Abstract
Introduction:
In vitro capacitation is essential in assisted reproductive technologies (ART) for embryo production. Recently, arginine has been proven to enhance capacitation in mammalian spermatozoa. However, the detailed mechanism of action of arginine remains elusive.
Aim:
This study investigated the effect of arginine-induced capacitation and motility enhancement on the spermatozoal RNA (spRNA) population in goats.
Material and methods:
Goat spermatozoa were treated with arginine for up to six hours and compared with non-treated or PHE (penicillamine, hypotaurine, and epinephrine)-treated spermatozoa at different intervals (0, 1, 2, 4, and 6 hours). Sperm parameters, including viability, individual motility, capacitation, acrosome reaction, and ROS production, were evaluated. The spRNA population was analyzed by short-read RNA sequencing (RNA-seq).
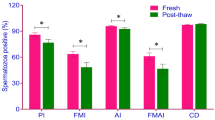
Results:
The percentage of capacitated (73.21 ± 4.22%) and acrosome reacted (18.35 ± 0.56%) spermatozoa was highest in arginine treatment, while PHE treatment showed the highest percentage (79.82 ± 4.31%) of motile spermatozoa from 0 to 4 hours of incubation. RNA-seq analysis identified 1,321 differentially expressed genes (DEGs) in arginine-treated spermatozoa compared to the control. The PGK2, RNASE10, ODF1, and ROPN1L genes involved in sperm motility and ACR, DKKL1, KCNJ11, and PRND genes involved in the capacitation process were upregulated in arginine-treated spermatozoa. The DEGs regulate sperm capacitation-related cAMP-PKA, PI3-Akt, calcium, and MAPK signaling pathways.
Conclusion:
The arginine-induced capacitation and enhanced sperm motility were associated with the upregulation of several genes involved in sperm motility and capacitation pathways. The comparative study also suggests that arginine may be used in lieu of PHE for motility enhancement and in vitro capacitation of goat spermatozoa.









Similar content being viewed by others
References
Almog T, Lazar S, Reiss N et al (2008) Identification of extracellular signal-regulated kinase 1/2 and p38 MAPK as regulators of human sperm motility and acrosome reaction and as predictors of poor spermatozoan quality. J Biol Chem 283:14479–14489. https://doi.org/10.1074/jbc.M710492200
Alonso CAI, Osycka-Salut CE, Castellano L et al (2017) Extracellular cAMP activates molecular signalling pathways associated with sperm capacitation in bovines. Mol Hum Reprod 23:521–534. https://doi.org/10.1093/molehr/gax030
Bergqvist A-S, Ballester J, Johannisson A et al (2007) Heparin and dermatan sulphate induced capacitation of frozen-thawed bull spermatozoa measured by merocyanine-540. Zygote 15:225–232. https://doi.org/10.1017/S0967199407004182
Bissonnette N, Lévesque-Sergerie JP, Thibault C, Boissonneault G (2009) Spermatozoal transcriptome profiling for bull sperm motility: A potential tool to evaluate semen quality. Reproduction 138:65–80. https://doi.org/10.1530/REP-08-0503
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Card CJ, Krieger KE, Kaproth M, Sartini BL (2017) Oligo-dT selected spermatozoal transcript profiles differ among higher and lower fertility dairy sires. Anim Reprod Sci 177:105–123. https://doi.org/10.1016/j.anireprosci.2016.12.011
Carrageta DF, Guerra-Carvalho B, Sousa M et al (2020) Mitochondrial activation and reactive oxygen-species overproduction during sperm capacitation are independent of glucose stimuli. Antioxidants 9:1–17. https://doi.org/10.3390/antiox9080750
