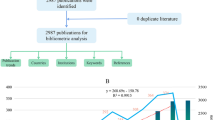
Abstract
Introduction
Acute kidney injury (AKI) is a global disease with high morbidity and mortality. At present, the treatment of AKI still lacks targeted measures. Ferroptosis, a form of regulated cell death, plays an essential role in the initiation and progression of AKI. Current evidence proves that targeting ferroptosis is supposed to be a novel potential strategy to cure AKI. In this study, we aim to use bibliometric analysis to identify research trends and hotspots in the field of “ferroptosis in AKI”.
Methods
We chose the Science Citation Index Expanded (SCI-EXPANDED) index of the Web of Science Core Collection (WoSCC) as the source database for data retrieval. Data were retrieved from the WoSCC on May 24, 2022. Full records and cited references of all the documents in WoSCC were collected. The R software and the Online Analysis Platform of Literature Metrology were used for data analysis and visual analysis.
Results
There were 120 documents on “ferroptosis in AKI” in the WOSCC from 2014 to 2022 (May 24, 2022). There was a clear upward trend each year in the number of documents published. According to WoS report, China, the United States, and Germany were the top three countries involved in this research area, the majority of publications were included in the subject area “Cell Biology”. Technical University of Dresden contributed the most publications, followed by Central South University and University of Pittsburgh. The Journal of Cell Death and Disease had the highest H-index and contributed the most publications. Linkermann A authored 16 articles and had the highest H-index. Multifactorial analysis of the keywords show that the research field is divided into two clusters. The most contributing publications and the most cited publications were also determined by factorial analysis.
Conclusion
This bibliometric analysis provides a comprehensive analysis of research trends and hot spots on the topic of “ferroptosis in AKI”. The study of ferroptosis-related AKI research remains in its early stages. There will be a dramatically increasing number of publications on this field. Further research should focus on exploring the mechanisms of crosstalk between ferroptosis and other programmed cell deaths, and improves clinical applications and therapeutic effects against AKI.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Rui Y, Li S, Luan F, Li D, Liu R, Zeng N (2022) Several alkaloids in chinese herbal medicine exert protection in acute kidney injury: focus on mechanism and target analysis. Oxid Med Cell Longev 2022:2427802
Feng Q, Yu X, Qiao Y, Pan S, Wang R, Zheng B, Wang H, Ren KD, Liu H, Yang Y (2022) Ferroptosis and acute kidney injury (aki): molecular mechanisms and therapeutic potentials. Front Pharmacol 13:858676
Li W, Xiang Z, Xing Y, Li S, Shi S (2022) Mitochondria bridge HIF signaling and ferroptosis blockage in acute kidney injury. Cell Death Dis 13(4):308
Ronco C, Bellomo R, Kellum JA (2019) Acute kidney injury. Lancet 394(10212):1949–1964
Hu Z, Zhang H, Yang SK, Wu X, He D, Cao K, Zhang W (2019) Emerging role of ferroptosis in acute kidney injury. Oxid Med Cell Longev 2019:8010614
Zhang J, Wang B, Yuan S, He Q, Jin J (2022) The role of ferroptosis in acute kidney injury. Front Mol Biosci 9:951275
Yao X, Li W (2021) Fang, Xiao C, Wu X, Li M, Luo Z: Emerging Roles of Energy Metabolism in Ferroptosis Regulation of Tumor Cells. Adv Sci (Weinh) 8(22):e2100997
Linkermann A (2016) Nonapoptotic cell death in acute kidney injury and transplantation. Kidney Int 89(1):46–57
Li J, Cao F, Yin HL, Huang ZJ, Lin ZT, Mao N, Sun B, Wang G (2020) Ferroptosis: past, present and future. Cell Death Dis 11(2):88
Wang Y, Bi R, Quan F, Cao Q, Lin Y, Yue C, Cui X, Yang H, Gao X, Zhang D (2020) Ferroptosis involves in renal tubular cell death in diabetic nephropathy. Eur J Pharmacol 888:173574
