Abstract
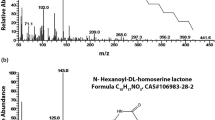
Acidovorax avenae subsp. citrulli is a Gram-negative bacterium and is the causal agent of bacterial fruit blotch (BFB) in cucurbits. In this study, the role played by the acyl-homoserine lactone (AHL)-type quorum sensing (QS) system in growth, swimming motility and virulence was characterized in A.avenae subsp. citrulli strain XJL12. The AHL synthase gene of the QS system from strain XJL12, defined as aacI, was cloned and characterized, and an aacI disruption mutant was generated. The aacI mutant XJL13 abolished the ability to produce AHL molecules, whereas the corresponding complemented strain CPXJL13 produced wild-type levels of AHL. The aacI mutant exhibited a significant decrease in growth rate relative to the wild type in minimal medium, and was partially impaired in swimming motility. In plants, the aacI mutant showed a significant reduction of virulence in watermelon fruits and melon seedlings when compared to the wild-type strain. However, the aacI mutation in strain XJL12 had no effects on biofilm formation, exopolysaccharide production, or induction of hypersentitive response in Nicotiana tabacum. Our data suggest that the AHL-type QS may play a key role in pathogen virulence and this may provide an opportunity to explore novel approaches for managing BFB in cucurbits by QS interference.






Similar content being viewed by others
References
Andersson RA, Eriksson ARB, Heikinheimo R, Mae A, Pirhonen M, Koiv V, HyytiĂinen H, Tuikkala A, Palvae T (2000) Quorum sensing in the plant pathogen Erwinia carotovora subsp. carotovora: the role of expREcc. Mol Plant Microbe Interact 13(4):384–393
Atkinson S, Chang CY, Sockett RE, Cámara M, Williams P (2006) Quorum sensing in Yersinia enterocolitica controls swimming and swarming motility. J Bacteriol 188:1451–1461
Balestrino D, Haagensen JA, Rich C, Forestier C (2005) Characterization of type 2 quorum sensing in Klebsiella pneumoniae and relationship with biofilm formation. J Bacteriol 187:2870–2880
Bassler BL (1999) How bacteria talk to each other: regulation of gene expression by quorum sensing. Curr Opin Microbiol 2:582–587
Burdman S, Kots N, Kritzman G, Kopelowitz J (2005) Molecular, physiological, and host-range characterization of Acidovorax avenae subsp. citrulli isolates from watermelon and melon in Israel. Plant Dis 89:1339–1347
Chen L, Hu BS, Qian GL, Wang C, Yang WF, Han ZC, Liu FQ (2009a) Identification and molecular characterization of twin-arginine translocation system (Tat) in Xanthomonas oryzae pv. oryzae strain PXO99. Arch Microbiol 191:163–170
Chen T, Qian GL, Yang XL, Ma JY, Hu BS, Liu FQ (2009b) Detection of quorum sensing signal molecule of Acidovorax avenae subsp. citrulli and its regulation on pathogenicity. Chin J Agri Biotechnol 6:49–53
Chesnokova O, Coutinho JB, Khan IH, Mikhail MS, Kado CI (1997) Characterization of flagella genes of Agrobacterium tumefaciens, and the effect of a bald strain on virulence. Mol Microbiol 23:579–590
Daniels MJ, Barber CE, Turner PC, Sawczyc MK, Byrde RJW, Fielding AH (1984) Cloning of genes involved in pathogenicity of Xanthomonas campestris pv. campestris using the broad host range cosmid pLAFR1. EMBO J 3:3323–3328
De Kievit TR, Iglewski BH (2000) Bacterial quorum sensing in pathogenic relationships. Infect Immun 68:4839–4849
Dong YH, Xu JL, Li XZ, Zhang LH (2000) AiiA, an enzyme that inactivates the acylhomoserine lactone quorumsensing signal and attenuates the virulence of Erwinia carotovora. Proc Natl Acad Sci USA 97:3526–3531
Dong YH, Gusti AR, Zhang Q, Xu JL, Zhang LH (2002) Identification of quorum-quenching N-acyl homoserine lactonases from Bacillus species. Appl Environ Microbiol 68:1754–1759
Feldman M, Bryan R, Rajan S, Scheffler L, Brunnert S, Tang H, Prince A (1998) Role of flagella in pathogenesis of Pseudomonas aeruginosa pulmonary infection. Infect Immun 66:43–51
