Abstract
Assessing the risk of recurrent venous thromboembolism (VTE), particularly when patients are anticoagulated, remains a major challenge largely due to the lack of biomarkers. Blood was sampled from adult VTE patients recruited between January 2018 and September 2020, while receiving therapeutic anticoagulation. Results were compared to 144 healthy subjects (34.7% male, median age 42 years). Overall haemostatic potential (OHP) assay, a spectrophotometric assay, was performed on platelet-poor plasma, in which fibrin formation (triggered by small amounts of thrombin (overall coagulation potential, OCP)) and fibrinolysis (by the addition of thrombin and tissue plasminogen activator (OHP)) are simultaneously measured. Results were obtained from 196 patients (52.6% male, mean age 57.1 years). Compared to healthy subjects, VTE patients displayed significantly higher OCP (39.6 vs 34.5 units, p < 0.001) and OHP (9.3 vs 6.4 units, p < 0.001) as well as lower overall fibrinolytic potential (75.6 v s81.1%, p < 0.001). All 16 VTE recurrences, including 11 unprovoked, occurred above an OCP cut-off of 40th percentile (recurrence rate 4.32/100 patient-years (100PY), 95% confidence interval (CI) 2.39–7.80, p = 0.002). Of 97 patients who subsequently discontinued anticoagulation, all unprovoked VTE recurrences (n = 9) occurred above the 40th OCP percentile (recurrence rate 9.10/100PY, 95% CI 4.74–17.49, p = 0.005) and the 40th OHP percentile (recurrence rate 8.46/100PY, 95% CI 4.40–16.25, p = 0.009). Our pilot study demonstrates that the OHP assay can detect a hypercoagulable and hypofibrinolytic state in anticoagulated VTE patients and may be able to risk stratify VTE recurrence, allowing for more individualised decision on long-term anticoagulation. Further larger prospective studies are required.
Similar content being viewed by others
Highlights
-
Compared to healthy subjects, VTE patients displayed significantly high OCP, OHP and lower overall fibrinolytic potential
-
All 16 VTE recurrences occurred above the 40th OCP percentile
-
Of patients who discontinued anticoagulation, all 9 unprovoked VTE recurrenced occurred above the 40th OCP and OHP percentiles
-
OCP performed better than D-dimer in ability to predict unprovoked recurrent VTE (ROC AUC 0.71 vs 0.43)
Introduction
Assessing the individual risk of recurrent venous thromboembolic (VTE) events remains one of the major challenges of VTE management. While large studies have reported up to 30% recurrence rate at 5 years following a first unprovoked proximal deep vein thrombosis (DVT) or pulmonary embolus (PE) [1], up to 70% of patients may not subsequently develop VTE recurrence [2]. Recommending indefinite anticoagulation to minimise the risk of recurrence is not without its risks and in situations of clinical equipoise, there is a need for an individualised and targeted approach to risk assessment.
Several biomarkers and assays have been explored as adjuncts to clinical decision making, and among the most well-known of these include D-dimer and thrombin generation assays [3], which have been associated with increased risk of VTE recurrence [4,5,6,7,8,9]. An important drawback of these biomarkers is the requirement to test when the patient has discontinued anticoagulation, which can be difficult in the VTE population as the risk of recurrent events is highest within the first 12 months following anticoagulation cessation [2, 10]. The only model to incorporate biomarker testing whilst anticoagulated is the HERDOO2 [11]. However, this score has been shown to lack discriminatory ability in males, with all males recommended for long-term anticoagulation following a first unprovoked VTE irrespective of their individual characteristics.
There has been a renewal of interest into examining the fibrinolytic system and its role in recurrence of VTE, with several recent studies demonstrating markers of reduced fibrinolysis including a prolonged clot lysis time (CLT) and low permeability coefficient (Ks) to be independently associated with VTE recurrence following a first DVT [12] or PE [13]. The Overall Haemostatic Potential (OHP) assay is a global coagulation assay that can examine both fibrin production and lysis within the same test system [14]. This assay has been shown to be able to detect hypercoagulable states following PE [15] and pregnancy-associated VTE [16], as well as potentially predict thrombosis in other hypercoagulable states such as diabetes mellitus [17], ischaemic heart disease [18] and antiphospholipid syndrome [19]. However, its role in risk stratification of future VTE recurrence has not been previously explored.
