469–2482. https://doi.org/10.1111/1462-2920.14258
Jamaloddin M, Anumalla MC, Guru G, Chintavaram B, Maliha A, Hitendra KP, Jauhar A (2021) Molecular Approaches for Disease Resistance in Rice. In: Ali J, Wani SH (eds) Rice Improvement. Springer, Cham (10.1007/978-3-030-66530-2_10)
Johnson LF, Curl EA (1977) Burgess Publishing Company. Minnea Polis, p. 247.
Kabdwal BC, Sharma R, Kumar A, Kumar S, Singh KP, Srivastava RM (2023) Efficacy of different combinations of microbial biocontrol agents against sheath blight of rice caused by Rhizoctonia solani. Egypt J Biol Pest Control 33:29. https://doi.org/10.1186/s41938-023-00671-6
Kannan C, Divya M, Rekha G, Maruthi P, Hajira S, Sundaram RM (2021) Diversity analysis of antagonistic microbes against bacterial leaf and fungal sheath blight diseases of rice. Egypt J Biol Pest Control 31:115. https://doi.org/10.1186/s41938-021-00462-x
Kannan C, Divya M, Rekha G, Barbadikar K, Maruthi P, Shaik H, Sundaram RM (2022) Whole genome sequencing data of native isolates of Bacillus and Trichoderma having potential biocontrol and plant growth promotion activities in rice. Data Brief 41:107923. https://doi.org/10.1016/j.dib.2022.107923
Kohl J, Kolnaar R, Ravensberg WJ (2019) Mode of action of microbial biological control agents against plant diseases: relevance beyond efficacy. Front Plant Sci 10:1664–2462. https://doi.org/10.3389/fpls.2019.00845
Kumar S, Stecher G, Tamura K (2015) MEGA7: molecular evolutionar genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Kumari P, Meena M, Upadhyay RS (2018) Characterization of plant growth promoting rhizobacteria (PGPR) isolated from the rhizosphere of Vigna radiata (mung bean). Biocatal Agric Biotechnol 16:155–162. https://doi.org/10.1016/j.bcab.2018.07.029
Kunkel BN, Harper CP (2018) The roles of auxin during interactions between bacterial plant pathogens and their hosts. J Exp Bot 69(2):245–254. https://doi.org/10.1093/jxb/erx447
Leon V, Manokaran R, Pandian RT, Aundy K, Sharma P (2018) Opportunistic endophytism of Trichoderma species in rice Pusa Basmati-1 (PB1). Indian J Exp Biol 56.
Liu Y, Fang XA, Chen G, Ye Y, Xu J, Ouyang G (2019) Recent development in sample preparation techniques for plant hormone analysis. Trends Analyt Chem 113:224–233. https://doi.org/10.1016/j.trac.2019.02.006
Lorck H (1948) Production of hydrocyanic acid by bacteria. Physiol Plant 1(2):142–146. https://doi.org/10.1111/j.1399-3054.1948.tb07118.x
Lu D, Willard D, Patel IR, Kadwell S, Overton L, Kost T, Luther M, Chen W, Woychik RP, Wilkison WO, Cone RD (1994) Agouti protein is an antagonist of the melanocyte-stimulating-hormone receptor. Nature 371(6500):799–802. https://doi.org/10.1038/371799a0
Ludwig-Müller J (2015) Plants and endophytes: equal partners in secondary metabolite production. Biotechnol Lett 37:1325–1334. https://doi.org/10.1007/s10529-015-1814-4
Makowski D, Ben-Shachar M, Patil I, Lüdecke D (2020) Methods and algorithms for correlation analysis in R. J Open Source Software 5(51):2306
Malick CP, Singh MB (1980) In: Plant Enzymology and Histo Enzymology. Kalyani publishers, New Delhi, p 286
Mathew SB, Raghu S, Prabhukarthikeyan SR, Arup KM, Manas KB, Srikanta L, Mayabini J (2020) Yield loss assessment in rice (Oryza sativa) due to false smut infection. Indian J Agric Sci 90(4):361–365. https://doi.org/10.56093/ijas.v90i2.99023.
Mhatre PH, Karthik C, Kadirvelu K, Divya KL (2019) Plant growth promoting rhizobacteria (PGPR): a potential alternative tool for nematodes bio-control. Biocatal Agric Biotechnol. https://doi.org/10.1016/j.bcab.2018.11.009
Mukherjee M, Mukherjee PK, Horwitz BA, Zachow C, Berg G, Zeilinger S (2012) Trichoderma-plant-pathogen interactions: advances in genetics of biological control. Indian J Microbiol 52(4):522–529. https://doi.org/10.1007/s12088-012-0308-5
Nakkeeran S, Rajamanickam SS, Karthikeyan M, Mahendra K, Renukadevi P, Johnson I (2021) Antimicrobial secondary metabolites from Trichoderma spp. as next generation fungicides, Biocontrol Agents and Secondary Metabolites, Woodhead Publishing, pp 257–282, ISBN 9780128229194. https://doi.org/10.1016/B978-0-12-822919-4.00011-9.
