Abstract
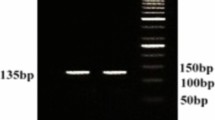
The abuse of acaricides for control of Rhipicephalus sanguineus sensu lato ticks led to a strong selection pressure for acaricide resistance. Data on acaricide resistance in R. sanguineus s.l. populations from India is meager though veterinarians frequently report treatment failures. The present study was aimed to characterize the level of resistance in R. sanguineus s.l. against most commonly used drugs, cypermethrin and ivermectin in Andhra Pradesh, south India. Fourteen adult female tick populations were collected from naturally infested dogs at veterinary clinics, residence and stray dog in nine state municipal corporations/municipalities. Information on the history of dog treatment with acaricides was obtained by interviewing dog owners. The larval packet test (LPT) and larval immersion test (LIT) were used on the larvae of ticks to test for resistance to cypermethrin and ivermectin, respectively. Mortality data of each tick population was analyzed by probit analysis. Corresponding to the most susceptible population, thirteen (92.6%) and six (42.9%) tick populations were regarded as resistant to cypermethrin and ivermectin, respectively. The phenotypic level of cypermethrin (resistance ratios at LC50% varied from 1.55 to 13.29) and ivermectin (resistance ratios at LC50% ranged from 1.16 to 4.79) resistance varied distinctly between the populations. Additionally, all the populations were tested using PCR to demonstrate the frequency of the point mutation in sodium channel gene that corresponds with resistance of R. sanguineus s.l. to cypermethrin. A nucleotide substitution (T2134C) on domain III segment VI of the sodium channel gene was noticed in phenotypically resistant tick populations on DNA sequencing. Ivermectin resistance in the brown dog ticks is reported for the first time from India.



Similar content being viewed by others
Code availability
Not applicable.
References
Alegria-Lopez MA, Rodriguez-Vivas RI, Torres-Acosta JFJ, Ojeda-Chi MM, Rosado-Aguilar JA (2015) Use of ivermectin as endoparasiticide in tropical cattle herds generates resistance in gastrointestinal nematodes and the tick Rhipicephalus microplus (Acari: Ixodidae). J Med Entomol 52(2):214–221
Becker S, Webster A, Doyle RL, Martins JR, Reck J, Klafke GM (2019) Resistance to deltamethrin, fipronil and ivermectin in the brown dog tick, Rhipicephalus sanguineus sensu stricto, Latreille (Acari: Ixodidae). Ticks Tick Borne Dis 10(5):1046–1050
Brengues C, Hawkes NJ, Chandre F, McCarroll L, Duchon S, Guillet P, Hemingway J (2003) Pyrethroid and DDT cross-resistance in Aedes aegypti is correlated with novel mutations in the voltage-gated sodium channel gene. Med Vet Entomol 17(1):87–94
Cafarchia C, Porretta D, Mastrantonio V, Epis S, Sassera D, Iatta R, Otranto D (2015) Potential role of ATP-binding cassette transporters against acaricides in the brown dog tick Rhipicephalus sanguineus sensu lato. Med Vet Entomol 29(1):88–93
Campbell WC (2016) Lessons from the history of ivermectin and other antiparasitic agents. Annu Rev Anim Biosci 4:1–14
Cen-Aguilar JF, Rodriguez-Vivas RI, Dominguez-Alpizar JL, Wagner GG (1998) Studies on the effect of infection by Babesia species on oviposition of Boophilus microplus engorged females naturally infected in the Mexican tropics. Vet Parasitol 78(4):253–257
Dantas-Torres F (2008) The brown dog tick, Rhipicephalus sanguineus (Latreille, 1806) (Acari: Ixodidae): from taxonomy to control. Vet Parasitol 152(3–4):173–185
Dong K, Du Y, Rinkevich F, Nomura Y, Xu P, Wang L, Zhorov BS (2014) Molecular biology of insect sodium channels and pyrethroid resistance. Insect Biochem Mol Biol 50:1–17
Dryden MW (2009) Flea and tick control in the 21st century: challenges and opportunities. Vet Dermatol 20(5–6):435–440
Eiden AL, Kaufman PE, Oi FM, Allan SA, Miller RJ (2015) Detection of permethrin resistance and fipronil tolerance in Rhipicephalus sanguineus (Acari: Ixodidae) in the United States. J Med Entomol 52(3):429–436