Cavarocchi E, Whitfield M, Chargui A et al (2021) The sodium/proton exchanger SLC9C1 (sNHE) is essential for human sperm motility and fertility. Clin Genet 99:684–693. https://doi.org/10.1111/cge.13927
Chen J, Wang Y, Xu X et al (2009) [Differential expression of ODF1 in human ejaculated spermatozoa and its clinical significance]. Zhonghua Nan Ke Xue 15:891–894
Chen S, Zhou Y, Chen Y, Gu J (2018) Fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34:i884–i890. https://doi.org/10.1093/bioinformatics/bty560
Chen Y, Lun ATL, Smyth GK (2016) From reads to genes to pathways: Differential expression analysis of RNA-Seq experiments using rsubread and the edgeR quasi-likelihood pipeline. F1000Research 5:1–51. https://doi.org/10.12688/F1000RESEARCH.8987.2
Civetta A (2003) Positive selection within sperm-egg adhesion domains of fertilin: An ADAM gene with a potential role in fertilization. Mol Biol Evol 20:21–29. https://doi.org/10.1093/molbev/msg002
Curry E, Safranski TJ, Pratt SL (2011) Differential expression of porcine sperm microRNAs and their association with sperm morphology and motility. Theriogenology 76:1532–1539. https://doi.org/10.1016/j.theriogenology.2011.06.025
Dai D-H, Qazi I, Ran M-X et al (2019) Exploration of miRNA and mRNA profiles in fresh and frozen-thawed boar sperm by Transcriptome and small RNA sequencing. Int J Mol Sci 20:802. https://doi.org/10.3390/ijms20040802
Danshina PV., Geyer CB, Dai Q et al (2010) Phosphoglycerate kinase 2 (PGK2) is essential for sperm function and male fertility in mice. Biol Reprod 82:136–145. https://doi.org/10.1095/biolreprod.109.079699
Dobrinski I, Smith TT, Suarez SS, Ball BA (1997) Membrane contact with oviductal epithelium modulates the intracellular calcium concentration of equine spermatozoa in vitro. Biol Reprod 56:861–869. https://doi.org/10.1095/biolreprod56.4.861
El-Shahat KH, Taysser MI, Badr MR, Zaki KA (2017) Effects of penicillamine, hypotaurine, and epinephrine on motility, hyperactivity, acrosome reaction of fresh ram sperm. Asian Pac J Reprod 6:283–288. https://doi.org/10.4103/2305-0500.217344
Etkovitz N, Rubinstein S, Daniel L, Breitbart H (2007) Role of PI3-kinase and PI4-kinase in actin polymerization during bovine sperm capacitation. Biol Reprod 77:263–273. https://doi.org/10.1095/biolreprod.106.056705
Etkovitz N, Tirosh Y, Chazan R et al (2009) Bovine sperm acrosome reaction induced by G protein-coupled receptor agonists is mediated by epidermal growth factor receptor transactivation. Dev Biol 334:447–457. https://doi.org/10.1016/j.ydbio.2009.08.002
Ferreira LM, Garcia-Herreros M, Domingos A et al (2017) Prion protein 2 (dublet) gene (PRND): role in ovine semen capacitation, cryopreservation and fertility. Reprod Fertil Dev 29:985–997. https://doi.org/10.1071/RD15214
Fiedler SE, Dudiki T, Vijayaraghavan S, Carr DW (2013) Loss of R2D2 Proteins ROPN1 and ROPN1L Causes Defects in Murine Sperm Motility, Phosphorylation, and Fibrous Sheath Integrity1. Biol Reprod 88. https://doi.org/10.1095/biolreprod.112.105262
Fujihara Y, Murakami M, Inoue N et al (2010) Sperm equatorial segment protein 1, SPESP1, is required for fully fertile sperm in mouse. J Cell Sci 123:1531–1536. https://doi.org/10.1242/jcs.067363
Galantino-Homer HL, Florman HM, Storey BT et al (2004) Bovine sperm capacitation: Assessment of phosphodiesterase activity and intracellular alkalinization on Capacitation-Associated protein tyrosine phosphorylation. Mol Reprod Dev 67:487–500. https://doi.org/10.1002/mrd.20034
