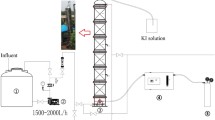
Abstract
Industrial production of manganese/silicon ore has generated a large number of tailing wastes which are difficult to dispose. A new method treating coking wastewater was proposed using the manganese/silicon tailing waste and demonstrated with good performances: the chemical oxygen demand (COD) removal rate was around 60 % without pH and temperature adjustment, with a reasonable reaction time of 2.5 h and tailing dosage of 0.2 g/L; while phenylamine was eliminated with a removal rate as high as 99 and 61.6 % for synthetic and real coking wastewater, respectively. Experimental results indicated that the removal of organic pollutants was mainly realized through chemical adsorption and/or oxidation by oxide components inside the tailing, rather than by physical adsorption. Operational parameters such as tailing dosage, reaction time, and temperature were optimized. Acid conditions were found to be favorable to remove the selected model organic pollutants, i.e., volatile phenols and phenylamine. Fortunately, the optimistic wastewater pH for COD removal was found to be around 7.0, right within the range of influent pH for real coking wastewater. The new method can treat coking wastewater and reuse mining tailing wastes simultaneously.






Similar content being viewed by others
References
Ajmal, M., Rao, R. A. K., Ahmad, R., & Ahmad, J. (2000). Adsorption studies on Citrus reticulata (fruit peel of orange): removal and recovery of Ni(II) from electroplating wastewater. Journal of Hazardous Materials, 79(1–2), 117–131.
Al-Johani, H., & Salam, M. A. (2011). Kinetics and thermodynamic study of aniline adsorption by multi-walled carbon nanotubes from aqueous solution. Journal of Colloid and Interface Science, 360(2), 760–767.
American Public Health Association (APHA), A.W.W.A.A., Water Environment Federation (WEFW). 1998. Standard methods for the examination of water and wastewater. 20th ed, Washington, DC.
Asghar, H. M. A., Hussain, S. N., Roberts, E. P. L., Campen, A. K., & Brown, N. W. (2013). Pre-treatment of adsorbents for waste water treatment using adsorption coupled-with electrochemical regeneration. Journal of Industrial and Engineering Chemistry, 19(5), 1689–1696.
Ba, Y.E., Li, Z.F. 2012. Study of the treatment of coking wastewater by biological nitrogen removal with physical-chemical process. Manufacturing Science and Technology, Pts 1-8, 383-390, 3729-3733.
Bai, X. (2010). Adsorption of organic pollutants from coking wastewater by activated coke. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 362(1), 140–146.
Blanco, G., Cauqui, M. A., Delgado, J. J., Galtayries, A., Perez-Omil, J. A., & Rodriguez-Izquierdo, J. M. (2004). Preparation and characterization of Ce-Mn-O composites with applications in catalytic wet oxidation processes. Surface and Interface Analysis, 36(8), 752–755.
Burmistrz, P., Rozwadowski, A., Burmistrz, M., & Karcz, A. (2014). Coke dust enhances coke plant wastewater treatment. Chemosphere, 117, 278–84.
Chang, E. E., Hsing, H. J., Chiang, P. C., Chen, M. Y., & Shyng, J. Y. (2008). The chemical and biological characteristics of coke-oven wastewater by ozonation. Journal of Hazardous Materials, 156(1–3), 560–567.
Chen, J.-C., Huang, J.-S., Chen, C.-M., & Guo, J.-S. (2008). Emission characteristics of PAHs, benzene and phenol group hydrocarbons in O2/RFG waste incineration processes. Fuel, 87(12), 2787–2797.
Chen, T. H., Huang, X. M., Pan, M., Jin, S., Peng, S. C., & Fallgren, P. H. (2009). Treatment of coking wastewater by using manganese and magnesium ores. Journal of Hazardous Materials, 168(2–3), 843–847.
Chu, L., Wang, J., Dong, J., Liu, H., & Sun, X. (2012). Treatment of coking wastewater by an advanced Fenton oxidation process using iron powder and hydrogen peroxide. Chemosphere, 86(4), 409–414.
Cieślik, B.M., Namieśnik, J., Konieczka, P. 2014. Review of sewage sludge management: standards, regulations and analytical methods. Journal of Cleaner Production.
Jablonska, B. (2012). Sorption of phenol on rock components occurring in mine drainage water sediments. International Journal of Mineral Processing, 104, 71–79.
Jin, L., Zhang, G., Zheng, X. 2014. Effects of different sludge disintegration methods on sludge moisture distribution and dewatering performance. Journal of Environmental Sciences.
Kondratenko, E. V. (2010). Using time-resolved methods to monitor and understand catalytic oxidation reactions. Catalysis Today, 157(1–4), 16–23.
Kossoff, D., Dubbin, W. E., Alfredsson, M., Edwards, S. J., Macklin, M. G., & Hudson-Edwards, K. A. (2014). Mine tailings dams: characteristics, failure, environmental impacts, and remediation. Applied Geochemistry, 51, 229–245.
Li, H. B., Cao, H. B., Li, Y. P., Zhang, Y., & Liu, H. R. (2010). Innovative biological process for treatment of coking wastewater. Environmental Engineering Science, 27(4), 313–322.
