Abstract
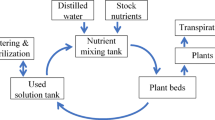
The aim of this study was examination possibility of Azolla caroliniana Willd. to aerate its medium enriched with Cd(II) and Hg(II) and comparison of DO and ODR methods in measurement aeration status of solutions. Azolla system has been chosen to verify the validity of the measuring ODR method in water solution. Water aeration measurements, one of important environmental tests, are performed most often by measurement of dissolved oxygen with oxygen sensors. Other similar method called oxygen diffusion rate is generally used in different porous materials such as soil. Our first objective was to check if these both methods are comparable and may be use exchangeable in water solution. The both types of measurements were performed in medium saturated at different oxygen concentrations. The linear relationship and high correlation (R = 0.89) were found between values of ODR and DO. The object of the second part of our studies was A. caroliniana Willd. (Azollaceae), a floating water fern living in symbiosis with cyanobacterium Anabaena azollae Strasb. (Nostoceae) that fixes atmospheric nitrogen. Azolla plants are used for centuries as a nitrogen biofertilizer. The second aim of our work was to find out, if the fern reduces or increases oxygen concentration in water. The ODR method was used to determine the aeration status of the nutrient solution. During 12 days of the experiment at laboratory conditions, an active role of A. caroliniana in aeration of the nutrient solution containing Cd(II) and Hg(II) was stated.







Similar content being viewed by others
References
Ashton, P. J., & Walmsey, R. D. (1976). The aquatic fern Azolla and its Anabaena symbiont. Endeavour, 35, 39–45.
Bennicelli, R., Stępniewska, Z., Banach, A., Szajnocha, K., & Ostrowski, J. (2004). The ability of Azolla caroliniana to remove heavy metals (Hg(II), Cr(III), Cr(VI)) from municipal waste water. Chemosphere, 55, 141–146.
Breitburg, D. L. (2002). Effects of hypoxia, and balance between hypoxia and enrichment, on coastal fishes and fisheries. Estuaries, 25, 767–781.
Carpenter, J. H. (1965). The Chesapeake Bay Institute technique for the Winkler dissolved oxygen method. Limnology and Oceanography, 10, 141–143.
Carrapiço, F., & Tavares, R. (1989a). New data on the Azolla–Anabaena symbiosis. I. Morphological and histochemical aspect. In A. Skinner, R. M. Boddey, & I. Frederik (Eds.), Nitrogen fixation with non-legume (pp. 89–94). Dordrecht, The Netherlands: Kluwer.
Carrapiço, F., & Tavares, R. (1989b). New data on the Azolla–Anabaena symbiosis. II. Cytochemical and immunocytochemical. In A. Skinner, R. M. Boddey, & L. Frederik (Eds.), Nitrogen fixation with non-legume (pp. 95–100). Dordrecht, The Netherlands: Kluwer.
Cavallini, A., Natali, L., Durante, M., & Maserti, B. (1999). Mercury uptake, distribution and DNA affinity in durum wheat (Triticum durum Desf.) plants. Science of the Total Environment, 243/244, 119–127.
Chen, Y. X., He, Y. M., Luo, Y. M., Yu, Y. L., Lin, Q., & Wong, M. H. (2003). Physiological mechanism of plant roots exposed to cadmium. Chemosphere, 50, 789–793.
Connell, E. L., Colmer, T. D., & Walker, D. I. (1999). Radial oxygen loss of Halophila ovalis as a function of distance behind the root tip and shoot illumination. Aquatic Botany, 63, 219–228.
Culberson, C. H. (1991). Dissolved oxygen. WHP operations and methods, July, pp. 1–15.
Dat, J. F., Capelli, N., Folzer, H., Bourgesde, P., & Badot, P. M. (2004). Sensing and signalling during plant flooding. Plant Physiology and Biochemistry, 42, 273–282.
Dawar, S., & Singh, P. K. (2001). Growth, nitrogen fixation and occurrence of epiphytic algae at different pH in the cultures of two species of Azolla. Biology and Fertility of Soils, 34, 210–214.
Dilhan, M., Estève, D., Gué, A. M., Mauvais, O., & Mercier, L. (1995). Electrochemical oxygen microsensors. Sensors and Actuators B, 26–27, 401–403.
Gliński, J., & Stępniewski, W. (1985). Oxygen diffusion rate (ODR). In Soil aeration and its role for plants (pp. 181–186). Boca Raton, Florida: CRC.
Hechler, W. D., & Dawson, J. O. (2000). Factors affecting nitrogen fixation in Azolla caroliniana. Transactions of the Illinois State Academy of Science, 88(3, 4), 97–107.
Jespersen, D. N., Sorrell, B. K., & Brix, H. (1998). Growth and root oxygen release by Typha latifolia and its effects on sediment methanogenesis. Aquatic Botany, 61, 165–180.