Gòdia M, Mayer FQ, Nafissi J et al (2018) A technical assessment of the porcine ejaculated spermatozoa for a sperm-specific RNA-seq analysis. Syst Biol Reprod Med 64:291–303. https://doi.org/10.1080/19396368.2018.1464610
Gonçalves FS, Barretto LSS, Arruda RP et al (2014) Heparin and penicillamine–hypotaurine–epinephrine (PHE) solution during bovine in vitro fertilization procedures impair the quality of spermatozoa but improve normal oocyte fecundation and early embryonic development. Vitr Cell Dev Biol - Anim 50:39–47. https://doi.org/10.1007/s11626-013-9675-4
Goodrich R, Johnson G, Krawetz SA (2007) The preparation of human spermatozoal RNA for clinical analysis. Arch Androl 53:161–167. https://doi.org/10.1080/01485010701216526
Grzmil P, Boinska D, Kleene KC et al (2008) Prm3, the fourth gene in the mouse protamine gene cluster, encodes a conserved acidic protein that affects sperm motility. Biol Reprod 78:958–967. https://doi.org/10.1095/biolreprod.107.065706
Guimarães Silva TV, da Silva BB, de Sá ALA et al (2014) Influence of L-arginine during bovine in vitro fertilization. Indian J Exp Biol 52:1159–1164
Gupta MK, Das ZC, Heo YT et al (2013) Transgenic chicken, mice, cattle, and pig embryos by somatic cell nuclear transfer into pig oocytes. Cell Reprogram 15:322–328. https://doi.org/10.1089/cell.2012.0074
Hassanpour H, Teshfam M, Goodarzi AK et al (2010) In vitro effects of l-arginine on motion parameters in ram epididymal sperm. Comp Clin Path 19:351–355. https://doi.org/10.1007/s00580-009-0876-x
Huang L, Xiao K, Zhang J et al (2021) Comparative transcriptome analysis reveals potential testosterone function-related regulatory genes/pathways of leydig cells in immature and mature buffalo (Bubalus bubalis) testes. Gene 802:145870. https://doi.org/10.1016/j.gene.2021.145870
Ickowicz D, Finkelstein M, Breitbart H (2012) Mechanism of sperm capacitation and the acrosome reaction: role of protein kinases. Asian J Androl 14:816–821. https://doi.org/10.1038/aja.2012.81
Jodar M (2019) Sperm and seminal plasma RNAs: what roles do they play beyond fertilization? Reproduction 158:R113–R123. https://doi.org/10.1530/REP-18-0639
Juma AR, Grommen SVH, O’Bryan MK et al (2017) PLAG1 deficiency impairs spermatogenesis and sperm motility in mice. Sci Rep 7:1–12. https://doi.org/10.1038/s41598-017-05676-4
KANG S-S, KOYAMA K, HUANG W et al (2015) Addition of D-penicillamine, hypotaurine, and epinephrine (PHE) mixture to IVF medium maintains motility and longevity of bovine sperm and enhances stable production of blastocysts in vitro. J Reprod Dev 61:99–105. https://doi.org/10.1262/jrd.2014-112
Kargar R, Forouzanfar M, Ghalamkari G, Nasr Esfahani MH (2017) Dietary flax seed oil and/or vitamin E improve sperm parameters of cloned goats following freezing-thawing. Cryobiology 74:110–114. https://doi.org/10.1016/j.cryobiol.2016.11.007
Karuthadurai T, Das DN, Kumaresan A et al (2022) Sperm transcripts Associated with odorant binding and olfactory transduction pathways are altered in breeding bulls producing poor-quality semen. Front Vet Sci 9:1–15. https://doi.org/10.3389/fvets.2022.799386
Keller DW, Polakoski KL (1975) L-Arginine stimulation of human sperm motility in vitro. Biol Reprod 13:154–157. https://doi.org/10.1095/biolreprod13.2.154
Kim D, Paggi JM, Park C et al (2019) Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat Biotechnol 37:907–915. https://doi.org/10.1038/s41587-019-0201-4