Guerrero-Hue M, Garcia-Caballero C, Palomino-Antolin A, Rubio-Navarro A, Vazquez-Carballo C, Herencia C, Martin-Sanchez D, Farre-Alins V, Egea J, Cannata P et al (2019) Curcumin reduces renal damage associated with rhabdomyolysis by decreasing ferroptosis-mediated cell death. FASEB J 33(8):8961–8975
Guler AT, Waaijer CJ, Palmblad M (2016) Scientific workflows for bibliometrics. Scientometrics 107:385–398
Wu H, Wang Y, Tong L, Yan H, Sun Z (2021) Global research trends of ferroptosis: a rapidly evolving field with enormous potential. Front Cell Dev Biol 9:646311
Dong X, Tan Y, Zhuang D, Hu T, Zhao M (2022) Global Characteristics and trends in research on ferroptosis: a data-driven bibliometric study. Oxid Med Cell Longev 2022:8661864
Banchi P, Rota A, Bertero A, Domain G, Ali Hassan H, Lannoo J, Van Soom A (2022) Trends in small animal reproduction: a bibliometric analysis of the literature. Animals 12(3):336
Xiao J, Yang Q, Zhang Y, Xu H, Ye Y, Li L, Yang Y, Jin S (2021) Maresin conjugates in tissue regeneration-1 suppresses ferroptosis in septic acute kidney injury. Cell Biosci 11(1):221
Li D, Liu B, Fan Y, Liu M, Han B, Meng Y, Xu X, Song Z, Liu X, Hao Q et al (2021) Nuciferine protects against folic acid-induced acute kidney injury by inhibiting ferroptosis. Br J Pharmacol 178(5):1182–1199
Meng X, Huang W, Mo W, Shu T, Yang H, Ning H (2021) ADAMTS-13-regulated nuclear factor E2-related factor 2 signaling inhibits ferroptosis to ameliorate cisplatin-induced acute kidney injuy. Bioengineered 12(2):11610–11621
Ma D, Li C, Jiang P, Jiang Y, Wang J, Zhang D (2021) Inhibition of ferroptosis attenuates acute kidney injury in rats with severe acute pancreatitis. Dig Dis Sci 66(2):483–492
Ding C, Ding X, Zheng J, Wang B, Li Y, Xiang H, Dou M, Qiao Y, Tian P, Xue W (2020) miR-182-5p and miR-378a-3p regulate ferroptosis in I/R-induced renal injury. Cell Death Dis 11(10):929
Dizaji R, Sharafi A, Pourahmad J, Abdollahifar MA, Vatanpour H, Hosseini MJ (2019) Induction of two independent immunological cell death signaling following hemoglobinuria -induced acute kidney injury: In vivo study. Toxicon 163:23–31
Zhao C, Yu D, He Z, Bao L, Feng L, Chen L, Liu Z, Hu X, Zhang N, Wang T et al (2021) Endoplasmic reticulum stress-mediated autophagy activation is involved in cadmium-induced ferroptosis of renal tubular epithelial cells. Free Radic Biol Med 175:236–248
Fan Z, Liu T, Huang H, Lin J, Zeng Z (2021) A ferroptosis-related gene signature for graft loss prediction following renal allograft. Bioengineered 12(1):4217–4232
Stoppe C, Averdunk L, Goetzenich A, Soppert J, Marlier A, Kraemer S, Vieten J, Coburn M, Kowark A, Kim BS et al (2018) The protective role of macrophage migration inhibitory factor in acute kidney injury after cardiac surgery. Sci Transl Med 10:441
Yoshioka H, Tominaga S, Nishikawa M, Shinohara Y, Nakao M, Yoshikawa M, Maeda T, Miura N (2021) Different renal chronotoxicity of bromobenzene and its intermediate metabolites in mice. Biol Pharm Bull 44(1):150–153
Xie X, Zhang Y, Su X, Wang J, Yao X, Lv D, Zhou Q, Mao J, Chen J, Han F et al (2022) Targeting iron metabolism using gallium nanoparticles to suppress ferroptosis and effectively mitigate acute kidney injury. Nano Res 15(7):6315–6327
von Massenhausen A, Zamora Gonzalez N, Maremonti F, Belavgeni A, Tonnus W, Meyer C, Beer K, Hannani MT, Lau A, Peitzsch M et al (2022) Dexamethasone sensitizes to ferroptosis by glucocorticoid receptor-induced dipeptidase-1 expression and glutathione depletion. Sci Adv 8(5):8920
Linkermann A, Chen G, Dong G, Kunzendorf U, Krautwald S, Dong Z (2014) Regulated cell death in AKI. J Am Soc Nephrol 25(12):2689–2701
Linkermann A, Skouta R, Himmerkus N, Mulay SR, Dewitz C, De Zen F, Prokai A, Zuchtriegel G, Krombach F, Welz PS et al (2014) Synchronized renal tubular cell death involves ferroptosis. Proc Natl Acad Sci U S A 111(47):16836–16841