We aimed to investigate the use of the OHP assay in newly diagnosed patients following VTE and explore its role in the risk stratification of VTE recurrence, including in patients whilst receiving anticoagulation.
Methods
This is a prospective observational study of adult patients aged 18 years and over with VTE, recruited through the haematology outpatient clinics of a sub-tertiary hospital, Northern Hospital, Victoria, Australia. At the time of recruitment, patients were required to be in their active phase of VTE treatment with therapeutic anticoagulation (warfarin, rivaroxaban or apixaban). Exclusion criteria included superficial thrombophlebitis, unconfirmed VTE diagnosis without objective radiological evidence of VTE, significant anaemia and inability to undergo venepunctures at our institution. For this analysis, non-lower limb DVT and non-PE patients such as those with upper limb DVT, cerebral venous sinus thrombosis (CVST) and splanchnic vein thrombosis were also excluded. Baseline information for all patients were collected at the initial clinical visit including age, gender, characteristics of the VTE, risk factors for VTE, smoking status, weight, and their relevant co-morbidities. Written informed consent was obtained from every study participant. This study was approved by the Human Research Ethics Committee of Austin Health (Austin429) and Northern Health (HREC14).
All blood samples were obtained by trained phlebotomists using standard venepuncture techniques via a 21G needle while the participant was still receiving therapeutic anticoagulation. The duration of anticoagulation was determined by the clinician, in discussion with the patient. Routine investigations including full blood count, coagulation studies, D-dimer and renal function tests, thrombophilia screen (including protein C, protein S, antithrombin, factor V Leiden mutation, prothrombin gene mutation), lupus anticoagulant, anticardiolipin IgG and b2-glycoprotein-1 IgG were performed using standard accredited laboratory protocol. The STA® fibrinogen kit was used to measure the fibrinogen levels by the Clauss method. D-dimer was measured using the immunoturbidimetric method with the STA-LIATEST D-Di Plus kit. The remaining citrate samples were double centrifuged at 2500 g for 10 min to obtain platelet-poor plasma and the supernatant stored at − 80 °C within two hours of collection. These samples were then thawed at 37 °C and batch-tested for the OHP assay.
Healthy subjects
The OHP results of the VTE cohort were compared to those of a previously published cohort of 144 healthy subjects (34.7% male, median age 42 years) [17, 20], using the same methodology as that described above. The inclusion criteria for healthy controls were stringent including no known cardiovascular risk factors and thrombosis history, medications such as anticoagulants, anti-platelets and oral contraceptives, as well as a negative thrombophilia screen.
Overall haemostatic potential (OHP) assay
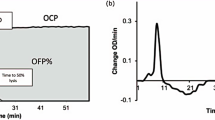
The OHP assay is derived from a fibrin aggregation curve formed from repeated spectrophotometric measurements of platelet-poor plasma (Fig. 1). 75 μL of thawed PPP was added to wells with 75 μL of buffer containing either (i) Tris, NaCl, CaCl2 (final concentration 66 nM Tris, 130 mM NaCl, 35 mL CaCl2; pH 7.0) and thrombin (0.006 IU/mL) to generate the overall coagulation potential (OCP) or (ii) Tris, NaCl, CaCl2, thrombin and tissue plasminogen activator (tPA) (600 ng/mL) to generate the OHP. The two fibrin-aggregation curves (OCP and OHP) are cumulatively calculated from the FLUOstar Optima (BMG Labtech) plate reader at 405 nM. The difference between the area underneath the two curves gives the overall fibrinolytic potential (OFP%).