Eiden AL, Kaufman PE, Oi FM, Dark MJ, Bloomquist JR, Miller RJ (2017) Determination of metabolic resistance mechanisms in pyrethroid-resistant and fipronil-tolerant brown dog ticks. Med Vet Entomol 31(3):243–251
Estrada-Pena A (2005) Etude de la resistance de la tique brune du chien Rhipicephalus Sanguineus Aux Acaricides. Rev Med Vet 156(2):67–69
Falconer DS, Mackay TFC (2009) Introduction to quantitative genetics, 4th edn. Pearson Education Ltd, United Kingdom
FAO: Food and Agriculture Organization of the United Nations (1971) Recommended methods for the detection and measurement of resistance of agricultural pests to pesticides–tentative method for larvae of cattle ticks, Boophilus microplus species FAO method No 7. FAO Plant Prot Bull 19:15–18
FAO: Food and Agriculture Organization (2004) Resistance management and integrated parasite control in ruminants–guidelines, module 1–ticks: acaricide resistance: diagnosis, management and prevention, pp 25–77
Farnham AW, Murray AWA, Sawicki RM, Denholm I, White JC (1987) Characterization of the structure-activity relationship of kdr and two variants of super-kdr to pyrethroids in the housefly (Musca domestica L). Pest Manag Sci 19:209–220
Guerrero FD, Davey RB, Miller RJ (2001) Use of an allele-specific polymerase chain reaction assay to genotype pyrethroid resistant strains of Boophilus microplus (Acari: Ixodidae). J Med Entomol 38(1):44–50
Guerrero FD, Jamroz RC, Kammlah D, Kunz SE (1997) Toxicological and molecular characterization of pyrethroid-resistant horn flies, Haematobia irritans: identification of kdr and super-kdr point mutations. Insect Biochem Mol Biol 27(8–9):745–755
Guerrero FD, Li AY, Hernandez R (2002) Molecular diagnosis of pyrethroid resistance in Mexican strains of Boophilus microplus (Acari: Ixodidae). J Med Entomol 39(5):770–776
Hawkins NJ, Bass C, Dixon A, Neve P (2019) The evolutionary origins of pesticide resistance. Biol Rev 94(1):135–155
Jamroz RC, Guerrero FD, Pruett JH, Oehler DD, Miller RJ (2000) Molecular and biochemical survey of acaricide resistance mechanisms in larvae from Mexican strains of the southern cattle tick, Boophilus microplus. J Insect Physiol 46(5):685–695
Klafke GM, Miller RJ, Tidwell J, Barreto R, Guerrero FD, Kaufman PE, Perez de Leon AA (2017) Mutation in the sodium channel gene corresponds with phenotypic resistance of Rhipicephalus sanguineus sensu lato (Acari: Ixodidae) to pyrethroids. J Med Entomol 54(6):1639–1642
Kumar R, Nagar G, Sharma AK, Kumar S, Ray DD, Chaudhuri P, Ghosh S (2013) Survey of pyrethroids resistance in Indian isolates of Rhipicephalus (Boophilus) microplus: identification of C190A mutation in the domain II of the para-sodium channel gene. Acta Trop 125(2):237–245
Kumar R, Sharma AK, Ghosh S (2020) Menace of acaricide resistance in cattle tick, Rhipicephalus microplus in India: Status and possible mitigation strategies. Vet Parasitol 278:108993
Kunz SE, Kemp DH (1994) Insecticides and acaricides: resistance and environmental impact. Rev Sci Tech Off Int Epiz 13(4):1249–1286
Latreille PA (1806) Genera crustaceorum et insectorum secundum ordinem naturalem in familia disposita, iconibus exemplisque plurimis explicate. Paris et Argentorati 1:302
Mathivathani C, Basith SA, Latha BR, Raj GD (2011) In vitro evaluation of synthetic pyrethroid resistance in Rhipicephalus sanguineus ticks of Chennai. J Vet Parasitol 25(1):56–58
Miller RJ, George JE, Guerrero F, Carpenter L, Welch JB (2001) Characterization of acaricide resistance in Rhipicephalus sanguineus (Latreille) (Acari: Ixodidae) collected from the Corozal army Veterinary quarantine center, Panama. J Med Entomol 38(2):298–302
Nandi A, Sagar SV, Chigure G, Fular A, Sharma AK, Nagar G, Ghosh S (2018) Determination and validation of discriminating concentration of ivermectin against Rhipicephalus microplus. Vet Parasitol 250:30–34
Needham GR, Teel PD (1991) Off-host physiological ecology of ixodid ticks. Annu Rev Entomol 36(1):659–681
Nolan J, Schnitzerling HJ (1986) Drug resistance in arthropod parasites. In: Chemotherapy of parasitic diseases. WC, Campbell, New York, pp 603–620
