Abstract
Bahiagrass (Paspalum notatum Flugge) is a warm season, low-input, perennial turfgrass used for residential lawns and sides of road ways. The turf quality of bahiagrass is limited by its open growth habit, light green color, and prolific production of tall seedheads. Genetic improvement of the tetraploid bahiagrass turf cultivar ‘Argentine’ by conventional breeding is difficult due to its apomictic mode of reproduction. Our objective was to explore the potential of in vitro chemical mutagenesis for the generation of uniform, mutant seed progeny with improved turf quality. Scarified, surface sterilized bahiagrass seeds were treated with different concentrations of the chemical mutagen ‘sodium azide’. Callus was induced from these seeds and 19,630 plants were regenerated via somatic embryogenesis. 2,035 of these plants were selected based on their morphological characteristics and transferred to soil. Wildtype bahiagrass and 46 mutant lines (M1 lines) with reduced stem lengths, higher tiller densities or reduced or delayed seedhead formation were established under field conditions. Mutant lines with improved traits and production of viable apomictic seeds were identified and their apomictic M2 progeny was evaluated. A superior line displayed higher density, finer leaves, an upright growth habit, dark green color, reduced seedhead formation and uniform seed progeny in replicated, two location field trials. Beside this improved turf quality, this line also retained the superior drought tolerance and persistence that make bahiagrass a popular low-input turfgrass.



Similar content being viewed by others
References
Acuña CA, Blount AR, Quesenberry KH, Kenworthy KE, Hanna WW (2011) Tetraploid bahiagrass hybrids: breeding technique, genetic variability and proportion of heterotic hybrids. Euphytica 179:227–235
Agharkar M, Lomba P, Altpeter F, Zhang H, Kenworthy K, Lange T (2007) Stable expression of AtGA2ox1 in a low-input turfgrass (Paspalum notatum Flugge) reduces bioactive gibberellin levels and improves turf quality under field conditions. Plant Biotechnol J 5:791–801
Ahloowalia BS, Maluszynski M (2001) Induced mutations: a new paradigm in plant breeding. Euphytica 118:167–173
Akashi R, Hashimoto A, Adachi T (1993) Plant regeneration from seed-derived embryogenic callus and cell suspension cultures of bahiagrass (Paspalum notatum). Plant Cell 90:73–80
Altpeter F, James VA (2005) Genetic transformation of turf-type bahiagrass (Paspalum notatum Flugge) by biolistic gene transfer. Int Turfgrass Soc Res J 10:485–489
Altpeter F, Positano M (2005) Efficient plant regeneration from mature seed derived embryogenic callus of turf-type bahiagrass (Paspalum notatum Flugge). Int Turfgrass Soc Res J 10:479–484
Bovo OA, Mroginski LA (1989) Somatic embryogenesis and plant regeneration from cultured mature and immature embryos of Paspalum notatum (Graminae). Plant Sci 65:217–223
Burton GW (1946) Bahia grass types. J Am Soc Agron 38:273–281
Burton GW (1948) The method of reproduction in common bahiagrass, Paspalum notatum. J Am Soc Agron 40:443–452
Burton GW (1985) Registration of Tifway II bermudagrass. Crop Sci 25:364
Busey P (1980) Gamma ray dosage and mutation breeding in St. augustinegrass. Crop Sci 20:181–184
Forbes I, Burton GW (1961) Cytology of diploids, natural and induced tetraploids, and interspecific hybrids of bahiagrass, Paspalum notatum Flugge. Crop Sci 1:204–206
Ganesan M, Han YJ, Bae TW, Hwang OJ, Chandrasekhar T, Shin AY, Goh CH, Nishiguchi S, Lee HY, Kim J, Song PS (2012) Overexpression of phytochrome A and its hyperactive mutant improves shade tolerance and turf quality in creeping bentgrass and zoysiagrass. Planta 236:1135–1150
Hanna WW, Bashaw EC (1987) Apomixis: its identification and use in plant breeding. Crop Sci 27:1136–1139
Hanna WW, Elsner JE (1999) Registration of TifEagle bermudagrass. Crop Sci 39:1258
Hanna WW, Carrow RN, Powell AJ (1997a) Registration of Tift 94 bermudagrass. Crop Sci 37:1012
Hanna WW, Dobson J, Duncan RR, Thompson D (1997b) Registration of TifBlair centipedegrass. Crop Sci 37:1017
Hanson AA, Juska FV (1959) A ‘Progressive’ mutation induced in Poa pratensis L. by ionizing radiation. Nature 184:1000–1001
Hodges AW, Stevens TJ (2010) Economic contributions of the turfgrass industry in the Florida. University of Florida, IFAS Extension, Gainesville, FL. http://www.fred.ifas.ufl.edu/economic-impact-analysis/pdf/Turfgrass%20Florida%202007.pdf
Jain SM, Brar DS, Ahloowalia BS (1998) Somaclonal variation and induced mutations in crop improvement. Kluwar, The Netherlands
Li R, Bruneau AH, Qu R (2010) Morphological mutants of St. Augustinegrass induced by gamma ray irradiation. Plant Breed 129:412–416