OHP assay. OCP is the area under the curve (AUC) of the OD curve obtained by the addition of thrombin (0.006 IU/ml) with CaCl2. OHP is the AUC of the curve by adding thrombin (0.006 IU/ml), CaCl2 and tPA. The OFP% is the difference between the AUCs OCP and OHP. OCP overall coagulation potential; OHP overall haemostatic potential; OFP% overall fibrinolytic potential
Patients were followed-up every 3 months during the initial anticoagulation period, then 6–12 months for up to 3 years following anticoagulation cessation, or for 3 years from VTE diagnosis for those who remained on indefinite anticoagulation. Each follow-up visit involved review for recurrent VTE. Recurrent VTE was defined as a new episode or progression of DVT or PE, objectively measured by compression ultrasonography and/or computed tomography pulmonary angiography (CTPA) or ventilation/perfusion (V/Q) scan. Superficial thrombophlebitis was not classified as recurrent VTE. Patients were classified as falling into a ‘clinical equipoise’ group with respects to VTE recurrence risk if they were following a first unprovoked or minimally provoked major VTE (defined as proximal DVT and/or PE). Minimally provoking factors included minor transient factors including long-haul flights more than 4 h duration, lower limb injury with partial immobilisation or hospital admission for medical reasons.
Statistical analysis
Statistical analysis was performed using Stata version 17.0 (StataCorp, College Stations, Texas, USA). Comparisons between patient groups were conducted using Student’s t-tests for those variables which were normally distributed and presented as means and standard deviation. Mann–Whitney (rank-sum) was performed for those variables found to be non-normally distributed and were presented as medians and interquartile ranges (IQR). Categorical variables were presented as counts and frequencies with χ2 tests to test for differences. For multivariate analysis, skewed variables were transformed into a normal distribution before linear regression was performed to account for confounders including age and sex. Receiver Operative Curve (ROC) analysis was used to determine the area-under-the-curve (AUC) for D-dimer and OCP. A two-tailed p-value of less than 0.05 was attributed statistical significance.
Time-to-event analysis was performed with recurrent VTE or recurrent unprovoked VTE as endpoints. Patients without VTE recurrence who were alive or lost to follow-up were censored at the date of last follow-up. Incidences of recurrence were compared between groups using cox proportional hazards regression analysis. Average rates of VTE recurrence are reported as events per 100 patient-years (100PY) to account for the differing lengths of follow up across patients. No deaths occurred in the subgroup without long-term anticoagulation, so this was not included as a competing risk.
Results
Between January 2018 and September 2020, a total of 235 patients were recruited, of which 208 had blood sampled by the time of this interim analysis (Fig. 2). 196 samples analysed were included in the final analysis, after exclusion of 3 patients with upper limb DVT, 2 with CVST, 1 with renal vein thrombus, 4 with failed assays and 2 who had blood collected prior to anticoagulation commencement. Five patients who developed VTE recurrence prior to blood sampling were excluded from the time-to-event analysis, due to a significantly shorter time interval to blood sampling in this group, compared to patients who developed VTE recurrence following blood sampling (mean number of days 20.2 v 91.9, p = 0.013).
Baseline characteristics of patients are displayed in Table 1. The median time from VTE diagnosis to time of blood collection was 79 days (range 41–114 days). There were 92 (46.9%) patients with PE with or without DVT, 47 (24%) with proximal DVT alone and 57 (29.1%) with isolated distal DVT (IDDVT). Anticoagulation was subsequently discontinued in 100 (51.0%) patients. There were 16 VTE recurrences following blood collection. The median duration from time of diagnosis to VTE recurrence was 1.35 years (range 0.36–4.38 years).
VTE patients vs healthy controls
Table 2 displays the OHP assay results for the 196 VTE study patients, compared with 144 previously recruited healthy subjects. After multivariate analysis adjusting for age and sex, OCP and OHP parameters remained significantly higher in VTE patients, and OFP% significantly lower, compared to healthy subjects.