Fessehaie A, Walcott RR (2005) Biological control to protect watermelon blossoms and seed from infection by Acidovorax avenae subsp. citrulli. Phytopathol 95(4):413–419
Fray RG, Throup JP, Daykin M, Wallace A, Williams P, Stewart GSAB, Grierson D (1999) Plants genetically modified to produce N-acylhomoserine lactones communicate with bacteria. Nat Biotechnol 17:1017–1020
Fuqua C, Greenberg EP (1998) Self perception in bacteria: quorum sensing with acylated homoserine lactones. Curr Opin Microbiol 1:183–189
Fuqua WC, Winans SC (1994) A LuxR-LuxI type regulatory system activates Agrobacterium Ti plasmid conjugal transfer in the presence of a plant tumor metabolite. J Bacteriol 176:2796–2806
Gao Y, Song JX, Hu BS, Zhang L, Liu QQ, Liu FQ (2009) The luxS gene is involved in AI-2 production, pathogenicity, and some phenotypes in Erwinia amylovora. Curr Microbiol 58:1–10
Greenberg EP (1997) Quorum sensing in gram-negative bacteria. ASM News 63:371–377
Hoang TT, Karkhoff-Schweizer RR, Kutchma AJ, Schweizer HP (1998) A broad-host-range Flp-FRT recombination system for site-specific excision of chromosomally-located DNA sequences: application for isolation of unmarked Pseudomonas aeruginosa mutants. Gene 212:77–86
Hopkins DL, Cucuzza JD, Watterson JC (1996) Wet seed treatments for the control of bacterial fruit blotch of watermelon. Plant Dis 80:529–532
Hu XJ, Roberts DP, Jiang ML, Zhang YB (2005) Decreased incidence of disease caused by Sclerotinia sclerotiorum and improved plant vigor of oilseed rape with Bacillus subtilis Tu-100. Appl Microbiol Biotechnol 68:802–807
Huang JJ, Han JI, Zhang LH, Leadbetter JR (2003) Utilization of acyl-homoserine lactone quorum signals for growth by a soil pseudomonad and Pseudomonas aeruginosa PAO1. Appl Environ Microbiol 69:05941–05949
Huber B, Riedel K, Riedel K, Hentzer M, Heydorn A, Gotschlich A, Givskov M, Molin S, Eberl L (2001) The cep quorum-sensing system of Burkholderia cepacia H111 controls biofilm formation and swarming motility. Microbiology 147:2517–2528
Hussain MBBM, Zhang HB, Xu JL, Liu QG, Jiang ZD, Zhang LH (2008) The acyl-homoserine lactone-type quorum sensing modulates cell motility and virulence of Eriwinia charysanthemi pv. zeae. J Bacteriol 190(3):1045–1053
Khajanchi BK, Sha J, Kozlova EV, Erova TE, Suarez G, Sierra JC, Popov VL, Horneman AJ, Chopra AK (2009) N-Acylhomoserine lactones involved in quorum sensing control the type VI secretion system biofilm formation, protease production and in vivo virulence in a clinical isolate of Aeromonas hydrophila. Microbiol 155:3518–3531
Koutsoudis MD, Tsaltas D, Minogue TD, Von Bodman SB (2006) Quorum sensing regulation governs bacterial adhesion, biofilm development, and host colonization in Pantoea stewartii subspecies stewartii. Proc Natl Acad Sci 103(15):5983–5988
Kovach ME, Elzer PH, Hill DS, Robertson GT, Farris MA, Roop RM II, Peterson KM (1995) Four new derivatives of the broad-host-range cloning vector pBBR1MCS, carrying different antibiotic-resistance cassettes. Gene 166:175–176
Lázaro M, Fabio R, Geneviève D, Brion D (2005) Autoinduction in Erwinia amlyvora: evidence of an acyl-homoserine lactone signal in the fire blight pathogen. J Bacteriol 187(9):3206–3213
Lemon KP, Higgins DE, Kolter R (2007) Flagellar motility is critical for Listeria monocytogenes biofilm formation. J Bacteriol 189(12):4418–4424
Lin YH, Xu JL, Hu J, Wang LH, Ong SL, Leadbetter JR, Zhang LH (2003) Acyl-homoserine lactone acylase from Ralstonia strain XJ12B represents a novel and potent class of quorum-quenching enzymes. Mol Microbiol 47:849–860
Maddula VS, Zhang Z, Pierson EA, Pierson LS (2006) Quorum sensing and phenazines are involved in Biofilm formation by Pseudomonas chlororaphis (aureofaciens) strain 30–84. Microb Ecol 52(2):289–301
