Abstract
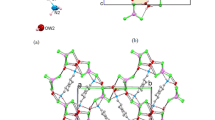
New organic-inorganic hybrid halometallates of the general formula (HpipeH2)-[M2X10] · 2H2O, where M = Sb, Bi; X = Br, I; Hpipe is homopiperazine (C5N2H12), were synthesized. The crystal structures of three new compounds, α-(HpipeH2)2[Sb2I10] · 2H2O (1), β-(HpipeH2)2[Sb2I10)]-2H2O (2), and (HpipeH2)2[Bi2Br10] · 2H2O (3), were determined and analyzed in comparison with the previously synthesized analog (HpipeH2)2[Bi2I10] · 2H2O (4). All four compounds have similar crystal structures, in which inorganic dioctahedral [M2X10]4− anions alternate with organic (HpipeH2)2+ cations and water molecules to form 3D systems based on (N)H⋯X, (N)H⋯O, and (O)H⋯X hydrogen bonds. In all structures, the (HpipeH2)2+ cation serves the same template function, forming three (N)H⋯X hydrogen bonds with halogen atoms of the inorganic anion and one (N)H⋯O bond with a water molecule. In going from Sb to Bi and from I to Br, the band gap width increases and reaches 2.89 eV for compound 3.
Similar content being viewed by others
References
Election of the Full Members (Academicians), Corresponding Members, and Foreign Members of the Russian Academy of Sciences, Russ. Chem. Bull., 2022, 71, 1559; DOI: https://doi.org/10.1007/s11172-022-3565-4.
W. L. Leong, Z. E. Ooi, D. Sabba, C. Y. Yi, S. M. Zakeeruddin, M. Graetzel, J. M. Gordon, E. A. Katz, N. Mathews, Adv. Mater., 2016, 28, 2439; DOI: https://doi.org/10.1002/adma.201505480.
Z. Zhu, C. C. Chueh, N. Li, C. Mao, A. K.-Y. Jen, Adv. Mater., 2018, 30, 1703800; DOI: https://doi.org/10.1002/adma.201703800.
Z. Shi, J. Guo, Y. Chen, Q. Li, Y. Pan, H. Zhang, Y. Xia, W. Huang, Adv. Mater., 2017, 29, 1605005; DOI: https://doi.org/10.1002/adma.201605005.
J. Shin, M. Kim, S. Jung, C. S. Kim, J. Park, A. Song, K.-B. Chung, S.-H. Jin, J. H. Lee, M. Song, Nano Res., 2018, 11, 6283; DOI: https://doi.org/10.1007/s12274-018-2151-4.
N. Dehnhardt, M. Axt, J. Zimmermann, M. Yang, G. Mette, J. Heine, Chem. Mater., 2020, 32, 4801; DOI: https://doi.org/10.1021/acs.chemmater.0c01605.
T. Y. Shao, Y. Fang, C. He, L. Zhang, K. Wang, Inorg. Chem., 2022, 61, 5184; DOI: https://doi.org/10.1021/acs.inorgchem.1c04032.
V. Morad, S. Yakunin, B. M. Benin, Y. Shynkarenko, M. J. Grotevent, I. Shorubalko, S. C. Boehme, M. V. Kovalenko, Adv. Mater., 2021, 33, 2007355; DOI: https://doi.org/10.1002/adma.202007355.
D. I. Pavlov, A. A. Rydun, D. G. Samsonenko, V. P. Fedin, A. S. Potapov, Russ. Chem. Bull., 2021, 70, 857; DOI: https://doi.org/10.1007/s11172-021-3159-6.
W. Bi, N. Leblanc, N. Mercier, P. Auban-Senzier, C. Pasquier, Chem. Mater., 2009, 21, 4099; DOI: https://doi.org/10.1021/cm9016003.
A. Piecha, A. Bialonska, R. Jakubas, J. Phys.; Condens. Matter, 2008, 20, 325224; DOI: https://doi.org/10.1088/0953-8984/20/32/325224.