VTE subtypes
In the overall study population (n = 196), the mean OCP, OHP and OFP parameters were not significantly different in patients following provoked versus unprovoked VTE (OCP 40.08 vs 40.83 units, p = 0.65; OHP 10.8 vs 10.55, p = 0.77 units; OFP 74.06 vs 74.66%, p = 0.62), or major versus minor VTE (OCP 40.87 vs 39.70 units, p = 0.52; OHP 10.92 vs 10.00 units, p = 0.30; OFP 74.14 vs 75.08%, p = 0.46).
Within the subgroup of 100 patients who subsequently discontinued anticoagulation, patients with unprovoked index VTE (n = 49) displayed significantly higher OCP than provoked VTE (43.06 v 38.70 units, p = 0.034) but no differences for OHP (11.35 v 10.01 units, p = 0.15) or OFP (74.01 v 74.88%, p = 0.55). Patients with major VTE index event (n = 57) also displayed higher OCP compared to minor VTE (42.72 v 38.34, p = 0.035) but no significant differences for OHP (11.34 v 9.79, p = 0.10), or OFP (74.03 v 75.00, p = 0.50).
VTE recurrence
At the time of this interim analysis, there was a combined total follow-up time of 443.20 patient years, during which there were 16 VTE recurrences, of which 11 were unprovoked events and 10 were major VTE recurrences. Four recurrent VTE occurred during anticoagulation. Baseline characteristics and the OHP assay results for 191 VTE patients with and without VTE recurrence are displayed in Table 1. There were no significant differences in the OHP assay results of those with had recurrent VTE compared with those who did not recur.
Time to event analysis
Time-to-event analysis of the entire cohort of 191 patients showed that an OCP cut-off corresponding to the 40th percentile of OCP results was able to stratify recurrent VTE events (n = 16) (Hazard ratio (HR) 5.34, 95% confidence interval (CI) 1.21–23.58, p = 0.027) (Fig. 3A). All recurrent unprovoked VTE (n = 11) occurred above the OCP 40th percentile (recurrence rate 4.32 per 100PY, 95% CI 2.39–7.80, p = 0.002) (Fig. 3C). OHP at the 40th percentile, on the other hand, was not able to stratify for recurrent events with statistical significance. (Fig. 3B, D).
In the time-to-event analysis of the subgroup of patients who subsequently discontinued anticoagulation (n = 97) (Fig. 4), all 9 unprovoked VTE recurrences occurred above the OCP 40th percentile cut-off, corresponding to a recurrence rate of 9.10 per 100PY (95% CI 4.74–17.49, p = 0.005) (Fig. 4A). The OHP 40th percentile was also a significant cut-off, above which all unprovoked VTE recurrences occurred, corresponding to a recurrence rate of 8.46 per 100PY (95% CI 4.40–16.25, p = 0.009) (Fig. 4B). No deaths occurred in this subgroup of patients.
Figure 5 displays the time-to-event analysis of patients who discontinued anticoagulation and were in ‘clinical equipoise’ with regards to VTE recurrence risk. This group of 34 patients comprised 25 patients following the first unprovoked major VTE, and 9 patients following a first minimally provoked major VTE. Within this group, there were 5 unprovoked recurrent VTE events, all of which occurred in patients with OCP above the 60th percentile and corresponding to a recurrence rate of 19.07 per 100 patient-years (95% CI 7.94–45.81, p = 0.018) (Fig. 5A). The OHP parameter was not able to risk stratify for unprovoked VTE recurrence with statistical significance.
A–C Kaplan–Meier curves of unprovoked VTE recurrence according to OCP in patients without long-term anticoagulation, and in ‘clinical equipoise’. A ‘clinical equipoise’ (n = 34, recurrences = 5); B after first unprovoked major VTE (n = 25, recurrences = 2); C after 1st minimally provoked major VTE (n = 9, recurrences = 3)
There were 62 patients not classified in the ‘clinical equipoise’ group, of which 3 unprovoked VTE occurred following IDDVT (n = 41), and 1 unprovoked VTE following provoked major VTE (n = 21). All 4 unprovoked recurrences occurred above a OCP cut-off of 36.25, equivalent to the 40th percentile and corresponding to a recurrence rate of 6.73 per 100 patient-years (95% CI 2.53–17.94, p = 0.058).
