Abstract
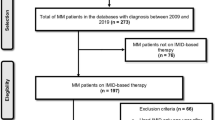
Venous thromboembolism (VTE) is among the complications of Multiple Myeloma (MM) and may occur in up to 10% of this patient population. However, medications used in MM therapy such as immunomodulators (IMID) may raise these rates. Thus, risk prediction models have been developed to quantify the risk of VTE in MM patients. The aim of this study is to compare the performance of three risk assessment models for VTE in newly diagnosed MM (NDMM) patients using immunomodulatory agents. A historical cohort study during a 10-year period in a Brazilian metropolis with NDMM treated with IMID. Data were collected from patient’s medical charts for the period of one year to calculate the scores using IMPEDE VTE, SAVED, and International Myeloma Working Group (IMWG) guidelines. The area under the curve (AUC) of the Receiver Operating Characteristic curve analysis was calculated to assess the discriminative power of three risk assessment models. We included 131 patients (9 in the VTE group versus 122 in the non VTE group). According to IMPEDE, 19.1, 62.6, and 18.3% of patients were considered low, intermediate, and high risk, respectively. SAVED classified 32.1% as high risk and 64.9% had ≥2 risk factors based on IMWG guidelines. The AUC of the IMPEDE VTE score was 0.80 (95% CI 0.66–0.95, p = 0.002), of the SAVED score was 0.69 (95% CI 0.49–0.89, p = 0.057), and of the IMWG risk score was 0.68 (95% CI 0.48–0.88, p = 0.075). IMPEDE VTE was the most accurate in predicting the development of VTE in Brazilian patients on IMID therapy. The SAVED score and the IMWG guidelines did not show discriminative ability in predicting VTE based on the population involved in this study.



Similar content being viewed by others
References
Malta JS, Silveira LP, de Drummond PLM, Costa NL, dos Santos RMM, Reis IA et al (2021) Validity and reliability of the QLQ-MY20 module for assessing the health-related quality of life in Brazilians with multiple myeloma. Curr Med Res Opin. 37(7):1163–1169
Fotiou D, Gavriatopoulou M, Terpos E (2020) Multiple myeloma and thrombosis: Prophylaxis and risk prediction tools. Cancers (Basel). 12(1):191
Baker HA, Brown AR, Mahnken JD, Shireman TI, Webb CE, Lipe BC (2019) Application of risk factors for venous thromboembolism in patients with multiple myeloma starting chemotherapy, a real-world evaluation. Cancer Med 8(1):455–462
Talamo GP, Ibrahim S, Claxton D, Tricot GJ, Fink LM, Zangari M (2009) Hypercoagulable states in patients with multiple myeloma can affect the thalidomide-associated venous thromboembolism. Blood Coagul Fibrinolysis 20(5):337–339
Palmaro A, Rougé-Bugat ME, Gauthier M, Despas F, Moulis G, Lapeyre-Mestre M (2017) Real-life practices for preventing venous thromboembolism in multiple myeloma patients: a cohort study from the French health insurance database. Pharmacoepidemiol Drug Saf 26(5):578–586
Kristinsson SY, Landgren O (2012) Thromboprophylaxis in multiple myeloma: Is the evidence there? Expert Rev Anticancer Ther 12(3):291–294
Zangari M, Barlogie B, Cavallo F, Bolejack V, Fink L, Tricot G (2007) Effect on survival of treatment-associated venous thromboembolism in newly diagnosed multiple myeloma patients. Blood Coagul Fibrinolysis 18(7):595–598
Leclerc V, Karlin L, Herledan C, Marchal L, Baudouin A, Gouraud A et al (2022) Thromboembolic events and thromboprophylaxis associated with immunomodulators in multiple myeloma patients: a real-life study. J Cancer Res Clin Oncol 148(4):975–984
Kandemir EA, Bayraktar-Ekincioglu A, Kilickap S (2021) Assessment of adherence to cancer-associated venous thromboembolism guideline and pharmacist’s impact on anticoagulant therapy. Support Care Cancer 29(3):1699–1709
Sanfilippo KM (2020) Assessing the risk of venous thromboembolism in multiple myeloma. Thromb Res 191(January):S74–S78
Anderson SM, Beck B, Sterud S, Lockhorst R, Ngorsuraches S (2019) Evaluating the use of appropriate anticoagulation with lenalidomide and pomalidomide in patients with multiple myeloma. J Oncol Pharm Pract 25(4):806–812
Carrier M, Le Gal G, Tay J, Wu C, Lee AY (2011) Rates of venous thromboembolism in multiple myeloma patients undergoing immunomodulatory therapy with thalidomide or lenalidomide: a systematic review and meta-analysis. J Thromb Haemost 9(4):653–663
Jha AK, Larizgoitia I, Audera-Lopez C, Prasopa-Plaizier N, Waters H, Bates DW (2013) The global burden of unsafe medical care: analytic modelling of observational studies. BMJ Qual Saf 22(10):809–815
Mosaad M, Elnaem MH, Cheema E, Ibrahim I, Ab Rahman J, Kori AN et al (2021) Cancer-associated thrombosis: a clinical scoping review of the risk assessment models across solid tumours and haematological malignancies. Int J Gen Med 14(May):3881–3897
Sanfilippo KM, Luo S, Wang TFTF, Fiala M, Schoen M, Wildes TM et al (2019) Predicting venous thromboembolism in multiple myeloma: development and validation of the IMPEDE VTE score. Am J Hematol. 94(11):1176–84
Li A, Wu Q, Luo S, Warnick GS, Zakai NA, Libby EN et al (2019) Derivation and validation of a risk assessment model for immunomodulatory drug-associated thrombosis among patients with multiple myeloma. JNCCN J Natl Compr Cancer Netw 17(7):840–847
Palumbo A, Rajkumar SV, Dimopoulos MA, Richardson PG, San Miguel J, Barlogie B et al (2008) Prevention of thalidomide- and lenalidomide-associated thrombosis in myeloma. Leukemia 22(2):414–423
(IHME) I for HM and E. GBD Results. [Internet]. IHME, University of Washington. 2020. https://vizhub.healthdata.org/gbd-results/ [accessed 2022 Aug 24].