Allam R, Kumar SV, Darisipudi MN, Anders HJ (2014) Extracellular histones in tissue injury and inflammation. J Mol Med 92(5):465–472
Song J, Sheng J, Lei J, Gan W, Yang Y (2022) Mitochondrial targeted antioxidant skq1 ameliorates acute kidney injury by inhibiting ferroptosis. Oxid Med Cell Longev 2022:2223957
Wang H, Liu C, Zhao Y, Gao G (2020) Mitochondria regulation in ferroptosis. Eur J Cell Biol 99(1):151058
Gao M, Yi J, Zhu J, Minikes AM, Monian P, Thompson CB, Jiang X (2019) Role of mitochondria in ferroptosis. Mol Cell 73(2):354–363
Hosohata K, Harnsirikarn T, Chokesuwattanaskul S (2022) Ferroptosis: a potential therapeutic target in acute kidney injury. Int J Mol Sci 23(12):6583
Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascon S, Hatzios SK, Kagan VE et al (2017) Ferroptosis: A regulated cell death nexus linking metabolism, redox biology, and disease. Cell 171(2):273–285
Martin-Sanchez D, Fontecha-Barriuso M, Martinez-Moreno JM, Ramos AM, Sanchez-Nino MD, Guerrero-Hue M, Moreno JA, Ortiz A, Sanz AB (2020) Ferroptosis and kidney disease. Nefrologia 40(4):384–394
Gao M, Monian P, Quadri N, Ramasamy R, Jiang X (2015) Glutaminolysis and transferrin regulate ferroptosis. Mol Cell 59(2):298–308
Sun Y, Zheng Y, Wang C, Liu Y (2018) Glutathione depletion induces ferroptosis, autophagy, and premature cell senescence in retinal pigment epithelial cells. Cell Death Dis 9(7):753
Kagan VE, Mao G, Qu F, Angeli JP, Doll S, Croix CS, Dar HH, Liu B, Tyurin VA, Ritov VB et al (2017) Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat Chem Biol 13(1):81–90
Lin Y, Xu W, Hou Y, Wang S, Zhang H, Ran M, Huang Y, Wang Y, Yang G (2022) The multifaceted role of ferroptosis in kidney diseases. Chem Biol Interact 365:110107
Thapa K, Singh TG, Kaur A (2022) Targeting ferroptosis in ischemia/reperfusion renal injury. Naunyn Schmiedebergs Arch Pharmacol 395(11):1331–1341
Hou L, Li X, Su C, Chen K, Qu M (2022) Current status and prospects of research on ischemia-reperfusion injury and ferroptosis. Front Oncol 12:920707
Pefanis A, Ierino FL, Murphy JM, Cowan PJ (2019) Regulated necrosis in kidney ischemia-reperfusion injury. Kidney Int 96(2):291–301
Priante G, Gianesello L, Ceol M, Del Prete D, Anglani F (2019) Cell Death in the Kidney. Int J Mol Sci 20(14):3598
Anders HJ (2018) Necroptosis in acute kidney injury. Nephron 139(4):342–348
Zhao Z, Wu J, Xu H, Zhou C, Han B, Zhu H, Hu Z, Ma Z, Ming Z, Yao Y et al (2020) XJB-5-131 inhibited ferroptosis in tubular epithelial cells after ischemia-reperfusion injury. Cell Death Dis 11(8):629
Sarhan M, von Massenhausen A, Hugo C, Oberbauer R, Linkermann A (2018) Immunological consequences of kidney cell death. Cell Death Dis 9(2):114
Xie Y, Hou W, Song X, Yu Y, Huang J, Sun X, Kang R, Tang D (2016) Ferroptosis: process and function. Cell Death Differ 23(3):369–379
Martin-Sanchez D, Ruiz-Andres O, Poveda J, Carrasco S, Cannata-Ortiz P, Sanchez-Nino MD, Ruiz Ortega M, Egido J, Linkermann A, Ortiz A et al (2017) Ferroptosis, but not necroptosis, is important in nephrotoxic folic acid-induced AKI. J Am Soc Nephrol 28(1):218–229
Wenzel SE, Tyurina YY, Zhao J, St Croix CM, Dar HH, Mao G, Tyurin VA, Anthonymuthu TS, Kapralov AA, Amoscato AA et al (2017) PEBP1 wardens ferroptosis by enabling lipoxygenase generation of lipid death signals. Cell 171(3):628–641
Moreno-Gonzalez G, Vandenabeele P, Krysko DV (2016) Necroptosis: A novel cell death modality and its potential relevance for critical care medicine. Am J Respir Crit Care Med 194(4):415–428
Deng F, Zheng X, Sharma I, Dai Y, Wang Y, Kanwar YS (2021) Regulated cell death in cisplatin-induced AKI: relevance of myo-inositol metabolism. Am J Physiol Renal Physiol 320(4):F578–F595
Tonnus W, Linkermann A (2017) The in vivo evidence for regulated necrosis. Immunol Rev 277(1):128–149
Ying Y, Padanilam BJ (2016) Regulation of necrotic cell death: p53, PARP1 and cyclophilin D-overlapping pathways of regulated necrosis? Cell Mol Life Sci 73(11–12):2309–2324