Figure 6 displays ROC analysis performed for D-dimer and OCP to examine their ability to predict unprovoked recurrent VTE in patients with at least 18 months follow-up from anticoagulation cessation (n = 70), and further subdivided to those following major VTE (n = 39) and IDDVT (n = 31). D-dimer performed poorly across all patient subgroups with low area under the ROC curve (AUC) (Figs. 6A–C). OCP demonstrated the best performance in the subgroup of patients following major VTE with an AUC of 0.79 (Fig. 6B).
Discussion
To the best our knowledge, this pilot study is the largest to study fibrin generation and fibrinolysis using the OHP assay in VTE patients, and the first to evaluate this assay during the acute anticoagulation period, particularly in its role to risk stratify for VTE recurrence. We demonstrate that despite being anticoagulated, patients with VTE possess higher fibrin generation capacity and reduced fibrinolytic potential compared to healthy subjects. The ability to interrogate coagulation parameters whilst anticoagulated is of practical importance and allows risk assessment without the need to cease anticoagulation, particularly in patients at high risk of VTE recurrence. This is advantageous over other global coagulation assays such as thromboelastography and thrombin generation assays, which are significantly impacted by the effect of anticoagulation.
Our findings demonstrating hypercoagulability in VTE patients are consistent with previous studies using the OHP assay in hypercoagulable disease states such as VTE, cardiovascular disease [18], pregnancy [23], vasculitis [24] and antiphospholipid syndrome [19]. Chow et al. [22] studied OHP in 67 long-term PE survivors who were no longer receiving therapeutic anticoagulation (mean sample collection 7.9 years after PE). Compared to 20 healthy controls, the mean OCP and OHP were significantly elevated, and OFP significantly lower in PE patients. Antovic et al. [16] demonstrated higher OHP and prolonged clot lysis time in 88 women with previous pregnancy-associated VTE compared with 25 healthy young women. Our study is unique, however, as we performed the assay on anticoagulated individuals.
Using time-to-event analysis, we found the 40th percentile of OCP in the entire cohort (Fig. 3) to be discriminatory for patients at higher risk of all VTE recurrences (HR 5.34, 95% CI 1.21–23.58, p = 0.027) as well as unprovoked VTE recurrence (recurrence rate 4.32/100PY, 95% CI 2.39–7.80, p = 0.002). In patients who subsequently discontinued anticoagulation, the 40th percentile for both OCP and OHP was able to stratify the risk of developing unprovoked VTE recurrence (Fig. 4). Within the ‘clinical equipoise’ subgroup in this study (n = 34), where the role of long-term anticoagulation requires further refinement particularly at an individual level, all 5 unprovoked VTE recurrences occurred above the 60th OCP percentile. Using this cut-off, 16 (47.1%) of the ‘clinical equipoise’ subgroup could be deemed low risk for unprovoked VTE recurrence and may potentially not need long-term prophylaxis. In addition, OCP was found to be superior to D-dimer at predicting unprovoked VTE recurrence, particularly in patients following an index major VTE event (ROC AUC of 0.79 vs 0.49, p = 0.037). These pilot results would suggest that the OHP assay, despite the patients being anticoagulated, may be discriminatory for unprovoked VTE recurrence risk, particularly for those in ‘clinical equipoise’.
The evidence supporting reduced fibrinolysis as a predictor of VTE recurrence has been conflicting. Other studies of plasminogen activator inhibitor-1 (PAI-1) [25, 26] and clot lysis time (CLT) [12, 26] have been mixed and have not translated to routine clinical use. The differences may be attributed to varying study designs and inclusion criteria Further study within this area is warranted. The OHP assay has the advantage of being able to simultaneously interrogate the fibrin generation and fibrinolytic pathways, with the OHP parameter being a representative of the balance between fibrin generation and lytic capacity within one individual. In this study, the OHP 40th percentile was the threshold above which all unprovoked VTE recurrences occurred within the subgroup of patients who subsequently discontinued anticoagulation.