Korrodi-Gregório L, Vieira SI, Esteves SLC et al (2013) TCTEX1D4, a novel protein phosphatase 1 interactor: connecting the phosphatase to the microtubule network. Biol Open 2:453–465. https://doi.org/10.1242/bio.20131065
Kovaka S, Zimin AV, Pertea GM et al (2019) Transcriptome assembly from long-read RNA-seq alignments with StringTie2. Genome Biol 20:278. https://doi.org/10.1186/s13059-019-1910-1
Lambard S (2004) Analysis and significance of mRNA in human ejaculated sperm from normozoospermic donors: relationship to sperm motility and capacitation. Mol Hum Reprod 10:535–541. https://doi.org/10.1093/molehr/gah064
Lasko J, Schlingmann K, Klocke A et al (2012) Calcium/calmodulin and cAMP/protein kinase-A pathways regulate sperm motility in the stallion. Anim Reprod Sci 132:169–177. https://doi.org/10.1016/j.anireprosci.2012.05.007
Li S, Ao L, Yan Y et al (2019) Differential motility parameters and identification of proteomic profiles of human sperm cryopreserved with cryostraw and cryovial. Clin Proteomics 16. https://doi.org/10.1186/s12014-019-9244-2
Liu XX, Cai L, Liu FJ (2018) An in silico analysis of human sperm genes associated with asthenozoospermia and its implication in male infertility. Med (United States) 97:1–7. https://doi.org/10.1097/MD.0000000000013338
Li Y, Li R-H, Ran M-X et al (2018) High throughput small RNA and transcriptome sequencing reveal capacitation-related microRNAs and mRNA in boar sperm. BMC Genomics 19:736. https://doi.org/10.1186/s12864-018-5132-9
Lybaert P, Vanbellinghen AM, Quertinmont E et al (2008) KATP channel subunits are expressed in the epididymal epithelium in several mammalian species. Biol Reprod 79:253–261. https://doi.org/10.1095/biolreprod.107.064659
Maciel VL, Caldas-Bussiere MC, Silveira V et al (2018) L-arginine alters the proteome of frozen-thawed bovine sperm during in vitro capacitation. Theriogenology 119:1–9. https://doi.org/10.1016/j.theriogenology.2018.06.018
Maidin MS, Aminudin NFAdanan, Tawang MT A (2014) In Vitro Supplements improves motility and progressive score of Spermatozoa in Jermasia Goats. APCBEE Procedia 8:329–333. https://doi.org/10.1016/j.apcbee.2014.03.049
NagDas SK, Winfrey VP, Olson GE (2002) Identification of ras and its downstream signaling elements and their potential role in hamster sperm motility. Biol Reprod 67:1058–1066. https://doi.org/10.1095/biolreprod67.4.1058
Nayernia K, Adham IM, Burkhardt-Göttges E et al (2002) Asthenozoospermia in Mice with Targeted Deletion of the Sperm Mitochondrion-Associated Cysteine-Rich Protein (Smcp) Gene. Mol Cell Biol 22:3046–3052. https://doi.org/10.1128/mcb.22.9.3046-3052.2002
O’Flaherty C, Rodriguez P, Srivastava S (2004) L-Arginine promotes capacitation and acrosome reaction in cryopreserved bovine spermatozoa. Biochim Biophys Acta - Gen Subj 1674:215–221. https://doi.org/10.1016/j.bbagen.2004.06.020
Oviedo N, Ortiz-Borrayo L, Hernández-Sánchez J et al (2018) Human CATSPER1 promoter is regulated by CREB1 and CREMτ transcriptional factors in Vitro. Arch Med Res 49:135–146. https://doi.org/10.1016/j.arcmed.2018.06.004
Parrish JJ, Susko-Parrish JL, First NL (1989) Capacitation of bovine sperm by heparin: inhibitory effect of glucose and role of intracellular pH. Biol Reprod 41:683–699. https://doi.org/10.1095/biolreprod41.4.683
Patra T, Gupta MK (2019) Cryopreservation of murine testicular leydig cells by modified solid surface vitrification with supplementation of antioxidants. Cryobiology 88:38–46. https://doi.org/10.1016/j.cryobiol.2019.04.002