A. Gągor, G. Banach, M. Węcławik, A. Piecha-Bisiorek, R. Jakubas, Dalton Trans., 2017, 46, 16605; DOI: https://doi.org/10.1039/c7dt03622a.
A. N. Usoltsev, N. A. Korobeynikov, A. S. Novikov, P. E. Plyusnin, B. A. Kolesov, V. P. Fedin, M. N. Sokolov, S. A. Adonin, Inorg. Chem., 2020, 59, 17320; DOI: https://doi.org/10.1021/acs.inorgchem.0c02599.
T. A. Shestimerova, N. A. Yelavik, A. V. Mironov, A. N. Kuznetsov, M. A. Bykov, A. V. Grigorieva, V. V. Utochnikova, L. S. Lepnev, A. V. Shevelkov, Inorg. Chem., 2018, 57, 4077; DOI: https://doi.org/10.1021/acs.inorgchem.8b00265.
T. A. Shestimerova, N. A. Golubev, N. A. Yelavik, M. A. Bykov, A. V. Grigorieva, Z. Wei, E. V. Dikarev, A. V. Shevelkov, Cryst. Growth Design, 2018, 18, 2572; DOI: https://doi.org/10.1021/acs.cgd.8b00179.
V. Yu. Kotov, A. B. Ilyukhin, A. A. Korlyukov, A. F. Smol’yakov, S. A. Kozyukhin, New J. Chem., 2018, 42, 6354; DOI: https://doi.org/10.1039/C7NJ04948J.
T. A. Shestimerova, N. A. Golubev, A. V. Grigorieva, M. A. Bykov, Z. Wei, E. V. Dikarev, A. V. Shevelkov, Russ. Chem. Bull., 2021, 70, 39; DOI: https://doi.org/10.1007/s11172-021-3054-1.
I. D. Gorokh, S. A. Adonin, A. S. Novikov, A. N. Usoltsev, P. E. Plyusnin, I. V. Korolkov, M. N. Sokolov, V. P. Fedin, Polyhedron, 2019, 166, 137; DOI: https://doi.org/10.1016/j.poly.2019.03.041.
T. A. Shestimerova, A. V. Mironov, M. A. Bykov, A. V. Grigorieva, Z. Wei, E. V. Dikarev, A. V. Shevelkov, Molecules, 2020, 25, 2765; DOI: https://doi.org/10.3390/molecules25122765.
I. Gorokh, S. Adonin, D. Samsonenko, M. Sokolov, V. Fedin, Russ. J. Coord. Chem., 2018, 44, 502; DOI: https://doi.org/10.1134/S1070328418080031.
K. Mencel, V. Kinzhybalo, R. Jakubas, J. K. Zaręba, P. Szklarz, P. Durlak, M. Drozd, A. Piecha-Bisiorek, Chem. Mater., 2021, 33, 8591; DOI: https://doi.org/10.1021/acs.chemmater.1c01266.
B. M. Benin, K. M. McCall, M. Wörle, D. Borgeaud, T. Vonderach, K. Sakhatskyi, S. Yakunin, D. Günther, M. V. Kovalenko, Chem. Mater., 2021, 33, 2408; DOI: https://doi.org/10.1021/acs.chemmater.0c04491.
N. A. Yelovik, A. V. Mironov, M. A. Bykov, A. N. Kuznetsov, A. V. Grigorieva, Z. Wei, E. V. Dikarev, A. V. Shevelkov, Inorg. Chem., 2016, 55, 4132; DOI: https://doi.org/10.1021/acs.inorgchem.5b02729.