Ochomo E, Bayoh MN, Brogdon WG, Gimnig JE, Ouma C, Vulule JM, Walker ED (2013) Pyrethroid resistance in Anopheles gambiae ss and Anopheles arabiensis in western Kenya: phenotypic, metabolic and target site characterizations of three populations. Med Vet Entomol 27(2):156–164
Pohl PC, Klafke GM, Carvalho DD, Martins JR, Daffre S, da Silva Vaz Jr I, Masuda A, (2011) ABC transporter efflux pumps: a defense mechanism against ivermectin in Rhipicephalus (Boophilus) microplus. Int J Parasitol 41(13–14):1323–1333
Rodriguez I, Fraga J, Noda AA, Mayet M, Duarte Y, Echevarria E, Fernandez C (2014) An alternative and rapid method for the extraction of nucleic acids from ixodid ticks by potassium acetate procedure. Braz Arch Biol Technol 57(4):542–547
Rodriguez-Vivas RI, Ojeda-Chi MM, Trinidad-Martinez I, Bolio-Gonzalez ME (2017a) First report of amitraz and cypermethrin resistance in Rhipicephalus sanguineus sensu lato infesting dogs in Mexico. Med Vet Entomol 31(1):72–77
Rodriguez-Vivas RI, Ojeda-Chi MM, Trinidad-Martine I, De Leon AP (2017b) First documentation of ivermectin resistance in Rhipicephalus sanguineus sensu lato (Acari: Ixodidae. Vet Parasitol 233:9–13
Robertson JL, Preisler HK (1992) Pesticide bioassays with arthropods. CRC Press, pp127
Scott JG, Georghiou GP (1986) Mechanisms responsible for high levels of permethrin resistance in the house fly. Pestic Sci 17(3):195–206
Shyma KP, Singh V, Gupta JP (2019) In vitro evaluation of effectiveness of synthetic pyrethroids against brown dog tick. Indian J Anim Res 53(10):1400–1402
Singh NK, Singh H, Prerna M, Rath SS (2015) First report of ivermectin resistance in field populations of Rhipicephalus (Boophilus) microplus (Acari: Ixodidae) in Punjab districts of India. Vet Parasitol 214(1–2):192–194
Sowjanya M (2019) Epidemiological surveillance of ectoparasitic infections in dogs in krishna-godavari zone of Andhra Pradesh. Thesis submitted to Sri Venkateswara Veterinary University Tirupati, Andhra Pradesh, India
Tucker NS, Kaufman PE, Weeks EN, Rowland J, Tidwell J, Miller RJ (2017) Characterization of a sodium channel mutation in permethrin-resistant Rhipicephalus sanguineus (Acari: Ixodidae). J Med Entomol 54(6):1633–1638
Tucker NS, Kaufman PE, Weeks EN (2019) Identification of permethrin and etofenprox cross-tolerance in Rhipicephalus sanguineus sensu lato (Acari: Ixodidae). Pest Manag Sci 75(10):2794–2801
Wharton RH (1976) Tick-borne livestock diseases and their vectors 5 Acaricide resistance and alternative methods of tick control. World Anim Rev 20:8–15
Whitehead GB (1965) Resistance in Acarina: Ticks. Adv Acarol 2:53–70
Acknowledgements
The authors are thankful to Sri Venkateswara Veterinary University, Tirupati, India, for providing the facilities to do the research work.
Author information
Authors and Affiliations
Contributions
Conceptualization: Sreedevi Chennuru, Prathyusha Sunkara, Formal analysis: Sudhakar Krovvidi, Investigation: Prathyusha Sunkara, Krovvidi Sudhakar Methodology: Sreedevi Chennuru, Sudhkar Krovvidi, Jyothisree Chitichoti, Writing – original draft: Sreedevi Chennuru, Prathyusha Sunkara, Writing – review and editing: Sreedevi Chennuru, Sudhkar Krovvidi, Jyothisree Chitichoti.
Corresponding author
Ethics declarations
Ethical statement
No experimentation was conducted on animals. Ticks were collected from dogs attending to the veterinary dispensaries and hence doesn’t necessitate for ethical approval. Study animals were subjected to minimum stress while collecting ticks.
Consent to participate
All authors contributed to the study conception and design. All authors read and approved the final manuscript.
Consent for publication
All authors gave their consent for research publication.
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sunkara, P., Chennuru, S., Krovvidi, S. et al. Cypermethrin and ivermectin resistance in field populations of Rhipicephalus sanguineus sensu lato (Latrielle, 1806) collected from dogs in south India. Vet Res Commun 46, 67–78 (2022). https://doi.org/10.1007/s11259-021-09830-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-021-09830-2