Mäe A, Montesano M, Koiv V, Tapio-Palva E (2001) Transgenic plants producing the bacterial pheromone N-acyl-homoserine lactone exhibit enhanced resistance to the bacterial phytopathogen Erwinia carotovora. Mol Plant Microbe Interact 14:1035–1042
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Miller MB, Bassler BL (2001) Quorum sensing in bacteria. Annu Rev Microbiol 55:165–199
Mobley HL, Belas R, Lockatell V, Chippendale G, Trifillis AL, Johnson DE, Warren JW (1996) Construction of a flagellum-negative mutant of Proteus mirabilis: effect on internalization by human renal epithelial cells and virulence in a mouse model of ascending urinary tract infection. Infect Immun 64:5332–5340
Novick RP, Muir TW (1999) Virulence gene regulation by peptides in staphylococci and other Gram-positive bacteria. Curr Opin Microbiol 2:40–45
Palumbo J, Yuen G, Jochum C, Tatum K, Kobayashi D (2005) Mutagenesis of b-1, 3-glucanase genes in Lysobacter enzymogenes strain C3 results in reduced biological control activity towards Bipolaris leaf spot of tall fescue and Pythium damping off of sugarbeet. Phytopathology 95:701–707
Park SY, Lee SJ, Oh TK, Oh JW, Koo BT, Yum DY, Lee JK (2003) AhlD, an N-acylhomoserine lactonase in Arthrobacter sp., and predicted homologues in other bacteria. Microbiology 149:1541–1550
Pesci EC, Pearson JP, Seed PC, Iglewski BH (1997) Regulation of las and rhl quorum sensing in Pseudomonas aeruginosa. J Bacteriol 179(10):3127–3132
Quinones B, Pujol CJ, Lindow SE (2004) Regulation of AHL production and its contribution to Epiphytic fitness in Pseudomonas syringae. Mol Plant Microbe Interact 17(5):521–531
Quiñones B, Dulla G, Lindow SE (2005) Quorum sensing regulates exopolysaccharide production, motility, and virulence in Pseudomonas syringae. Mol Plant Microbe Interact 18(7):682–693
Ray SK, Rajeshwari R, Sonti RV (2000) Mutants of Xanthomonas oryzae pv. oryzae deficient in general secretory pathway are virulence deficient and unable to secrete xylanase. Mol Plant Microbe Interact 13:394–401
Reimmann C, Ginet N, Michel C, Keel C, Michanux P, Zala M, Heurlier K, Triandafillu K, Harms H, Défago G, Haas D (2002) Genetically programmed autoinducer destruction reduces virulence gene expression and swarming motility in Pseudomonas aeruginosa PAO1. Microbiology 148:923–932
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory mannual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Schaad NW, Sowell G, Goth RW, Colwell RR, Webb RE (1978) Pseudomonas pseudoalcaligenes subsp. citrulli subsp. nov. Int J Syst Bact 28:117–125
Shaw PD, Ping G, Daly SL, Cha C, Cronan JE Jr, Rinehart KL, Farrand SK (1997) Detecting and characterizing N-acyl-homoserine lactone signal molecules by thin-layer chromatography. Proc Natl Acad Sci 94:6036–6041
Simon R, Priefer U, Pühler A (1983) A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in gram negative bacteria. Biotechnology 1:787–796
Somodi GC, Jones JB, Hopkins DL, Stall RE, Kucharek TA, Hodge NC, Watterson JC (1991) Occurrence of bacterial watermelon fruit blotch in Florida. Plant Dis 75:1053–1056
Tang JL, Feng JX, Li QQ, Wen HX, Zhou DL, Wilson TJG (1996) Cloning and characterization of the rpfC gene of Xanthomonas oryzae pv. oryzae: involvement in exopolysaccharide production and virulence to rice. Mol Plant Microbe Interact 9:664–666
Von Bodman SB, Bauer WD, Coplin DL (2003) Quorum sensing in plant-pathogenic bacteria. Annu Rev Phytopathol 41:455–482
Walcott RR, Gitaitis RD (2000) Detection of Acidovorax avenae subsp. citrulli in watermelon seed using immunomagetic separation and the polymerase chain reaction. Plant Dis 84:470–474
Walcott RR, Langston DB Jr, Sanders FH Jr, Gitaitis RD (2000) Investigating intraspecific variation of Acidovorax avenae subsp. citrulli using DNA fingerprinting and whole fatty cell acid analysis. Phytopathology 90:191–196