V. Petricek, M. Dusek, L. Palatinus, Z. Kristallogr., 2014, 229, 345.
R. A. Doyle, C. R. A. Muchmore, M. Blum, Marccd Software Manual, Rayonix LLC, Evanston (IL, USA), 2011.
T. G. G. Battye, L. Kontogiannis, O. Johnson, H. R. Powell, A. G. W. Leslie, Acta Cryst., 2011, D67, 271.
P. Evans, Acta Cryst., 2006, D62, 72.
L. Krause, R. Herbst-Irmer, G. M. Sheldrick, D. Stalke, J. App. Cryst. Intern. Un. Cryst., 2015, 48, 3; DOI: https://doi.org/10.1107/S1600576714022985.
G. Sheldrick, Acta Crystallogr. C, 2015, 71, 3; DOI: https://doi.org/10.1107/S2053229614024218.
P. Kubelka, F. Munk, Z. Tech. Phys. (Leipzig), 1931, 12, 593.
S. A. Adonin, Russ. J. Struct. Chem., 2021, 62, 1345; DOI: https://doi.org/10.1134/S0022476621080126.
S. A. Adonin, I. D. Gorokh, D. G. Samsonenko, O. V. Antonova, I. V. Korolkov, M. N. Sokolov, V. P. Fedin, Inorg. Chim. Acta, 2017, 469, 32; DOI: https://doi.org/10.1016/j.ica.2017.08.058.
S. A. Adonin, I. D. Gorokh, D. G. Samsonenko, A. S. Novikov, I. V. Korolkov, P. E. Plyusnin, M. N. Sokolov, V. P. Fedin, Polyhedron, 2019, 159, 318; DOI: https://doi.org/10.1016/j.poly.2018.12.017.
S. A. Adonin, M. N. Sokolov, V. P. Fedin, Coord. Chem. Rev., 2016, 312, 1; DOI: https://doi.org/10.1016/j.ccr.2015.10.010.
C. Hrizi, A. Trigui, Y. Abid, N. Chniba-Boudjada, P. Bordet, S. Chaabouni, J. Solid State Chem., 2001, 184, 3336; DOI: https://doi.org/10.1016/j.jssc.2011.10.004.
T. A. Shestimerova, M. A. Bykov, Z. Wei, E. V. Dikarev, A. V. Shevelkov, Russ. Chem. Bull., 2019, 68, 1520; DOI: https://doi.org/10.1007/s11172-019-2586-0.
B. Wagner, F. Weigend, J. Heine, Inorg. Chem., 2021, 60, 4352; DOI: https://doi.org/10.1021/acs.inorgchem.1c00096.
T. A. Shestimerova, A. V. Shevelkov, Russ. Chem. Rev., 2018, 87, 28; DOI: https://doi.org/10.1070/RCR4762.
A. J. Dennington, M. T. Weller, Dalton Trans., 2016, 45, 17974; DOI: https://doi.org/10.1039/C6DT03602C.
B. Saparov, F. Hong, J.-P. Sun, H.-S. Duan, W. Meng, S. Cameron, I. G. Hill, Y. Yan, D. B. Mitzi, Chem. Mater., 2015, 27, 5622; DOI: https://doi.org/10.1021/acs.chemmater.5b01989.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by the Russian Foundation for Basic Research (Project No. 20-03-00280). The X-ray diffraction studies were performed using the equipment granted by the Lomonosov Moscow State University Program of Development.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Andrei Vladimirovich Shevelkov, born in 1961, Doctor of Chemical Sciences, Head of the Chair of Inorganic Chemistry of the Department of Chemistry of the Lomonosov Moscow State University, expert in the field of chemistry of inorganic functional materials, elected as a Corresponding Member of the Russian Academy of Sciences in 2022 (for more detailed information, see Ref. 1).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, Vol. 72, No. 1, pp. 167–176, January, 2023.
Rights and permissions
About this article
Cite this article
Bykov, A.V., Shestimerova, T.A., Bykov, M.A. et al. New lead-free hybrid halometallates with dioctahedral anions synthesized using the template function of homopiperazine. Russ Chem Bull 72, 167–176 (2023). https://doi.org/10.1007/s11172-023-3721-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-023-3721-5