Martin-Sanchez D, Gallegos-Villalobos A, Fontecha-Barriuso M, Carrasco S, Sanchez-Nino MD, Lopez-Hernandez FJ, Ruiz-Ortega M, Egido J, Ortiz A, Sanz AB (2017) Deferasirox-induced iron depletion promotes BclxL downregulation and death of proximal tubular cells. Sci Rep 7:41510
Su L, Jiang X, Yang C, Zhang J, Chen B, Li Y, Yao S, Xie Q, Gomez H, Murugan R et al (2019) Pannexin 1 mediates ferroptosis that contributes to renal ischemia/reperfusion injury. J Biol Chem 294(50):19395–19404
Tonnus W, Meyer C, Steinebach C, Belavgeni A, von Massenhausen A, Gonzalez NZ, Maremonti F, Gembardt F, Himmerkus N, Latk M et al (2021) Dysfunction of the key ferroptosis-surveilling systems hypersensitizes mice to tubular necrosis during acute kidney injury. Nat Commun 12(1):4402
Fan X, Zhang X, Liu LC, Zhang S, Pelger CB, Lughmani HY, Haller ST, Gunning WT 3rd, Cooper CJ, Gong R et al (2022) Hemopexin accumulates in kidneys and worsens acute kidney injury by causing hemoglobin deposition and exacerbation of iron toxicity in proximal tubules. Kidney Int 102(6):1320–1330
Zhou L, Xue X, Hou Q, Dai C (2022) Targeting ferroptosis attenuates interstitial inflammation and kidney fibrosis. Kidney Dis (Basel) 8(1):57–71
Yu P, Zhang J, Ding Y, Chen D, Sun H, Yuan F, Li S, Li X, Yang P, Fu L et al (2022) Dexmedetomidine post-conditioning alleviates myocardial ischemia-reperfusion injury in rats by ferroptosis inhibition via SLC7A11/GPX4 axis activation. Hum Cell 35(3):836–848
Wang Y, Quan F, Cao Q, Lin Y, Yue C, Bi R, Cui X, Yang H, Yang Y, Birnbaumer L et al (2021) Quercetin alleviates acute kidney injury by inhibiting ferroptosis. J Adv Res 28:231–243
Zhang J, Bi J, Ren Y, Du Z, Li T, Wang T, Zhang L, Wang M, Wei S, Lv Y et al (2021) Involvement of GPX4 in irisin’s protection against ischemia reperfusion-induced acute kidney injury. J Cell Physiol 236(2):931–945
Jiang GP, Liao YJ, Huang LL, Zeng XJ, Liao XH (2021) Effects and molecular mechanism of pachymic acid on ferroptosis in renal ischemia reperfusion injury. Mol Med Rep. https://doi.org/10.3892/mmr.2020.11704
Yang J, Sun X, Huang N, Li P, He J, Jiang L, Zhang X, Han S, Xin H (2022) Entacapone alleviates acute kidney injury by inhibiting ferroptosis. FASEB J 36(7):e22399
Sui M, Xu D, Zhao W, Lu H, Chen R, Duan Y, Li Y, Zhu Y, Zhang L, Zeng L (2021) CIRBP promotes ferroptosis by interacting with ELAVL1 and activating ferritinophagy during renal ischaemia-reperfusion injury. J Cell Mol Med 25(13):6203–6216
Huang YB, Jiang L, Liu XQ, Wang X, Gao L, Zeng HX, Zhu W, Hu XR, Wu YG (2022) Melatonin alleviates acute kidney injury by inhibiting nrf2/slc7a11 axis-mediated ferroptosis. Oxid Med Cell Longev 2022:4776243
Acknowledgements
This work was not supported by National Natural Science Foundation of China (82200780, 81871581).
Funding
National Natural Science Foundation of China (82200780, 81871581).
Author information
Authors and Affiliations
Contributions
CL and WHZ: contributed equally to this work. CL and WHZ: conceived the study, participated in the design, collected the data, performed statistical analyses, and drafted the manuscript. XML and QM: performed statistical analyses and helped to draft the manuscript. RF and ZM: critically revised the manuscript for important intellectual content. YBZ and LZ: contributed to collected data; QH and XFS: contributed to analyze data; FHZ: performed statistical analyses and helped to critically revise the manuscript for important intellectual content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
The ethics approval was not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, C., Zhou, W., Mao, Z. et al. Bibliometric analysis of ferroptosis in acute kidney injury from 2014 to 2022. Int Urol Nephrol 55, 1509–1521 (2023). https://doi.org/10.1007/s11255-022-03456-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-022-03456-2