Abstract
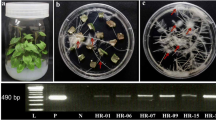
Hairy root cultures of Mitracarpus hirtus L. were obtained after transforming leaf-disc explants with wild strain Agrobacterium rhizogenes A13. The root cultures of M. hirtus showed high efficiency of shoot formation in both transformed and non-transformed cultures when illuminated with light. However, transformed hairy root proliferation was approximately 3.8–5 times higher than the control in both solidified and liquid plant growth regulator free media. Putatively transformed roots were identified by the presence of the rol gene via PCR. Integration of the rol gene into the plant genome was confirmed via Southern blot analysis after 5 months with no detection in non-transformed roots. In addition, the effect of 2-chloro-4-pyridyl-N-phenylurea (CPPU), a synthetic cytokinin, when applied as an elicitor for hairy root cultures of M. hirtus was investigated. The 24-day-old hairy root cultures of high root proliferation line R107-3, were incubated for 48 h in media supplemented with 0 or 5 mg l−1 CPPU. The methanolic extracts of root tissues were subsequently analyzed for biochemical constituents using Gas Chromatography Mass Spectrometry (GC-MS). The alteration of plant secondary metabolites produced after CPPU treatment was analyzed. Compared to the control (with quality higher than 80 %), six unique compounds were found, five original compounds absent, 11 with increased, and five with decreased contents. Increased contents of two metabolites, chrysophanol and 2-methoxy-4-vinylphenol, showed pharmaceutical potential. CPPU was also found to elicit the alkaloid compound, Eseroline, 7-bromo-, methylcarbamate (ester), which could not be detected in the non-treated sample. The findings of this study demonstrate the establishment of transgenic hairy root of M. hirtus and the application of CPPU as an elicitor to induce variations in plant secondary metabolite that shows its potential to apply for bio-reactor system.



Similar content being viewed by others
References
Abere TA, Onwukaeme DN, Eboka CJ (2007) Pharmacognostic evaluation of the leaves of Mitracarpus scaber Zucc (Rubiaceae). Trop J Pharm Res 6(4):849–853
Akutsu M, Ishizaki T, Sato H (2004) Transformation of the monocot Alstroemeria by Agrobacterium rhizogenes. Mol Breed 13:69–78
Amoo SO, Aremu AO, Van Staden J (2012) In vitro plant regeneration, secondary metabolite production and antioxidant activity of micropropagated Aloe arborescens Mill. Plant Cell Tissue Org 111:345–358
Anderson JA (1985) Conversion of emodin to chrysophanol in a cell-free system from Pyrenochaeta terrestris. Phytochemistry 25(1):103–106
Bourgaud F, Gravot A, Milesi S, Gonteir E (2001) Production of plant secondary metabolites: a historical perspective. Plant Sci 161:839–851
Burkill HM (1970). The useful plants of west tropical Africa (vol 4). Royal Botanic Gardens, Kew. 969 pages
Caspeta L, Quintero R, Villarreal ML (2005) Novel airlift reactor fitting for hairy root cultures: developmental and performance studies. Biotechnol Prog 21:735–740
Chang L, Ramireddy E, Schmulling T (2013) Lateral root formation and growth of Arabidopsis is redundantly regulated by cytokinin metabolism and signaling genes. J Exp Bot 64(16):5021–5032
Chen M, Chory J, Fankhauser C (2004) Light signal transduction in higher plants. Annu Rev Genet 38:87–117
Cimanga RK, Kambu K, Tona L, Bruyne TD, Sandra A, Totte J, Pieters L, Vlietinck AJ (2004) Antibacterial and antifungal activities of some extracts and fractions of Mitracarpus scaber Zucc (Rubiaceae). J Nat Remedies 4(1):17–25
Crozier A, Clifford MN, Ahi hara H (2006) Plant secondary metabolites: occurrence, structure and role in the human diet, vol 1. Blackwell, Oxford, p 372
Daimon H, Fukami M, Mii M (1990) Hairy root formation in peanut by the wild type strains of Agrobacterium rhizogenes. Plant Tissue Cult Lett 7:31–34