Walcott RR, Fessehaie A, Castro AC (2004) Differences in pathogenicity between two genetically distinct groups of Acidovorax avenae subsp. citrulli on cucurbit hosts. J Phytopathol 152:277–285
Wang Y, Zheng HM, Yang MH, Zhong ZT, Zhu J (2007) Screening for autoinducer synthase gene in Sinorhizobium meliloti and analysis of the autoinducer produced by recombinant expression in Escherichia coli. Acta Microbiol Sin 47(5):838–842
Wang P, Zhong ZT, Zhou J, Cai T, Zhu J (2008) Exopolysaccharide biosynthesis is important for Mesorhizobium tianshanense: plant host interaction. Arch Microbiol 189:525–530
Want WZ, Morohoshi T, Ikenoya M, Someya N, Ikeda T (2010) AiiM, a novel class of N-acylhomoerine lactonase from the leaf-associated bacterium Microbacterium testaceum. Appl Environ Microbiol 76:2524–2530
Waters C, Bassler B (2005) Quorum sensing: cell-to-cell communication in bacteria. Annu Rev Cell Dev Biol 21:319–346
Whitehead NA, Barnard AM, Slater H, Simpson NJ, Salmond GP (2001) Quorum-sensing in gram-negative bacteria. FEMS Microbiol Rev 25:365–404
Willems A, Goor M, Thielemans S, Gillis M, Kersters K, Ley JD, De Ley J (1992) Transfer of several phytopathogenic Pseudomonas species to Acidovorax as Acidovorax avenae subsp. avenae subsp. nov., comb. nov., Acidovorax avenae subsp. citrulli, Acidovorax avenae subsp. cattleyae, and Acidovorax konjaci. Int J Syst Bact 42:107–119
Williams P, Camara M, Hardman A, Swift S, Milton D, Hope VJ, Winzer K, Middleton B, Pritchard DI, Bycroft BW (2000) Quorum sensing and the population dependent control of virulence. Philos Trans R Soc Lond B Biol Sci 355:667–680
Withers H, Swift S, Williams P (2001) Quorum sensing as an integral component of gene regulatory networks in Gram-negative bacteria. Curr Opin Microbiol 4:186–193
Yin XT, Xu L, Fan SS, Xu LN, Li DC, Liu ZY (2010) Isolation and characterization of an AHL lactonase gene from Bacillus amyloliquefaciens. World J Microbiol Biotechnol 26:1361–1367
Zhang HB, Wang LH, Zhang LH (2002) Genetic control of quorum-sensing signal turnover in Agrobacterium tumefaciens. Proc Natl Acad Sci 99(7):4638–4643
Zheng HM, Zhong ZT, Lai X, Chen WX, Li SP, Zhu J (2006) A LuxR/LuxI-type quorum sensing system in a plant bacterium, Mesorhizobium tianshanense, controls symbiotic nodulation. J Bacteriol 188:1943–1949
Zhou H, Yao F, Roberts DP, Lessie TG (2003) AHL-deficient mutants of Burkholderia ambifaria BC-F have decreased antifungal activity. Curr Microbiol 47(3):174–179
Zhu J, Chai Y, Zhong Z, Li S, Winans SC (2003) Agrobacterium bioassay strain for ultrasensitive detection of N-acylhomoserine lactone-type quorum-sensing molecules: detection of autoinducers in Mesorhizobium huakuii. Appl Environ Microbiol 69:6949–6953
Acknowledgments
We are very grateful to professor Jun Zhu (Nanjing Agricultural University) for kindly providing AHL bioassay strain JZA1 and A. tumefaciens R10. We thank Dr. Tao Cai (Nanjing Agricultural University) for the technical assistant in AHL bioassay. We also thank Becky Shirk (University of Geogia, Dr. Suomeng Dong and Yu Chen (Nanjing Agricultural University) for their critical revisions of this manuscript. This work was supported by the National Basic Research program of China (“973” program, No. 2009CB119200), a U.S. National Science Foundation PIRE Grant (OISE 0730218) and Science and Technology support XinJiang project (No. 200840102-03).
Author information
Authors and Affiliations
Corresponding author
Additional information
Jiaqin Fan, Guoliang Qian, Tao Chen contribute equally to this research.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fan, J., Qian, G., Chen, T. et al. The acyl-homoserine lactone (AHL)-type quorum sensing system affects growth rate, swimming motility and virulence in Acidovorax avenae subsp. citrulli . World J Microbiol Biotechnol 27, 1155–1166 (2011). https://doi.org/10.1007/s11274-010-0562-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-010-0562-9