Abstract
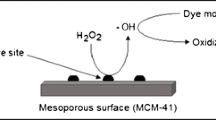
Vanadium-containing mesoporous molecular sieves have been prepared by hydrothermal treatment at 373 K. These materials showed spherical morphology with a narrow particle size distribution between 2 and 4 μm. The techniques used for their physicochemical characterization were: XRD, AAS, N2 physisorption, SEM, TEM and DR–UV–Vis spectroscopy. All the materials presented high specific surface area (>900 m2/g), characteristic of MCM-41 materials. A well-defined mesoporous structure was observed by TEM measures although there was no one-dimensional ordering of pores characteristic of such materials. Additionally, secondary mesoporosity domains were determined in the BJH size distribution. The sample synthesized with the highest content of V presented marked differences in their structural characteristics, which were attributed to the blockage of channels by the presence of nano-clusters and/or VxOy nano-oxides. From the DR–UV–Vis analysis, a successful incorporation of V ions to silica structure in tetrahedral coordination with oxygen of the network could be inferred. The catalytic activity of these materials was evaluated in the test reaction of cyclohexene oxidation using H2O2 as oxidizing agent, showing a high conversion of about 93% respect to the maximum, resulting dominant the radicalary mechanism over the direct oxidation mechanism. Apparently, the isolated V ions incorporated into the silica structure would be responsible for the high catalytic activity of these materials.







Similar content being viewed by others
References
Soller-Illia GJAA, Sanchez C, Lebeau B, Patarin J (2002) Chem Rev 4093:102
Kresge CT, Leonowicz ME, Roth WJ, Vartuli JC, Beck JS (1992) Nature 359:710
Beck JS, Vartuli JC, Roth WJ, Leonowicz ME, Kresge CT, Schmitt KD, Chu CT-W, Olson DH, Sheppard EW, McCullen SB, Higgins JB, Schlenker JL (1992) J Am Chem Soc 114:10834
Reddy EP, Davydov L, Smirniotis PG (2002) J Phys Chem B 106:3394
Eimer GA, Casuscelli SG, Ghione GE, Crivello ME, Herrero ER (2006) Appl Catal A 298:232
Eimer GA, Casuscelli SG, Chanquía CM, Elías V, Crivello ME, Herrero ER (2008) Catal Today 133–135:639
Grün M, Lauer I, Unger KK (1997) Adv Mater 9:254
Szegedi Á, Kónya Z, Méhn D, Solymár E, Pál-Borbély G, Horváth ZE, Biró LP, Kiricsi I (2004) Appl Catal A 272:257
Liu X, Sun H, Chen Y, Yang Y, Borgna A (2009) Microporous Mesoporous Mater 121:73
Jha RK, Shylesh S, Bhoware SS, Singh AP (2006) Microporous Mesoporous Mater 95:154
Pârvulescu V, Anastasescu C, Su BL (2003) J Mol Catal A 198:249
Shylesh S, Singh AP (2005) J Catal 233:359
Reddy KM, Moudrakovski I, Sayari A (1994) J Chem Soc Chem Commun 1059
Gontier S, Tuel A (1995) Microporous Mater 5:161
Morey M, Davidson A, Eckert H, Stucky G (1996) Chem Mater 8:486
Moudrakovski I, Sayari A, Ratcliffe CI, Ripmeester JA, Preston KF (1994) J Phys Chem 98:10895
Pârvulescu V, Su BL (2001) Catal Today 69:315
Chao KJ, Wu CN, Chang H, Lee LJ, Hu S-f (1997) J Phys Chem B 101:6341
George J, Shylesh S, Singh AP (2005) Appl Catal A 290:148
Peña ML, Dejoz A, Fornés V, Rey F, Vázquez MI, López Nieto JM (2001) Appl Catal A 209:155
Gao F, Zhang Y, Wan H, Kon Y, Wu X, Dong L, Li B, Chen Y (2008) Microporous Mesoporous Mater 110:508
Sanderson W (2000) Pure Appl Chem 72:1289
Weckhuysen BM, Keller DE (2003) Catal Today 78:25
Gregg SJ, Sing KSW (1982) Adsorption, surface area and porosity. Academic Press, New York
Sayari A, Liu P, Kruk M, Jaroniec M (1997) Chem Mater 9:2499
Hao X-Y, Zhang Y-Q, Wang J-W, Zhou W, Zhang C, Liu S (2005) Microporous Mesoporous Mater 88:38
Velu S, Wang L, Okazaki M, Suzuki K, Tomura S (2002) Microporous Mesoporous Mater 54:113
Parvulescu V, Anastasescu C, Su BL (2003) J Mol Catal A Chem 3919:1
Gomes HT, Selvam P, Dapurkar SE, Figueiredo JL, Faria JL (2005) Microporous Mesoporous Mater 86:287
Eimer GA, Chanquía CM, Sapag K, Herrero ER (2008) Microporous Mesoporous Mater 116:670
Sing KSW, Everett DH, Haul RAW, Moscou Pierotti L, Rouquerol RA, Siemieniewska J (1985) Pure Appl Chem 57:603
Kruk M, Jaroniec M, Ryoo R, Kim JM (1999) Chem Mater 11:2568
Díaz I, Pérez-Pariente J (2002) Chem Mater 14:4641
Lim S, Haller GL (2002) J Phys Chem B 106:8437
Rouquerol F, Rouquerol J, Sing K (1999) Adsorption by powders and porous solids. Principles, methodology and application. Academic Press, London
Efremov D, Fenelonov V (1991) Stud Surf Sci Catal 62:62
Chanquía CM, Sapag K, Rodríguez-Castellón E, Herrero ER, Eimer GA (2010) J Phys Chem C 114:1481
Solsona B, Blasco T, López JM, Peña ML, Rey F, Vidal-Moya A (2001) J Catal 203:443
Chanquía CM, Andrini L, Fernández JD, Crivello ME, Requejo FG, Herrero ER, Eimer GA (2010) J Phys Chem C 114:12221
Eimer GA, Díaz I, Sastre E, Casuscelli SG, Crivello ME, Herrero ER, Perez-Pariente J (2008) Appl Catal A 343:77
Notari B (1996) Adv Catal 41:253
Hu Y, Higashimoto S, Takahashi S, Nagai Y, Anpo M (2005) Catal Lett 100:35
Luan Z, Xu J, He H, Klinowski J, Kevan L (1996) J Phys Chem 100:19595
Farzaneh F, Zamanifar E, Williams CD (2004) J Mol Catal A 218:203
Acknowledgment
G.A.E., K.S. and S.G.C are CONICET Researchers; C.M.Ch. and A.L.C received CONICET Doctoral Fellowship. This work was supported by the CONICET, the UTN-FRC and the UNSL of Argentina, and UdeC of Chile. We thank J. Bazán Aguirre (UTN-FRC Student) for valuable help on some experimental activities. Finally, C.M.Ch. acknowledge to Dra. M.E. Crivello for the participation in international cooperation project MINCYT–CONICYT, Code CH/08/03.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chanquía, C.M., Cánepa, A.L., Sapag, K. et al. Mesoporous Silicates with Spherical Morphology Modified with Vanadium Highly Active in Oxidation of Cyclohexene with H2O2 . Top Catal 54, 160–169 (2011). https://doi.org/10.1007/s11244-011-9635-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-011-9635-8