Paudel B, Gervasi MG, Porambo J et al (2019) Sperm capacitation is associated with phosphorylation of the testis-specific radial spoke protein Rsph6a. Biol Reprod 100:440–454. https://doi.org/10.1093/biolre/ioy202
Pereira R, Sá R, Barros A, Sousa M (2017) Major regulatory mechanisms involved in sperm motility. Asian J Androl 19:5–14. https://doi.org/10.4103/1008-682X.167716
Prakash MA, Kumaresan A, Ebenezer Samuel King JP et al (2021) Comparative transcriptomic analysis of Spermatozoa from High- and low-fertile crossbred Bulls: implications for fertility prediction. Front Cell Dev Biol 9:1–14. https://doi.org/10.3389/fcell.2021.647717
Radany EW, Atherton RW (1981) Arginine induced stimulation of rabbit sperm motility. Arch Androl 7:351–355. https://doi.org/10.3109/01485018108999330
Rathi R, Colenbrander B, Bevers MM, Gadella BM (2001) Evaluation of in vitro capacitation of stallion spermatozoa. Biol Reprod 65:462–470. https://doi.org/10.1095/biolreprod65.2.462
Rodriguez PC, Valdez LB, Zaobornyj T et al (2011) Nitric oxide and Superoxide Anion Production during Heparin-Induced Capacitation in Cryopreserved bovine spermatozoa. Reprod Domest Anim 46:74–81. https://doi.org/10.1111/j.1439-0531.2010.01583.x
Roy SC, Atreja SK (2008) Tyrosine phosphorylation of a 38-kDa capacitation-associated buffalo (Bubalus bubalis) sperm protein is induced by L-arginine and regulated through a cAMP/PKA-independent pathway. Int J Androl 31:12–24. https://doi.org/10.1111/j.1365-2605.2007.00745.x
Sahoo B, Guttula PK, Gupta MK (2021) Comparison of spermatozoal RNA extraction methods in goats. Anal Biochem 614:114059. https://doi.org/10.1016/j.ab.2020.114059
Schlingmann K, Michaut MA, Mcelwee JL et al (2007) Calmodulin and CaMKII in the sperm principal piece: evidence for a motility-related calcium/calmodulin pathway. J Androl 28:706–716. https://doi.org/10.2164/jandrol.106.001669
Shannon P, Markiel A, Ozier O et al (2003) Cytoscape: A Software Environment for Integrated Models of Biomolecular Interaction Networks. 2498–2504. https://doi.org/10.1101/gr.1239303.metabolite
Somlev B, Subev M (1998) Effect of kininase II inhibitors on bradykinin-stimulated bovine sperm motility. Theriogenology 50:651–657. https://doi.org/10.1016/S0093-691X(98)00169-1
Song Y, Milon B, Ott S et al (2018) A comparative analysis of library prep approaches for sequencing low input translatome samples. BMC Genomics 19:1–16. https://doi.org/10.1186/s12864-018-5066-2
Spehr M, Schwane K, Riffell JA et al (2004) Particulate adenylate cyclase plays a key role in human sperm olfactory receptor-mediated chemotaxis. J Biol Chem 279:40194–40203. https://doi.org/10.1074/jbc.M403913200
Srivastava S, Desai P, Coutinho E, Govil G (2006) Mechanism of action of L-arginine on the vitality of spermatozoa is primarily through increased biosynthesis of nitric oxide. Biol Reprod 74:954–958. https://doi.org/10.1095/biolreprod.105.046896
Staicu FD, Lopez-Úbeda R, Romero-Aguirregomezcorta J et al (2019) Regulation of boar sperm functionality by the nitric oxide synthase/nitric oxide system. J Assist Reprod Genet 36:1721–1736. https://doi.org/10.1007/s10815-019-01526-6
Stegemann JP (2007) Characterization of a Novel Tektin Member, TEKT5, in Mouse Sperm. Tissue Eng 23:1–7. https://doi.org/10.2164/jandrol.109.009456
Szklarczyk D, Gable AL, Lyon D et al (2019) STRING v11: protein – protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. 47:607–613. https://doi.org/10.1093/nar/gky1131
Tourmente M, Sanchez-Rodriguez A, Roldan ERS (2022) Effect of motility factors D-Penicillamine, hypotaurine and epinephrine on the performance of Spermatozoa from five Hamster species. Biology (Basel) 11:526. https://doi.org/10.3390/biology11040526