Nawaz ME, Malik K, Hassan MN (2022) Rice-associated antagonistic bacteria suppress the Fusarium fujikoroi causing rice bakanae disease. Biol Control 67:101–109. https://doi.org/10.1007/s10526-021-10122-6
Ozimek E, Jaroszuk Sciseł J, Bohacz J, Kornillowicz Kowalska T, Tyskiewicz R, Słomka A, Nowak A, Hanaka A (2018) Synthesis of indoleacetic acid, gibberellic acid and ACC-deaminase by Mortierella strains promote winter wheat seedlings growth under different conditions. Int J Mol Sci 19:3218. https://doi.org/10.3390/ijms19103218
Palmieri D, Vitale S, Lima G, Di Pietro A, Turra D (2020) A bacterial endophyte exploits chemotropism of a fungal pathogen for plant colonization. Nat Commun 11(1):5264
Pandit MA, Kumar J, Gulati S, Bhandari N, Mehta P, Katyal R, Rawat CD, Mishra V, Kaur J (2022) Major biological control strategies for plant pathogens. Pathogens (basel, Switzerland) 11(2):273. https://doi.org/10.3390/pathogens11020273
Park J, Lee Y, Martinoia E, Geisler M (2017) Plant hormone transporters: what we know and what we would like to know. BMC Biol 15(1):93
Parul T, Ayushi S, Anamika G, Mrinalini P, Rajiv R (2022) Mechanism and function of salicylate in plant toward biotic stress tolerance, Emerging Plant Growth Regulators in Agriculture, Academic Press pp 131–164. https://doi.org/10.1016/B978-0-323-91005-7.00018-7. ISBN 9780323910057.
Parveen SR, Latha D (2019) Characterization of Siderophore producing Pseudomonas spp. for its Plant Growth Promoting Properties. Biosci Biotechnol Res Commun 12(4).
Patten CL, Glick BR (2002) Role of Pseudomonas putida indole acetic acid in development of the host plant root system. Appl Environ Microbiol 68(8):3795–3801. https://doi.org/10.1128/aem.68.8.3795-3801.2002
Qiu J, Meng S, Deng Y, Huang S, Kou Y (2019) Ustilaginoidea virens: a fungus infects rice flower and threats world rice production. Rice Sci 26(4):199–206. https://doi.org/10.1016/j.rsci.2018.10.007
Rahman MM, Lamsal BP (2021) Ultrasound-assisted extraction and modification of plant-based proteins: Impact on physicochemical, functional, and nutritional properties. Compr Rev Food Sci Food Saf 20(2):1457–1480
Rais A, Jabeen Z, Shair F, Hafeez F, Hassan M (2017) Bacillus spp. a bio-control agent enhances the activity of antioxidant defense enzymes in rice against Pyricularia oryzae. PLoS ONE 12:e0187412. https://doi.org/10.1371/journal.pone.0187412
Raja Gopalan NS, Sharma R, Mohapatra S (2022) Probing into the unique relationship between a soil bacterium, Pseudomonas putida AKMP7 and Arabidopsis thaliana: a case of “conditional pathogenesis.” Plant Physiol Biochem 183:46–55. https://doi.org/10.1016/j.plaphy.2022.05.003
Rajwar A, Sahgal M (2016) Phylogenetic relationships of fluorescent pseudomonads deduced from the sequence analysis of 16S rRNA, Pseudomonas-specific and rpoD genes. 3 Biotech 6(1):80. https://doi.org/10.1007/s13205-016-0386-x
Sakakibara H (2006) Cytokinins: activity, biosynthesis, and translocation. Annu Rev Plant Biol 57:431–449. https://doi.org/10.1146/annurev.arplant.57.032905.105231
Sánchez Márquez M, Bills GF, Zabalgogeazcoa I (2007) The endophytic mycobiota of the grass Dactylis glomerata. Fungal Divers 27:10–31
Shandeep SG, Shanthi A, Kalaiarasan P, Swarnapriya R (2021) Compatibility Studies between Different Bacterial and Fungal Biocontrol Agents and Neem cake for Management of Root Knot Nematode, Meloidogyne incognita in okra. J Pharma Innov 10(12):73–76
Sinclair JB, Dhingra OD (2017) Basic plant pathology methods. CRC Press, Boca Raton, pp 287–300. https://doi.org/10.1201/9781315138138.
Singh R, Kumar V, Jalali BL (2003) Evaluation of resistance to stem rot and yield losses caused by the disease in rice. Indian Phytopathol 403–407.
Singh JS, Koushal S, Kumar A, Vimal SR, Gupta VK (2016) Book review: microbial inoculants in sustainable agricultural Productivity-Vol. II: functional application. https://doi.org/10.3389/fmicb.2016.02105.
Soesanto L, Mugiastuti E, Rahayuniati R (2011) Morphological and physiological features of Pseudomonas fluorescens P60 ER—4th International seminar of Indonesian society for microbiology.