There are several limitations to our study. Given the pilot nature of this study, the study population was relatively small. This was further compounded by a high proportion of patients (49%) who went on to indefinite anticoagulation, which may have limited the number of VTE recurrences. The high proportion of long-term anticoagulation may reflect current clinical practice following evidence supporting the use of long-term low-dose direct oral anticoagulants [27, 28]. Despite small numbers, our pilot results have been able to demonstrate that the OHP assay, particularly the OCP parameter, is able to risk stratify VTE recurrence risk and warrant validation in larger studies. The VTE subjects were significantly older (57 vs 42 years, p < 0.001) and more likely male (52.6% vs 34.7%, p < 0.001) compared to the healthy subject group. However, differences in the OHP assay results remained significant after adjusting for age and sex by multivariate analysis and these appear to discriminate for the risk of VTE recurrence despite testing occurring whilst on anticoagulation (see Table 2). Our OHP assay was modified to have a lower dose of thrombin (0.006U/mL vs 0.04U/mL) compared to the original assay developed by He et al. [14] and hence may not be directly comparable with other studies [19]. Our rationale for using the lower thrombin concentration was to enhance the sensitivity of the OHP assay to detect increased fibrin generation capacity in patients with prothrombotic tendency. The tPA concentration used was unchanged however, and in excess of physiological concentrations. This may have contributed the OHP and OFP parameters being unable to detect differences in VTE recurrence risk within the entire patient cohort and the ‘clinic equipoise’ subgroup.
In conclusion, we have demonstrated in this pilot study the ability of the OHP assay to detect a hypercoagulable and hypofibrinolytic state in anticoagulated patients following VTE, compared to healthy subjects. In this interim analysis of an ongoing study, the OHP assay was also able to discriminate the risk of VTE recurrence and suggests this assay may hold promise as an adjunct to further refine risk assessment allowing for more individualised targeting of long-term anticoagulation prophylaxis. Further long-term and larger studies evaluating fibrin generation assays are required.
References
Kearon C, Kahn SR (2020) Long-term treatment of venous thromboembolism. Blood 135:317–325. https://doi.org/10.1182/blood.2019002364
Khan F, Tritschler T, Kimpton M, Wells PS, Kearon C, Weitz JI, Büller HR, Raskob GE, Ageno W, Couturaud F, Prandoni P, Palareti G, Legnani C, Kyrle PA, Eichinger S, Eischer L, Becattini C, Agnelli G, Vedovati MC, Geersing G, Takada T, Cosmi B, Aujesky D, Marconi L, Palla A, Siragusa S, Bradbury CA, Parpia S, Mallick R, Lensing AWA, Gebel M, Grosso MA, Shi M, Thavorn K, Hutton B, Gal GL, Rodger M, Fergusson D (2021) Long-term risk of recurrent venous thromboembolism among patients receiving extended oral anticoagulant therapy for first unprovoked venous thromboembolism. J Thromb Haemost. https://doi.org/10.1111/jth.15491
Wang J, Lim HY, Ho P (2021) individualised risk assessments for recurrent venous thromboembolism: new frontiers in the era of direct oral anticoagulants. Hemato 2:64–78. https://doi.org/10.3390/hemato2010003
Bruinstroop E, Klok FA, Ree MAVD, Oosterwijk FL, Huisman MV (2009) Elevated D-dimer levels predict recurrence in patients with idiopathic venous thromboembolism: a meta-analysis. J Thromb Haemost 7:611–618. https://doi.org/10.1111/j.1538-7836.2009.03293.x