Doyle JJ, Doyle JJ (1990) Isolation of plant DNA from fresh tissues. Focus 12:13–15
Emam SS, Abd El-Moaty HI (2009) Glucosinolates, phenolic acids and anthraquinones of Isatis microcarpa boiss and Pseuderucaria clavate (Boiss&Reut.) family: Cruciferae. J Appl Sci Res 5(12):2315–2322
Emery RJ, Ma Q, Atkins CA (2000) The forms and sources of cytokinins in developing white lupine seeds and fruits. Plant Physiol 123:1593–1604
Farah A, Donangelo CM (2006) Phenolic compounds in coffee. Braz J Plant Physiol 18(1):23–36
Gao B, Chen Y, Zhang M, Xu Y, Pan S (2011) Chemical composition, antioxidant and antimicrobial activity of Pericarpium Citri Reticulatae essential oil. Molecules 16:4082–4096
Hoshino Y, Mii M (1998) Bialaphos stimulates shoot regeneration from hairy roots of snapdragon (Antirrhinum majus L.) transformed by Agrobacterium rhizogenes. Plant Cell Rep 17:256–261
Hosokawa K, Matsuki R, Oikawa Y, Yamamura S (1997) Genetic transformation of gentian using wild-type Agrobacterium rhizogenes. Plant Cell Tissue Org 51:137–140
Kalaivani CS, Sathish SS, Janakiraman N, Johnson M (2012) GC-MS studies on Andrographis paniculata (Burm.f.) Wall ex Nees - A medicinally important plant. Int J Med Arom Plants 2(1):69–74
Kim OT, Yoo NH, Kim GS, Kim YC, Bang KH, Hyun DY, Kim SH, Kim MY (2013) Stimulation of Rg3 ginsenoside biosynthesis in ginseng hairy roots elicited by methyl jasmonate. Plant Cell Tissue Org 112(1):87–93
Kiyokawa S, Kikuchi Y, Kamada H, Harada H (1992) Detection of rol genes of Ri plasmids by PCR method and its application to confirmation of transformation. Plant Tissue Cult Lett 9:94–98
Liu Y, Wang H, Ye HC, Li GF (2005) Advances in the plant isoprenoid biosynthesis pathway and its metabolic engineering. J Interg Plant Biol 47(7):769–782
Martin D, Tholl D, Gershenzon J, Bohlmann J (2002) Methyl jasmonate induces traumatic resin ducts, terpenoid resin biosynthesis, and terpenoid accumulation in developing xylem of Norway spruce stems. Plant Physiol 129:1003–1018
Mok DWS, Mok MC (2001) Cytokinin metabolism and action. Annu Rev Plant Physiol Plant. Mol Biol 52:89–118
Moubayidin L, Mambro RD, Sabatini S (2009) Cytokinin–auxin crosstalk. Trends Plant Sci 14(10):557–562
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Plant Physiol 15:473–497
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4325
Ni CH, Chen PY, Lu HF, Yang JS, Huang HY, Wu SH, Ip SW, Wu CT, Chiang SY, Lin JG, Wood WG, Chung JC (2012) Chrysophanol-induced necrotic-like cell death through an impaired mitochondrial ATP synthesis in Hep3B human liver cancer cells. Arch Pharm Res 35(5):887–895
Ohara A, Akasaka Y, Daimon H, Mii M (2000) Plant regeneration from hairy roots induced by infection with Agrobacterium rhizogenes in Crotalaria juncea L. Plant Cell Rep 19:563–568
Owolabi MS, Lawal OA, Dosoky NS, Satyal P, Setzer WN (2013) Chemical composition, antimicrobial, and cytotoxic assessment of Mitracarpus scaber Zucc. (Rubiaceae) essential oil from southwestern Nigeria. Am J Ess Oils Nat Prod 1(1):4–6
Pal A, Swain SS, Mukherjee AK, Chand PK (2013) Agrobacterium pRi TL-DNA rolB and TR-DNA Opine Genes Transferred to the Spiny Amaranth (Amaranthus spinosus L.), A Nutraceutical Crop. Food Technol Biotechnol 51(1):26–35
Pansuksan K, Sangthong R, Nakamura I, Mii M, Supaibulwatana K (2014) Tetraploid induction of Mitracarpus hirtus L. by colchicine and its characterization including antibacterial activity. Plant Cell Tissue Org 117(3):381–391
Peleg H, Naim M, Zehavi U, Rouseff RL, Nagy S (1992) Pathways of 4-vinylguaiacol formation from ferulic acid in model solutions of orange juice. J Agric Food Chem 40(5):764–767
Pichersky E, Gershenzon J (2002) The formation and function of plant volatiles: perfumes for polliator attraction and defense. Curr Opin Plant Biol 5:237–243