Lemon, E. R., & Erickson, A. E. (1952). The measurement of oxygen diffusion in the soil with a platinum microelectrode. Soil Science, 16, 160.
Lucassen, E. C. H. E. T., Bobbink, R., Smolders, A. J. P., Van der Ven, P. J. M., Lamers, L. P. M., & Roelofs, J. G. M. (2002). Interactive effects of low pH and high ammonium levels responsible for the decline of Cirsium dissectum (L.) Hill. Plant Ecology, 165, 45–52.
Lucassen, E. C. H. E. T., Smolders, A. J. P., & Roelofs, J. G. M. (2000). Increased groundwater levels cause iron toxicity in Glyceria fluitans (L.). Aquatic Botany, 66, 321–327.
Malicki, M., & Walczak, R. (1983). A gauge for redox potential and the oxygen diffusion rate in the soils with automatic regulation of cathode potential. Zeszyty Problemowe Postêpów Nauk Rolniczych, 220, 447–452.
Mandal, B., Vlek, P. L. G., & Mandal, L. N. (1999). Beneficial effects of blue-green algae and Azolla, excluding supplying nitrogen, on wetland rice fields: A review. Biology and Fertility of Soils, 28, 329–342.
McConnachie, A. J., de Wit, M. P., Hill, M. P., & Byrne, M. J. (2003). Economic evaluation of the successful biological control of Azolla filiculoides in South Africa. Biological Control, 28, 25–32.
Meharg, A. A. (1994). Integrated tolerance mechanisms: Constitutive and adaptive plant responses to elevated metal concentrations in the environment. Plant, Cell and Environment, 17, 989–993.
Mommer, L., Pedersen, O., & Visser, E. J. W. (2004). Acclimation of a terrestrial plant to submergence facilitates gas exchange under water. Plant, Cell and Environment, 27, 1281–1287.
Nei, L., & Compton, R. G. (1996). An improved Clark-type galvanic sensor for dissolved oxygen. Sensors and Actuators B, 30, 83–87.
Prasand, M. N. V. (1995). Cadmium toxicity and tolerance in vascular plants. Environmental and Experimental Botany, 35(4), 525–545.
Sanità di Toppi, L., & Gabbrielli, R. (1999). Response to cadmium in higher plants. Environmental and Experimental Botany, 41, 105–130.
Snowden, R. E. D., & Wheeler, B. D. (1993). Iron toxicity to fen plant species. Journal of Ecology, 81(1), 35–46.
Stępniewska, Z., Bennicelli, R. P., Balakhina, T. I., Szajnocha, K., Banach, A., & Wolińska, A. (2005). Potential of Azolla caroliniana for the removal of Pb and Cd from wastewaters. International Agrophysics, 19(3), 251–256.
Susarla, S., Medina, V. F., & McCutcheon, S. (2002). Phytoremediation: An ecological solution to organic chemical contamination. Ecological Engineering, 18, 647–658.
Vasailev, A., Tsonev, T., & Yordanov, I. (1998). Physiological response of barley plants (Hordeum vulgare) to cadmium contamination in soil during ontogenesis. Environmental Pollution, 103, 287–293.
Visser, E. J. W., Colmer, T. D., Blom, C. W. P. M., & Voesenek, L. A. C. J. (2000). Changes in growth, porosity, and radial oxygen loss from adventitious roots of selected mono- and dicotyledonous wetland species with contrasting types of aerenchyma. Plant, Cell and Environment, 23, 1237–1245.
Voesenek, L. A. C. J., Armstrong, W., Bőgemann, G. M., McDonald, M. P., & Colmer, T. D. (1999). A lack of aerenchyma and high rates of radial oxygen loss from the root base contribute to the waterlogging intolerance of Brassica napus Aust. Journal of Plant Physiology, 26, 87–93.
Wackett, L. P. (2002). Mechanism and applications of Rieske non-heme iron dioxygenases. Enzyme and Microbial Technology, 31, 577–587.
Watanabe, I. (1982). Azolla–Anabaena symbiosis – Its physiology and use in tropical agriculture. In Y. R. Dommergues & H. G. Diem (Eds.), Microbiology of tropical soils and plant productivity (pp. 168–185). The Hague: Martinus Nijhoff.
Watanabe, I., Roger, P. A., Landha, J. K., & Van Hove, C. (1992). Biofertilizer germoplasm collections at IRRI. Manila: International Rice Research Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bennicelli, R.P., Stępniewska, Z., Banach, K. et al. Comparison of Aeration Status Measurements by Clark Sensor (DO) and ODR-Meter during Azolla caroliniana Willd. Growth in the Presence of Cd(II) and Hg(II). Water Air Soil Pollut 180, 29–37 (2007). https://doi.org/10.1007/s11270-006-9247-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-006-9247-2