Tosetto A, Iorio A, Marcucci M, Baglin T, Cushman M, Eichinger S, Palareti G, Poli D, Tait RC, Douketis J (2012) Predicting disease recurrence in patients with previous unprovoked venous thromboembolism: a proposed prediction score (DASH). J Thromb Haemost 10:1019–1025. https://doi.org/10.1111/j.1538-7836.2012.04735.x
Tosetto A, Testa S, Martinelli I, Poli D, Cosmi B, Lodigiani C, Ageno W, Stefano VD, Falanga A, Nichele I, Paoletti O, Bucciarelli P, Antonucci E, Legnani C, Banfi E, Dentali F, Bartolomei F, Barcella L, Palareti G (2017) External validation of the DASH prediction rule: a retrospective cohort study. J Thromb Haemost 15:1963–1970. https://doi.org/10.1111/jth.13781
Eichinger S, Hron G, Kollars M, Kyrle PA (2008) Prediction of recurrent venous thromboembolism by endogenous thrombin potential and D-dimer. Clin Chem 54:2042–2048. https://doi.org/10.1373/clinchem.2008.112243
Hron G, Kollars M, Binder BR, Eichinger S, Kyrle PA (2006) Identification of patients at low risk for recurrent venous thromboembolism by measuring thrombin generation. JAMA 296:397–402. https://doi.org/10.1001/jama.296.4.397
Tripodi A, Legnani C, Chantarangkul V, Cosmi B, Palareti G, Mannucci PM (2008) High thrombin generation measured in the presence of thrombomodulin is associated with an increased risk of recurrent venous thromboembolism. J Thromb Haemost 6:1327–1333. https://doi.org/10.1111/j.1538-7836.2008.03018.x
Kearon C (2003) Natural History of Venous Thromboembolism. Circulation 107:22I – 30. https://doi.org/10.1161/01.cir.0000078464.82671.78
Rodger MA, Kahn SR, Wells PS, Anderson DA, Chagnon I, Le Gal G, Solymoss S, Crowther M, Perrier A, White R, Vickars L, Ramsay T, Betancourt MT, Kovacs MJ (2008) Identifying unprovoked thromboembolism patients at low risk for recurrence who can discontinue anticoagulant therapy. Can Med Assoc J 179:417–426. https://doi.org/10.1503/cmaj.080493
Cieslik J, Mrozinska S, Broniatowska E, Undas A (2018) Altered plasma clot properties increase the risk of recurrent deep vein thrombosis: a cohort study. Blood 131:797–807. https://doi.org/10.1182/blood-2017-07-798306
Zabczyk M, Plens K, Wojtowicz W, Undas A (2018) Prothrombotic Fibrin Clot Phenotype Is Associated With Recurrent Pulmonary Embolism After Discontinuation of Anticoagulant Therapy. Arterioscler Thromb Vasc Biol 37:365–373. https://doi.org/10.1161/atvbaha.116.308253
He S, Antovic A (2001) A simple and rapid laboratory method for determination of haemostasis potential in plasma: II. Modifications for use in routine laboratories and research work. Thromb Res 103:355–361. https://doi.org/10.1016/s0049-3848(01)00332-2
Chow V, Reddel C, Pennings G, Scott E, Pasqualon T, Ng ACC, Yeoh T, Curnow J, Kritharides L (2015) Global hypercoagulability in patients with schizophrenia receiving long-term antipsychotic therapy. Schizophr Res 162:175–182. https://doi.org/10.1016/j.schres.2014.12.042
Antovic A, Blombäck M, Bremme K, Rooijen MV, He S (2003) Increased hemostasis potential persists in women with previous thromboembolism with or without APC resistance. J Thromb Haemost 1:2531–2535. https://doi.org/10.1111/j.1538-7836.2003.00451.x
Lim HY, Lui B, Tacey M, Kwok A, Varadarajan S, Donnan G, Nandurkar H, Ho P (2021) Global coagulation assays in patients with diabetes mellitus. Res Pract Thrombosis Haemostasis 5:e12611. https://doi.org/10.1002/rth2.12611