Transl J, Zhao Y, Li MC et al (2021) TPM, FPKM, or normalized Counts ? A comparative study of quantification measures for the analysis of RNA – seq data from the NCI patient – derived models repository. J Transl Med 1–15. https://doi.org/10.1186/s12967-021-02936-w
Urizar-Arenaza I, Osinalde N, Akimov V et al (2019) Phosphoproteomic and functional analyses reveal sperm-specific protein changes downstream of kappa opioid receptor in human spermatozoa. Mol Cell Proteomics 18:S118–S131. https://doi.org/10.1074/mcp.RA118.001133
Valdivia A, Cortés L, Beitia M et al (2020) Role of Angiotensin-(1–7) via MAS receptor in human sperm motility and acrosome reaction. Reproduction 159:241–249. https://doi.org/10.1530/REP-19-0274
Vickram AS, Srikumar PS, Srinivasan S et al (2021) Seminal exosomes – an important biological marker for various disorders and syndrome in human reproduction. Saudi J Biol Sci 28:3607–3615. https://doi.org/10.1016/j.sjbs.2021.03.038
Wang T, Yin Q, Ma X et al (2018) Ccdc87 is critical for sperm function and male fertility. Biol Reprod 99:817–827. https://doi.org/10.1093/biolre/ioy106
Wang X, Yang C, Guo F et al (2019) Integrated analysis of mRNAs and long noncoding RNAs in the semen from Holstein bulls with high and low sperm motility. Sci Rep 9:1–10. https://doi.org/10.1038/s41598-018-38462-x
Zhang X, Zhang P, Song D et al (2019) Expression profiles and characteristics of human lncRNA in normal and asthenozoospermia sperm†. Biol Reprod 100:982–993. https://doi.org/10.1093/biolre/ioy253
Zhang Y, Dai D, Chang Y et al (2017) Cryopreservation of boar sperm induces differential microRNAs expression. Cryobiology 76:24–33. https://doi.org/10.1016/j.cryobiol.2017.04.013
Zhu F, Yan P, Zhang J et al (2019) Deficiency of TPPP2, a factor linked to oligoasthenozoospermia, causes subfertility in male mice. J Cell Mol Med 23:2583–2594. https://doi.org/10.1111/jcmm.14149
Acknowledgements
This research project was performed at the Department of Biotechnology and Medical Engineering, National Institute of Technology, Rourkela.
Funding
This work was supported by a grant (#BT/PR40124/BTIS/137/14/2021) from the Department of Biotechnology, Government of India.
Author information
Authors and Affiliations
Contributions
BS: Designed and performed the experiments, analyzed the data, and wrote the manuscript. MKG: Provided resources, supervised the work, and finalized and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Ethics approval
This article does not contain any studies with human participants performed by authors.
Consent
Not applicable.
Compliance with ethical standards
All the experiments on abattoir-derived testes samples were performed according to National guidelines of the Committee for the Purpose of Control and Supervision on Experiments on Animals (CPCSEA) of the Ministry of Fisheries, Animal Husbandry and Dairying, Government of India.
Research involving human participants and/or animals
This article does not contain any studies with human participants performed by any authors.
Informed consent
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.




Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sahoo, B., Gupta, M.K. Effect of arginine-induced motility and capacitation on RNA population in goat spermatozoa. Vet Res Commun 47, 1427–1444 (2023). https://doi.org/10.1007/s11259-023-10092-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10092-3