Spaepen S, Vanderleyden J (2011) Auxin and plant-microbe interactions. Cold Spring Harb Perspect Biol 3(4):a001438. https://doi.org/10.1101/cshperspect.a001438
Stuti S, Shweena K, Rajni S (2021) Pseudomonas mediated nutritional and growth promotional activities for sustainable food security. Microbiol Curr Res 2:2666–5174. https://doi.org/10.1016/j.crmicr.2021.100084
Sun JQ, Guo LD, Zang W, Ping WX, Chi DF (2008) Diversity and ecological distribution of endophytic fungi associated with medicinal plants. Life Sci 51:751–759
Swapnil P, Meena M, Singh SK, Dhuldhaj UP, Harish Marwal A (2021) Vital roles of carotenoids in plants and humans to deteriorate stress with its structure, biosynthesis, metabolic engineering and functional aspects. Curr Plant Biol 26:00203. https://doi.org/10.1016/j.cpb.2021.100203
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Thesiya M, Rakholiya K, Shekhada M (2020) Comparative efficacy of fungicides and biocontrol agents against stem blight and fruit rot disease of brinjal under pot culture conditions. Int J Curr Microbiol Appl Sci. https://doi.org/10.20546/ijcmas.2020.910
Tian J, Ge F, Zhang D, Deng S, Liu X (2021) Roles of phosphate solubilizing microorganisms from managing soil phosphorus deficiency to mediating biogeochemical P cycle. Biol J 10(2):158. https://doi.org/10.3390/biology10020158
Vanitha S, Umesha S (2011) Pseudomonas fluorescens mediated systemic resistance in tomato is driven through an elevated synthesis of defense enzymes. Biol Plant 55:317–322. https://doi.org/10.1007/s10535-011-0045-3
Vinay K, Lata J, Sanjay KJ, Sameer C, Pankaj K (2020) Bacterial endophytes of rice (Oryza sativa L.) and their potential for plant growth promotion and antagonistic activities. S Afr J Bot 134:50–63. https://doi.org/10.1016/j.sajb.2020.02.017
Wang GL, Jiang BH, Rue EA, Semenza GL (1995) Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci 92(12):5510–5514
Wang X, Li Q, Sui J, Zhang J, Liu Z, Du J, Xu R, Zhou Y, Liu Z (2019) Isolation and characterization of antagonistic bacteria Paenibacillus jamilae HS-26 their effects on plant growth. Biomed Res Int 19:1–13
War AR, Paulraj MG, War MY, Ignacimuthu S (2011) Role of salicylic acid in induction of plant defense system in chickpea (Cicer arietinum L.). Plant Signal Behav 6(11):1787–1792. https://doi.org/10.4161/psb.6.11.17685
Wen-Ming W, Jing F, John MJJ (2019) Rice False Smut: An Increasing Threat to Grain Yield and Quality, Protecting Rice Grains in the Post-Genomic Era. Intech Open, Rijeka. https://doi.org/10.5772/intechopen.84862.
Wiesel L, Newton AC, Elliott I, Booty D, Gilroy EM, Birch PRJ, Hein I (2014) Molecular effects of resistance elicitors from biological origin and their potential for crop protection. Plant Signal Behav 5:655. https://doi.org/10.3389/fpls.2014.00655
Xie XL, Mai YW, Zhou XP (2005) Dispersion and alignment of carbon nanotubes in polymer matrix: a review. Mater Sci Eng R Rep 49(4):89–112. https://doi.org/10.1016/j.mser.2005.04.002
Zeng Q, Man X, Huang Z, Zhuang L, Yang H, Sha Y (2023) Effects of rice blast biocontrol strain Pseudomonas alcaliphila Ej2 on the endophytic microbiome and proteome of rice under salt stress. Front Microbiol 14:1129614. https://doi.org/10.3389/fmicb.2023.1129614
Acknowledgements
The authors thank Mrs. Jyothi Chaitanya and Mr. Gnanadhar for their help towards this work. We would also like to acknowledge the help and support received from the Department of plant pathology, PJTSAU, and ICAR-IIRR, Rajendranagar, Hyderabad for this study.
Funding
This study was funded by DARE, ministry of government, India. corresponding author has received research support from ICAR-IIRR.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by SV, MP, RGNS, KC is the principal investigator of this work. Contributed reagents/materials/analysis tools by BM, LLD, KP, JG, SR, SPM, SRM. The manuscript was written by SV, MP, RGNS, SM, UDG, RP commented and checked the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This research has no potential conflicts of interest and did not involve Human Participants and/or Animals.
Consent to participate
All authors agreed with the content of the work and with explicit consent for submission.
Consent for publication
All authors approved the version to be published.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vanama, S., Gopalan, N.S.R., Pesari, M. et al. Native bio-control agents from the rice fields of Telangana, India: characterization and unveiling the potential against stem rot and false smut diseases of rice. World J Microbiol Biotechnol 40, 2 (2024). https://doi.org/10.1007/s11274-023-03782-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-023-03782-2