LoSchiavo F, Pitto L, Giuliano G, Torti G, Nuti-Ronchi V, Marazatti D, Vergara R, Orselli S, Terzi M (1989) DNA methylation of embryogenic carrot cell cultures and its variations as caused by mutation, differentiation, hormones, and hypomethylating drugs. Theor Appl Genet 77:325–331
Lu S, Wang Z, Niu Y, Chen Y, Chen H, Fan Z, Lin J, Yan K, Guo Z, Li H (2009) Gamma-ray radiation induced dwarf mutants of turf-type bermudagrass. Plant Breed 128:205–209
Maluszynski M, Ahloowalia BS, Sigurbjornsson B (1995) Application of in vivo and in vitro mutation techniques for crop improvement. Euphytica 85:303–315
Martinez EJ, Urbani MH, Quarin CL, Ortiz JPA (2001) Inheritance of apospory in bahiagrass, Paspalum notatum. Hereditas 135:19–25
Mba C, Afza R, Jankowicz-Cieslak J, Bado S, Matijevic M, Huynh O, Till BJ (2009) Enhancing genetic diversity through induced mutagenesis in vegetatively propagated plants. In: Shu QY (ed) Induced plant muations in the genomics era. Food and Agriculture Organization of the United Nations, Rome, pp 262–265
Morris KN, Shearman RC (2006) NTEP turfgrass evaluation guidelines. http://www.ntep.org/pdf/ratings.pdf
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nagatomi S, Degi K (2009) Mutation breeding of chrysanthemum by gamma field irradiation and in vitro culture. In: Shu QY (ed) Induced plant muations in the genomics era. Food and Agriculture Organization of the United Nations, Rome, pp 258–261
Newman Y, Vendramini J, Blount A (2010) Bahiagrass (Paspalum notatum): overview and management. EDIS Publication SS-AGR-332 Agronomy Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL
Pande S, Khetmalas M (2012) Biological effects of sodium azide and colchicine on seed germination and callus induction in Stevia rebaudiana. Asian J Exp Biol Sci 3:93–98
Pederson JF, Dickens R (1985) Registration of AU centennial centipedegrass. Crop Sci 25:364
Quarin CL, Espinoza F, Martinez EJ, Pessino SC, Bovo OA (2001) A rise of ploidy level induces the expression of apomixis in Paspalum notatum. Sex Plant Reprod 13:243–249
Reynolds WC, Li R, de Silva K, Bruneau AH, Qu R (2009) Field performance of mutant and somaclonal variation lines of St. Augustinegrass [Stenotaphrum secundatum (Walt.) Kuntze]. Int Turfgrass Soc Res J 11:573–582
Rios E, Blount A, Erickson J, Quesenberry K, Altpeter A, Cellon C, Kenworthy K (2013) Root and shoot characterization of mutant turf-type bahia grasses. Int Turfgrass Soc Res J 12:509–516
Sadiq MF, Owais WM (2000) Mutagenicity of sodium azide and its metabolite azidoalanine in Drosophila melanogaster. Mutat Res 469:253–257
Scott JM (1920) Bahia grass. J Am Soc Agron 12:112–113
Shatters RG, Wheeler RA, West SH (1994) Somatic embryogenesis and plant regeneration from callus culture of ‘Tifton 9’ bahiagrass. Crop Sci 34:1378–1384
Smith RL, Grando MF, Li YY, Seib JC, Shatters RG (2002) Transformation of bahiagrass (Paspalum notatum Flugge). Plant Cell Rep 20:1017–1021
Srivastava P, Marker S, Pandey P, Tiwari DK (2011) Mutagenic effects of sodium azide on the growth and yield characteristics in wheat (Triticum aestivum L. em. Thell.). Asian J Plant Sci 10:190–201
Tomlekova NB (2010) Induced mutagenesis for crop improvement in Bulgaria. Plant Mutat Rep 2:4–27
Trenholm LE, Cisar JL, Unruh JB (2003) Bahiagrass for Florida lawns. EDIS Publication ENH6 Environmental Horticulture Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL
Zhang H, Lomba P, Altpeter F (2007) Improved turf quality of transgenic bahiagrass (Paspalum notatum Flugge) constitutively expressing the ATHB 16 gene, a repressor of cell expansion. Mol Breed 20:415–423
Acknowledgments
We would like to thank the Southwest Florida Water Management District for funding this research project, Loan Ngo for technical assistance, and Dr. Janice Zale for contributing to the editing of this manuscript and Sun Gro Horticulture for donation of the Fafard #2 potting mix.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
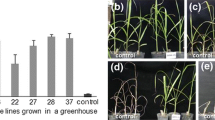
Visual Ratings for desirable turf characteristic in the field. a stem length b density c leaf texture. Visual ratings were assigned a rating using a 1–9 scale with 1 being (a) shortest stem length (b) least dense or (c) coarsest leaf texture and 9 being (a) longest stem length, (b) most dense or (c) finest leaf texture. * Indicates significant difference compared to WT at α = 0.05. (PPT 152 kb)
Rights and permissions
About this article
Cite this article
Kannan, B., Davila-Olivas, N.H., Lomba, P. et al. In vitro chemical mutagenesis improves the turf quality of bahiagrass. Plant Cell Tiss Organ Cult 120, 551–561 (2015). https://doi.org/10.1007/s11240-014-0621-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-014-0621-2