Lim, B.-R., Hu, H.-Y., & Fujie, K. (2003a). Biological degradation and chemical oxidation characteristics of coke-oven wastewater. Water, Air, and Soil Pollution, 146(1–4), 23–33.
Lim, B. R., Hu, H. Y., & Fujie, K. (2003b). Biological degradation and chemical oxidation characteristics of coke-oven wastewater. Water Air and Soil Pollution, 146(1–4), 23–33.
Liu, T., Wu, K., & Zeng, L. H. (2012). Removal of phosphorus by a composite metal oxide adsorbent derived from manganese ore tailings. Journal of Hazardous Materials, 217, 29–35.
Maciuca, A. L., Dumitriu, E., Fajula, F., & Hulea, V. (2007). Catalytic oxidation processes for removing dimethylsulfoxide from wastewater. Chemosphere, 68(2), 227–233.
Oubagaranadin, J. U. K., & Murthy, Z. V. P. (2010). Isotherm modeling and batch adsorber design for the adsorption of Cu(II) on a clay containing montmorillonite. Applied Clay Science, 50(3), 409–413.
Rashed, M. N. (2010). Monitoring of contaminated toxic and heavy metals, from mine tailings through age accumulation, in soil and some wild plants at Southeast Egypt. Journal of Hazardous Materials, 178(1–3), 739–746.
Samolada, M. C., & Zabaniotou, A. A. (2014). Comparative assessment of municipal sewage sludge incineration, gasification and pyrolysis for a sustainable sludge-to-energy management in Greece. Waste Management, 34(2), 411–420.
Shao, D., Hu, J., Chen, C., Sheng, G., Ren, X., & Wang, X. (2010). Polyaniline multiwalled carbon nanotube magnetic composite prepared by plasma-induced graft technique and its application for removal of aniline and phenol. The Journal of Physical Chemistry C, 114(49), 21524–21530.
Smeink, R., & Mahy, J. (2005). Determination of preferential adsorption of Depramin® on mineral surfaces by ToF-SIMS. Minerals Engineering, 18(2), 247–255.
Song, G. X., Zhu, C. Y., Hu, Y. M., Chen, J. M., & Cheng, H. F. (2013). Determination of organic pollutants in coking wastewater by dispersive liquid-liquid microextraction/GC/MS. Journal of Separation Science, 36(9–10), 1644–1651.
Wang, J. L., Quan, X. C., Wu, L. B., Qian, Y., & Hegemann, W. (2002). Bioaugmentation as a tool to enhance the removal of refractory compound in coke plant wastewater. Process Biochemistry, 38(5), 777–781.
Wasylenki, L. E., Rolfe, B. A., Weeks, C. L., Spiro, T. G., & Anbar, A. D. (2008). Experimental investigation of the effects of temperature and ionic strength on Mo isotope fractionation during adsorption to manganese oxides. Geochimica Et Cosmochimica Acta, 72(24), 5997–6005.
Wenzl, T., Simon, R., Anklam, E., & Kleiner, J. (2006). Analytical methods for polycyclic aromatic hydrocarbons (PAHs) in food and the environment needed for new food legislation in the European Union. TrAC Trends in Analytical Chemistry, 25(7), 716–725.
Yang, Z.S., Hsu, F.H., Lin, L.Y., Lee, H.J., Lian, N.T., Yang, T., Chen, K.C., Lu, C.Y. 2013. Investigation of shape etching on multi-layer SiO2/Poly-Si for 3D NAND architecture. 2013 24th Annual Semi Advanced Semiconductor Manufacturing Conference (Asmc), 24-26.
Zhang, S., Zhao, X., Niu, H., Shi, Y., Cai, Y., & Jiang, G. (2009). Superparamagnetic Fe3O4 nanoparticles as catalysts for the catalytic oxidation of phenolic and aniline compounds. Journal of Hazardous Materials, 167(1), 560–566.
Zhang, W. H., Wei, C. H., Feng, C. H., Yan, B., Li, N., Peng, P. A., & Fu, J. M. (2012). Coking wastewater treatment plant as a source of polycyclic aromatic hydrocarbons (PAHs) to the atmosphere and health-risk assessment for workers. Science of the Total Environment, 432, 396–403.
Ziolek, M. (2006). Catalytic processes of liquid-phase oxidation. Przemysl Chemiczny, 85(2), 118–123.
Zuo, L. N., Song, W. Z., Shi, T. H., Lv, C., Yao, J., Liu, J. F., & Weng, Y. H. (2014). Adsorption of aniline on template-synthesized porous carbons. Microporous and Mesoporous Materials, 200, 174–181.
Acknowledgments
This study was financially supported by the Natural Science Foundation of China (NSFC No. 21206092) and RTTC-China Program (2014). The authors are very grateful to Dr. Xu Shuyun from the University of Shanghai for Science and Technology and Dr. Chen Xiaoguang from Donghua University for proof reading.
Conflict of Interest
The authors have declared no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Liu, H., Gao, S., Zhu, M. et al. Use of Manganese/Silicon Tailing Waste for Coking Wastewater Treatment: Evaluation of Phenol and Phenylamine Removal Efficiencies. Water Air Soil Pollut 226, 78 (2015). https://doi.org/10.1007/s11270-015-2303-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-015-2303-z