Quevedo C, Perassolo M, Alechine E, Corach D, Giulietti AM, Talou JR (2010) Increasing anthraquinone production by overexpression of 1-deoxy-D-xylulose-5-phosphate synthase in transgenic cell suspension cultures of Morinda citrifolia. Biotechnol Lett 32(7):997–1003
Roy S, Rao K, Bhuvaneswari CH, Giri A, Mangamoori LN (2010) Phytochemical analysis of Andrographis paniculata extract and its antimicrobial activity. World J Microbiol Biotechnol 26:85–91
Sa G, Mi M, He-chun Y, Ben-ye l, Guo-feng L, Kang C (2001) Effects of ipt gene expression on the physiological and chemical characteristics of Artemisia annua L. Plant Sci 160(4):691–698
Saitou T, Kamada H, Harada H (1992) Light requirement for shoot regeneration in horseradish hairy roots. Plant Physiol 99:1336–1341
Sakakibara H (2006) Cytokinins: activity, biosynthesis, and translocation. Annu Rev Plant Biol 57:431–449
Saniewski M, Miyamoto K, Ueda J (1998) Methyl jasmonate induces gums and stimulates anthocyanin accumulation in peach shoots. J Plant Growth Regul 17:121–124
Satdive RK, Fulzele DP, Eapen S (2007) Enhanced production of azadirachtin by hairy root cultures of Azadirachta indica A. Juss by elicitation and media optimization. J Biotechnol 128(2):281–289
Silici S, Kutluca S (2005) Chemical composition and antibacterial activity of propolis collected by three different races of honeybees in the same region. J Ethnopharmacol 99:69–73
Srivastava S, Srivastava AK (2007) Hairy root culture for mass-production of high-value secondary metabolites. Crit Rev Biotechnol 27(1):29–43
Tatsuo K (2011) Identification of plant cytokinin biosynthetic enzymes as dimethylallyl diphosphate: ATP/ADP isopentenyltransferases. Plant Cell Physiol 42(7):677–685
Theboral J, Sivanandhan G, Subramanyam K, Arun M, Selvaraj N, Manickavasagam M, Ganapathi A (2014) Enhanced production of isoflavones by elicitation in hairy root cultures of Soybean. Plant Cell Tissue Org 117(3):477–481
Wang JW, Zhang Z, Tan RX (2001) Stimulation of artemisinin production in Artemisia annua hairy roots by the elicitor from the endophytic Colletotrichum sp. Biotechnol Lett 23(11):857–860
Wink M (1998) A short history of alkaloids. In: Roberts MF, Wink M (eds) Alkaloids: biochemistry, ecology, and medicinal applications. Plenum Press, New York, pp 11–44
Xie D, Wang L, Ye H, Li G (2000) Isolation and production of artemisinin and stigmasterol in hairy root cultures of Artemisia annua. Plant Cell Tissue Org 63:161–166
Zhai DD, Jin HZ, Zong JJ (2010a) A new sesquiterpene from hairy root culture of Artemisia annua. Chinese Chem Lett 21:590–592
Zhai DD, Supaibulwatana K, Zhong JJ (2010b) Inhibition of tumor cell proliferation and induction of apoptosis in human lung carcinoma 95-D cells by a new sesquiterpene from hairy root cultures of Artemisia annua. Phytomedicine 17:856–861
Zhang L, Demain A (2005) Natural products: drug discovery and therapeutic medicine. Humana Press, Totowa, pp 197–228
Acknowledgments
This work was supported by Mahidol University and a partial Grant by the “Strategic Consortia for Capacity Building of University Faculties and Staff of Thailand Research Fund” from the Commission on Higher Education (CHE), Thailand. The authors are grateful for partial support of the Excellent International Student Scholarship at Chiba University Program from the Japanese government through Chiba University. This research work was conducted under the collaboration of Mahidol University and Chiba University for the double degree program of K. Pansuksan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pansuksan, K., Mii, M. & Supaibulwatana, K. Phytochemical alteration and new occurring compounds in hairy root cultures of Mitracarpus hirtus L. induced by phenylurea cytokinin (CPPU). Plant Cell Tiss Organ Cult 119, 523–532 (2014). https://doi.org/10.1007/s11240-014-0552-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-014-0552-y