Reddel CJ, Curnow JL, Voitl J, Rosenov A, Pennings GJ, Morel-Kopp M-C, Brieger DB (2013) Detection of hypofibrinolysis in stable coronary artery disease using the overall haemostatic potential assay. Thromb Res 131:457–462. https://doi.org/10.1016/j.thromres.2013.03.015
Curnow JL, Morel-Kopp M-C, Roddie C, Aboud M, Ward CM (2007) Reduced fibrinolysis and increased fibrin generation can be detected in hypercoagulable patients using the overall hemostatic potential assay. J Thromb Haemost 5:528–534. https://doi.org/10.1111/j.1538-7836.2007.02362.x
Lim HY, Choy KW, Wang J, Nandurkar H, Ho P (2021) Global coagulation assays—proposed reference intervals for healthy controls. Int J Lab Hematol. https://doi.org/10.1111/ijlh.13746
Miyakis S, Lockshin MD, Atsumi T, Branch DW, Brey RL, Cervera R, Derksen RHWM, Groot PGD, Koike T, Meroni PL, Reber G, Shoenfeld Y, Tincani A, Vlachoyiannopoulos PG, Krilis SA (2006) International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost 4:295–306. https://doi.org/10.1111/j.1538-7836.2006.01753.x
Chow V, Reddel C, Pennings G, Chung T, Ng ACC, Curnow J, Kritharides L (2015) Persistent global hypercoagulability in long-term survivors of acute pulmonary embolism. Blood Coagul Fibrin 26:537–544. https://doi.org/10.1097/mbc.0000000000000285
He S, Bremme K, Blombck M (1999) A Laboratory method for determination of overall haemostatic potential in plasma. I. method design and preliminary results. Thromb Res 96:145–156. https://doi.org/10.1016/s0049-3848(99)00092-4
Antovic A, Svensson E, Lövström B, Illescas VB, Nordin A, Börjesson O, Arnaud L, Bruchfeld A, Gunnarsson I (2020) Venous thromboembolism in anti-neutrophil cytoplasmic antibody-associated vasculitis: an underlying prothrombotic condition? Rheumatology Adv Pract. https://doi.org/10.1093/rap/rkaa056
Crowther MA, Roberts J, Roberts R, Johnston M, Stevens P, Skingley P, Patrassi GM, Sartori MT, Hirsh J, Prandoni P, Weitz JI, Gent M, Ginsberg JS (2001) Fibrinolytic variables in patients with recurrent venous thrombosis: a prospective cohort study. Thromb Haemostasis 85:390–394
Meltzer ME, Lisman T, de Groot PG, Meijers JCM, le Cessie S, Doggen CJM, Rosendaal FR (2010) Venous thrombosis risk associated with plasma hypofibrinolysis is explained by elevated plasma levels of TAFI and PAI-1. Blood 116:113–121. https://doi.org/10.1182/blood-2010-02-267740
Weitz JI, Lensing AWA, Prins MH, Bauersachs R, Beyer-Westendorf J, Bounameaux H, Brighton TA, Cohen AT, Davidson BL, Decousus H, Freitas MCS, Holberg G, Kakkar AK, Haskell L, van Bellen B, Pap ÁF, Berkowitz SD, Verhamme P, Wells PS, Prandoni P (2017) Rivaroxaban or aspirin for extended treatment of venous thromboembolism. N Engl J Med 376:1211–1222. https://doi.org/10.1056/nejmoa1700518
Agnelli G, Buller HR, Cohen A, Curto M, Gallus AS, Johnson M, Porcari A, Raskob GE, Weitz JI, Investigators P-E (2013) Apixaban for extended treatment of venous thromboembolism. New Engl J Medicine 368:699–708. https://doi.org/10.1056/nejmoa1207541
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This research was supported by a research grant from the Bristol-Myers-Squibb/Pfizer alliance. The sponsor had no input in the scientific conduct of the study including conception, data collection, data analysis or manuscript formulation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, J., Lim, H.Y., Brook, R. et al. Overall haemostatic potential (OHP) assay can risk stratify for venous thromboembolism recurrence in anticoagulated patients. J Thromb Thrombolysis 55, 32–41 (2023). https://doi.org/10.1007/s11239-022-02686-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-022-02686-6