de Crusoé EQ, Pimenta FCF, Maiolino A, de Castro NS, Pei H, Trufelli D et al (2021) Results of the daratumumab monotherapy early access treatment protocol in patients from Brazil with relapsed or refractory multiple myeloma. Hematol Transfus Cell Ther. 43(4):417–23
Silveira LP, de Pádua CAM, de Drummond PLM, Malta JS, dos Santos RMM, Costa NL et al (2021) Adherence to thalidomide in patients with multiple myeloma: a cross-sectional study in a Brazilian metropolis. J Oncol Pharm Pract. 28:373–380
Mandrekar JN (2010) Receiver operating characteristic curve in diagnostic test assessment. J Thorac Oncol 5(9):1315–1316
Covut F, Ahmed R, Chawla S, Ricaurte F, Samaras CJ, Anwer F et al (2021) Validation of the IMPEDE VTE score for prediction of venous thromboembolism in multiple myeloma: a retrospective cohort study. Br J Haematol 193(6):1213–1219
Bao L, Wang Y, Lu M, Zhuang J, Chu B, Shi L et al (2022) Validation of the Impede VTE score for predction of venous thromboembolism in Chinese patients with multiple myeloma: a retrospective cohort study. Blood 140(Supplement 1):5146–5147
Calafiore V, Giamporcaro S, Conticello C, Romano A, Parisi M, Giuffrida G et al (2020) A real-life survey of venous thromboembolic events occurring in myeloma patients treated in third line with second-generation novel agents. J Clin Med 9(9):1–14
Chalayer E, Talbot A, Frenzel L, Karlin L, Collet P, Guyotat D et al (2022) Prediction of venous thromboembolism in patients with multiple myeloma treated with lenalidomide, bortezomib, dexamethasone, and transplantation: Lessons from the substudy of IFM/DFCI 2009 cohort. J Thromb Haemost. 20:1–9
Teste A, Thollot H, Tinquaut F, Sotton S, Tardy B, Moreau P et al (2021) PO-82 Impede VTE vs saved scores to predict the risk of venous thromboembolism in newly diagnosed multiple myeloma with immunomodulatory drugs: how to choose? Thromb Res 200:S61–S62
Sanfilippo KM, Carson KR, Wang TF, Luo S, Edwin N, Kuderer N et al (2022) Evaluation of the Khorana score for prediction of venous thromboembolism in patients with multiple myeloma. Res Pract Thromb Haemost 6(1):1–7
Hosmer D, Lemeshow S, Sturdivant R (2013) Applied logistic regression, 3rd edn. Wiley, Hoboken
Oliveira JADQ, Ribeiro ALP, Ribeiro DD, Nobre V, Rocha MODC, Martins MAP (2018) Predictive ability of scores for bleeding risk in heart disease outpatients on warfarin in Brazil. PLoS ONE 13(10):1–15
Kumar SK, Callander NS, Adekola K, Anderson LD Jr, Baljevic M, Campagnaro E et al (2022) Multiple myeloma. Natl Compr Cancer Netw 4(5):1–107
Baljevic M, Sborov DW, Lim MY, Hillengass J, Martin T, Castillo JJ et al (2022) Optimizing thromboembolism prophylaxis for the contemporary age of multiple myeloma. JNCCN J Natl Compr Cancer Netw 20(1):91–95
Acknowledgements
The authors would like to thank the Empresa Brasileira de Serviços Hospitalares (Ebserh) for all the support provided.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by IHFdC, CAMdP, and AMMR. The first draft of the manuscript was written by IHFdC and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial conflict of interest or relevant disclosures related to this study.
Ethical approval
The present study was designed in accordance with the ethical standards of research involving human participants of the national research committee (CNS 466/2012) and was approved by the Research Ethics Committee of the Federal University of Minas Gerais (No. 3.305.726). Patients who agreed to participate in the research signed a free and informed consent form.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
da Costa, I.H.F., de Pádua, C.A.M., de Miranda Drummond, P.L. et al. Comparison of three risk assessment models for thromboembolism in multiple myeloma patients receiving immunomodulators: a Brazilian historical cohort. J Thromb Thrombolysis 56, 147–155 (2023). https://doi.org/10.1007/s11239-023-02817-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-023-02817-7